Abstract
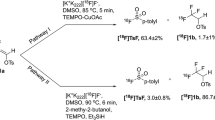
N-Succinimidyl 3-(di-tert-butyl[18F]fluorosilyl)benzoate ([18F]SiFB) is a highly reactive prosthetic group for radiolabeling of proteins for use in positron emission tomography (PET). It is similar to N-succinimidyl-4-[18F]fluorobenzoate ([18F]SFB), the 'gold-standard' prosthetic group for protein 18F-labeling, but can be synthesized using a much shorter and technically easier procedure. A recently reported simple procedure to obtain anhydrous 18F− by avoiding time-consuming azeotropic drying is applied with a slight modification to prevent basic hydrolysis of the active N-hydroxysuccinimide (NHS) ester moiety of [18F]SiFB. The labeling of [18F]SiFB is performed by a fast 18F–19F isotopic exchange (IE) reaction at room temperature (20–25 °C) within 30 min. [18F]SiFB is purified using a C18 cartridge instead of HPLC, further decreasing the overall time required for protein labeling. High specific activities >18.5 GBq μmol−1 (>500 Ci mmol−1) can be obtained. Finally, incubation of [18F]SiFB with the desired protein in an aqueous solution at pH 9, followed by HPLC purification, provides the final solution of the labeled protein ready for in vivo applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
23 October 2012
In the version of this article initially published online, Katy Orchowski's name was spelled incorrectly. The surname Orchovski has been corrected to read Orchowski. The error has been corrected in all versions of this article.
References
Vaidyanathan, G. & Zalutsky, M.R. Labeling proteins with fluorine-18 using N-succinimidyl 4-[18F]fluorobenzoate. Nucl. Med. Biol. 19, 275–281 (1992).
Vaidyanathan, G. & Zalutsky, M.R. Improved synthesis of N-succinimidyl 4-[18F]fluorobenzoate and its application to the labeling of a monoclonal antibody fragment. Bioconjug. Chem. 5, 352–356 (1994).
Wester, H.J., Hamacher, K. & Stöcklin, G.A. A Comparative study of N.C.A. Fluorine-18 labeling of proteins via acylation and photochemical conjugation. Nucl. Med. Biol. 23, 365–372 (1996).
Toretsky, J. et al. Preparation of F-18 labeled annexin V: a potential PET radiopharmaceutical for imaging cell death. Nucl. Med. Biol. 31, 747–752 (2004).
Wüst, F., Hultsch, C., Bergmann, R., Johanssen, B. & Henle, T. Radiolabelling of isopeptide Nɛ-(γ-glutamyl)-L-lysine by conjugation with N-succinimidyl-4-[18F]fluorobenzoate. Appl. Radiat. Isot. 59, 43–48 (2003).
Tang, G., Tang, X. & Wang, X. A facile automated synthesis of N-succinimidyl 4-[18F]fluorobenzoate ([18F]SFB) for 18F-labeled cell-penetrating peptide as PET tracer. J. Label Compd. Radiopharm 53, 543–547 (2010).
Cai, L., Lu, S. & Pike, V.W. Chemistry with [18F]fluoride ion. Eur. J. Org. Chem. 2008, 2853–2873 (2008).
Smith, G.E., Sladen, H.L., Biagini, C.G. & Blower, P.J. Inorganic approaches for radiolabelling biomolecules with fluorine-18 for imaging with positron emission tomography. Dalton Trans. 40, 6196–6205 (2011).
Mu, L., Schubiger, A.P. & Ametamey, S.M. [18F]fluorosilicon- and [18F]fluoroboron-based biomolecules for PET imaging. Curr. Radiopharm. 3, 224–242 (2010).
Mamat, C., Ramenda, T. & Wuest, F.R. Recent applications of click chemistry for the synthesis of radiotracers for molecular imaging. Mini-Rev. Org. Chem. 6, 21–34 (2009).
Wängler, C., Schirrmacher, R., Bartenstein, P. & Wängler, B. Click-chemistry reactions in radiopharmaceutical chemistry: fast & easy introduction of radiolabels into biomolecules for in vivo imaging. Curr. Med. Chem. 17, 1092–116 (2010).
