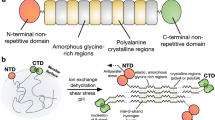
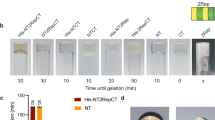
Abstract
The extreme strength and elasticity of spider silks originate from the modular nature of their repetitive proteins. To exploit such materials and mimic spider silks, comprehensive strategies to produce and spin recombinant fibrous proteins are necessary. This protocol describes silk gene design and cloning, protein expression in bacteria, recombinant protein purification and fiber formation. With an improved gene construction and cloning scheme, this technique is adaptable for the production of any repetitive fibrous proteins, and ensures the exact reproduction of native repeat sequences, analogs or chimeric versions. The proteins are solubilized in 1,1,1,3,3,3-hexafluoro-2-propanol (HFIP) at 25–30% (wt/vol) for extrusion into fibers. This protocol, routinely used to spin single micrometer-size fibers from several recombinant silk-like proteins from different spider species, is a powerful tool to generate protein libraries with corresponding fibers for structure–function relationship investigations in protein-based biomaterials. This protocol may be completed in 40 d.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Peters, H.M. Über den spinnapparat von Nephila madagascariensis (Radnetzspinnen Argiopidae). Z. Naturforsch 10, 395–404 (1955).
Lucas, F. Spiders and their silks. Discovery 25, 20–26 (1964).
Foelix, R.F. Spider webs. In Biology of Spiders, 2nd edn. (eds. Foelix, R.F.) 110–149 (Oxford University Press Inc. & Georg Thieme Verlag, New York, USA, 1996).
Akai, H. The structure and ultrastructure of the silk gland. Experientia 39, 443–449 (1983).
Sehnal, F. & Akai, H. Insect silk glands: their types, development and function, and effects of environmental factors and morphogenetic hormones on them. Int. J. Insect. Morphol. Embryol. 19, 79–132 (1990).
Kovoor, J. La soie et les glandes séricigènes des arachnides. Ann. Biol. 16, 97–171 (1977).
Kovoor, J. Chapter IV Comparative structure and histochemistry of silk producing organs in arachnids. In Ecophysiology of Spiders (ed. Nentwig, W.) 160–186 (Springer Verlag, Berlin, Heidelberg, New York, 1986).
Kovoor, J. The silk gland system in some Tetragnatinae (Aranea: Araneidae). Comparative anatomy and histochemistry. Acta Zool. Fenn. 190, 215–222 (1990).
Vollrath, F. & Knight, D. Structure and function of the silk production pathway in the spider Nephila edulis . Int. J. Biol. Macromol. 24, 243–249 (1999).
Knight, D.P. & Vollrath, F. Liquid crystals and flow elongation in a spider's silk production line. Proc. R. Soc. Lond. B 266, 519–523 (1999).
Teulé, F. Spinning from protein solutions. In Biologically Inspired Textiles, Chapter 3 (eds. Abbott, A.G. & Ellison, M.S.) 44–73 (Woodhead Publishing Ltd, Cambridge, UK, 2008).
Knight, D.P. & Vollrath, F. Changes in element composition along the spinning duct in a Nephila spider. Naturwissnschaften 88, 179–182 (2001).
Vollrath, F. & Knight, D.P. Liquid crystalline spinning of spider silk. Nature 410, 541–548 (2001).
Craig, C.L. Evolution of arthropod silks. Annu. Rev. Entomol. 42, 231–267 (1997).
Denny, M.W. The physical properties of spider silks and their role in the design of orb webs. J. Exp. Biol. 65, 483–505 (1976).
Stauffer, S., Coguill, S. & Lewis, R. Comparison of physical properties of three silks from Nephila clavipes and Araneus gemmoides . J. Arachnol. 22, 5–11 (1994).
Gosline, J.M., Denny, M.W. & DeMont, M.E. Spider silk as rubber. Nature 309, 551–552 (1984).
Gosline, J.M., Guerette, P.A., Ortlepp, C.S. & Savage, K.N. The mechanical design of spider silks: from fibroin sequence to mechanical function. J. Exp. Biol. 202, 3295–3303 (1999).
Lewis, R.V. Spider silk: ancient ideas for new biomaterials. Chem. Rev. 106, 3762–3774 (2006).