Glaser, M. & Robins, E.G. Click labelling in PET radiochemistry. J. Label Compd. Radiopharm. 52, 407–414 (2009).
Wängler, C. et al. One-step 18F-labeling of peptides for positron emission tomography imaging using the SiFA methodology. Nat. Protoc. 7, 1946–1955 (2012).
Wessmann, S., Henriksen, G. & Wester, H.J. Cryptate mediated nucleophilic 18F−fluorination without azeotropic drying. Nuklearmedizin. 51, 1–31 (2012).
Kostikov, A.P. et al. Oxalic acid supported Si-18F radiofluorination: one step radiosynthesis of N-succinimidyl-3-(di-tert-butyl[18F]fluorosilyl)benzoate ([18F]SiFB) for protein labeling. Bioconjug. Chem. 23, 104–114 (2012).
Nikula, T.K. et al. Impact of high tyrosine fraction in complementary determining regions: measured and predicted effects of radioiodination on IgG immunoreactivity. Mol. Immunol. 32, 865–872 (1995).
Wängler, C., Moldenhauer, G., Eisenhut, M., Haberkorn, U. & Mier, W. Antibody-dendrimer conjugates: the number, not the size of the dendrimers, determines the immunoreactivity. Bioconjug. Chem. 19, 813–820 (2008).
Wängler, B. et al. Protein labeling with the labeling precursor [18F]SiFA-SH for positron emission tomography. Nat. Protoc. 7, 1964–1969 (2012).
Acknowledgements
We thank the cyclotron crew of the McConnell Brain Imaging Centre for providing fluorine-18. The financial support of the following grant agencies are gratefully acknowledged: Canada Foundation for Innovation project no. 203639 to R.S., Natural Sciences and Engineering Research Council of Canada (NSERC) Strategic Project Grant Program (STPGP) no. 412893 to R.S., Bayern-Quebec-Allianz to S.N., Deutsche Forschungsgemeinschaft (DFG) grant no. WA 2132/3-1 to B.W., and Fonds der Chemischen Industrie to C.W.
Author information
Authors and Affiliations
Contributions
A.P.K., S.N., J.C., K.O., E.S., L.I.-B., K.J. and C.W. performed the experimental work. R.S., C.W. and B.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figure 1
1H NMR spectrum of compound 1 (PDF 426 kb)
Supplementary Figure 2
13C NMR spectrum of compound 1 (PDF 445 kb)
Supplementary Figure 3
1H NMR spectrum of compound 2 (PDF 441 kb)
Supplementary Figure 4
13C NMR spectrum of compound 2 (PDF 435 kb)
Supplementary Figure 5
1H NMR spectrum of compound 3 (PDF 429 kb)
Supplementary Figure 6
13C NMR spectrum of compound 3 (PDF 428 kb)
Supplementary Figure 7
1H NMR spectrum of compound 4 (PDF 428 kb)
Supplementary Figure 8
13C NMR spectrum of compound 4 (PDF 429 kb)
Supplementary Figure 9
1H NMR spectrum of compound 5 (PDF 431 kb)
Supplementary Figure 10
13C NMR spectrum of compound 5 (PDF 439 kb)
Supplementary Figure 11
Radio-TLC of [18F]5 (PDF 485 kb)
Supplementary Figure 12
HPLC chromatogram of [18F]5-RSA conjugate (PDF 87 kb)
Rights and permissions
About this article
Cite this article
Kostikov, A., Chin, J., Orchowski, K. et al. Synthesis of [18F]SiFB: a prosthetic group for direct protein radiolabeling for application in positron emission tomography. Nat Protoc 7, 1956–1963 (2012). https://doi.org/10.1038/nprot.2012.110
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2012.110
This article is cited by
-
Using 5-deoxy-5-[18F]fluororibose to glycosylate peptides for positron emission tomography
Nature Protocols (2014)
-
One-step 18F-labeling of peptides for positron emission tomography imaging using the SiFA methodology
Nature Protocols (2012)
-
Protein labeling with the labeling precursor [18F]SiFA-SH for positron emission tomography
Nature Protocols (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.