Bittencourt, D. et al. Spidroins from the Brazilian spider Nephilengys cruentata (Araneae: Nephilidae). Comp. Biochem. Physiol. 147, 597–606 (2007).
Hayashi, C.Y., Shipley, N.H. & Lewis, R.V. Hypotheses that correlate the sequence, structure, and mechanical properties of spider silk proteins. Int. J. Biol. Macromol. 24, 271–275 (1999).
Xu, M. & Lewis, R.V. Structure of a protein superfiber: spider dragline silk. Proc. Natl Acad. Sci. USA 87, 7120–7124 (1990).
Hinman, M.B. & Lewis, R.V. Isolation of a clone encoding a second dragline silk fibroin. J. Biol. Chem. 267, 19320–19324 (1992).
Hayashi, C.Y. & Lewis, R.V. Evidence from flagelliform silk cDNA for the structural basis of elasticity and modular nature of spider silks. J. Mol. Biol. 275, 773–784 (1998).
Colgin, M.A. & Lewis, R.V. Spider minor ampullate silk proteins contain new repetitive sequences and highly conserved non-silk-like 'spacer regions'. Protein Sci. 7, 667–672 (1998).
Hayashi, C.Y. & Lewis, R.V. Molecular architecture and evolution of a modular spider silk protein gene. Science 287, 1477–1479 (2000).
Hirijidia, D.H. et al. C13 NMR of Nephila clavipes major ampullate silk gland. Biophys. J. 71, 3442–3447 (1996).
Simmons, A., Michal, C. & Jelinski, L. Molecular orientation and two component nature of the crystalline fraction of spider dragline silk. Science 271, 84–87 (1996).
Teulé, F., Furin, W.A., Cooper, A.R., Duncan, J.R. & Lewis, R.V. Modifications of spider silk sequences in an attempt to control the mechanical properties of the synthetic fibers. J. Mater. Sci. 42, 8974–8985 (2007).
Brooks, A.E. et al. Properties of synthetic spider silk fibers based on Argiope aurantia MaSp2. Biomacromolecules 9, 1506–1510 (2008).
Teulé, F., Marcotte, W.R., Lewis, R.V. & Abbott, A.G. Recombinant DNA methods applied to the production of protein-based fibers as biomaterials. In Biologically Inspired Textiles, Chapter 1 (eds. Abbott, A.G. & Ellison, M.S.) 3–25 (Woodhead Publishing Ltd, Cambridge, UK, 2008).
Arcidiacono, S., Mello, C., Kaplan, D.L., Cheley, S. & Bayley, H. Purification and characterization of recombinant spider silk expressed in Escherichia coli . Appl. Microbiol. Biotechnol. 49, 31–38 (1998).
Lazaris, A. et al. Spider silk fibers spun from soluble recombinant silk produced in mammalian cells. Science 295, 472–476 (2002).
Miao, Y. et al. Expression of spider flagelliform silk protein in Bombyx mori cell line by a novel Bac-to-Bac/BmNPV baculovirus expression system. Appl. Microbiol. Biotechnol. 71, 192–199 (2006).
Zhang, Y. et al. Expression of EGFP-spider dragline silk fusion protein in BmN cells and larvae of silkworm showed the solubility is primary limit for dragline protein yield. Mol. Biol. Rep. 35, 329–335 (2008).
Prince, J.T., McGrath, K.P., DiGirolamo, C.M. & Kaplan, D.L. Construction, cloning and expression of genes encoding spider dragline silk. Biochemistry 34, 10879–10885 (1995).
Lewis, R.V., Hinman, M.B., Kothakota, S. & Fournier, M.J. Expression and purification of a spider silk protein: a new strategy for producing repetitive proteins. Prot. Expr. Purif. 7, 400–406 (1996).
Fahnestock, S.R. & Irwin, S.L. Synthetic spider dragline silk proteins and their production in Escherichia coli . Appl. Microbiol. Biotechnol. 47, 23–32 (1997).
Fukushima, Y. Genetically engineered synthesis of tandem repetitive polypeptides consisting of glycine-rich sequence of spider dragline silk. Biopolymer 45, 269–279 (1998).
Winkler, S. et al. Designing recombinant spider silk proteins to control assembly. Int. J. Biol. Macrom. 24, 265–270 (1999).
Qu, Y., Payne, S.C., Apkarian, R.P. & Conticello, V.P. Self-assembly of a polypeptide multi-block copolymer modeled on dragline silk proteins. J. Am. Chem. Soc. 122, 5014–5015 (2000).
Winkler, S., Wilson, D. & Kaplan, D.L. Controlling β-sheet assembly in genetically engineered silk by enzymatic phosphorylation/dephosphorylation. Biochem. 39, 12739–12746 (2000).
Zhou, Y., Wu, S. & Conticello, V.P. Genetically directed syntheses and spectroscopic analysis of a protein polymer derived from a Flagelliform silk sequence. Biomacromolecules 2, 111–125 (2001).
Ouroudjev, E. et al. Segmented nanofibers of spider dragline silk: atomic force microscopy and single-molecule force microscopy. Proc. Natl. Acad. Sci. 99, 6460–6465 (2002).
Fahnestock, S.R. & Bedzyk, L.A. Production of synthetic spider dragline silk protein in Pichia pastoris . Appl. Microbiol. Biotechnol. 47, 33–39 (1997).
Scheller, J., Gurhuns, K.H., Grosse, F. & Conrad, U. Production of spider silk proteins in tobacco and potato. Nat. Biotech. 19, 573–577 (2001).
Piruzian, E.S. et al. Construction of the synthetic genes for protein analogs of spider silk spidroin 1 and their expression in tobacco plants. Mol. Biol (Mosk) 27, 554–560 (2003).
Rammensee, S., Slotta, U., Scheibel, T. & Baush, A.R. Assembly mechanism of recombinant spider silk proteins. PNAS 105, 6590–6595 (2008).
Candelas, G.C. et al. Translational pauses during a spider fibroin synthesis. Biochem. Biophy. Res. Commun. 116, 1033–1038 (1983).
Candelas, G.C. et al. Features of the cell-free translation of a spider fibroin mRNA. Biochem. Cell. Biol. 67, 173–176 (1989).
Candelas, G.C. et al. Spider silk glands contain a tissue specific alanine tRNA that accumulates in vitro in response to the stimulus for silk protein synthesis. Dev. Biol. 140, 215–220 (1990).
Sambrook, J. & Russell, D.W. (eds.) Molecular Cloning: A Laboratory Manual, 3rd edn., Vol. 1–3 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA, 2001).
Lock, R.L. Process for making silk fibroin fibers. US Patent no. 5252285 (1993).
Fahnestock, S.L. Novel, recombinantly produced spider silk analogs. European Patent no. 1413585 (1994).
Fahnestock, S.R. Recombinantly produced spider silk. US Patent no. 6268169 (2001).
Birnboim, H.C. & Doly, J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 7, 1513–1523 (1979).
Costa, G.L. & Weiner, M.P. Bidirectional and directional cloning of PCR products. In PCR Primer, 2nd edn. (eds. Dieffenbach, C.W. & Dveksler, G.S.) (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA, 2003).
Ausubel, F.M. et al. (eds). Current Protocols in Molecular Biology, Vol. 1ndash;3 (John Wiley & Sons, New York, USA, 1998).
Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities utilizing the principle of protein dye binding. Anal. Biochem. 72, 248–254 (1976).
Wong, C., Sridhara, S., Bardwell, J. & Jakob, U. Heating greatly speeds coomassie blue staining and destaining. Biotechniques 28, 426–432 (2000).
Acknowledgements
These studies were funded by NSF, NIH and DOD grants awarded to the University of Wyoming and CNPQ grants awarded to the Brazilian Agricultural Research Corporation.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Teulé, F., Cooper, A., Furin, W. et al. A protocol for the production of recombinant spider silk-like proteins for artificial fiber spinning. Nat Protoc 4, 341–355 (2009). https://doi.org/10.1038/nprot.2008.250
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.250
This article is cited by
-
Replicating shear-mediated self-assembly of spider silk through microfluidics
Nature Communications (2024)
-
Thermoplastic moulding of regenerated silk
Nature Materials (2020)
-
A marine photosynthetic microbial cell factory as a platform for spider silk production
Communications Biology (2020)
-
Orb-weaving spider Araneus ventricosus genome elucidates the spidroin gene catalogue
Scientific Reports (2019)
-
Extraordinary Mechanical Properties of Composite Silk Through Hereditable Transgenic Silkworm Expressing Recombinant Major Ampullate Spidroin
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.