Abstract
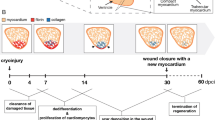
Although amphibian and fish models of heart regeneration have existed for decades, a mammalian equivalent has long remained elusive. Our discovery of a brief postnatal window for heart regeneration in neonatal mice has led to the establishment of surgical models for cardiac regenerative studies in mammals for the first time. This protocol describes a 10-min surgical procedure to induce cardiac injury in 1-d-old neonatal mice. This allows for the analysis of cardiac regeneration after surgical amputation of the left ventricle (LV) (apical resection) and coronary artery occlusion (myocardial infarction (MI)). A comparative analysis of neonatal and adult responses to myocardial injury should enable identification of the key differences between regenerative and nonregenerative responses to cardiac injury. This protocol can also be adapted to the growing repertoire of genetic models available in the mouse, and it provides a valuable tool for unlocking the molecular mechanisms that guide mammalian heart regeneration during early postnatal life.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Mosterd, A. & Hoes, A.W. Clinical epidemiology of heart failure. Heart 93, 1137–1146 (2007).
Jessup, M. & Brozena, S. Heart failure. New Engl. J. Med. 348, 2007–2018 (2003).
Sutton, M.G. & Sharpe, N. Left ventricular remodeling after myocardial infarction: pathophysiology and therapy. Circulation 101, 2981–2988 (2000).
Laflamme, M.A. & Murry, C.E. Heart regeneration. Nature 473, 326–335 (2011).
Murry, C.E., Reinecke, H. & Pabon, LM. Regeneration gaps: observations on stem cells and cardiac repair. J. Am. Coll. Cardiol. 47, 1777–1785 (2006).
Laflamme, M.A. & Murry, C.E. Regenerating the heart. Nat. Biotechnol. 23, 845–856 (2005).
Poss, K.D., Wilson, L.G. & Keating, M.T. Heart regeneration in zebrafish. Science 298, 2188–2190 (2002).
Oberpriller, J.O. & Oberpriller, J.C. Response of the adult newt ventricle to injury. J. Exp. Zool. 187, 249–253 (1974).
Mahmoud, A.I. & Porrello, E.R. Turning back the cardiac regenerative clock: Lessons from the neonate. Trends Cardiovasc. Med. 22, 128–133 (2012).
Jopling, C. et al. Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 464, 606–609 (2010).
Kikuchi, K. et al. Primary contribution to zebrafish heart regeneration by gata4+ cardiomyocytes. Nature 464, 601–605 (2010).
Porrello, E.R. et al. Transient regenerative potential of the neonatal mouse heart. Science 331, 1078–1080 (2011).
Porrello, E.R. et al. Regulation of neonatal and adult mammalian heart regeneration by the mir-15 family. Proc. Natl. Acad. Sci. USA 110, 187–192 (2013).
Jesty, S.A. et al. C-kit+ precursors support postinfarction myogenesis in the neonatal, but not adult, heart. Proc. Natl. Acad. Sci. USA 109, 13380–13385 (2012).
Gonzalez-Rosa, J.M., Martin, V., Peralta, M., Torres, M. & Mercader, N. Extensive scar formation and regression during heart regeneration after cryoinjury in zebrafish. Development 138, 1663–1674 (2011).
Egger, B., Ladurner, P., Nimeth, K., Gschwentner, R. & Rieger, R. The regeneration capacity of the flatworm Macrostomum lignano—on repeated regeneration, rejuvenation, and the minimal size needed for regeneration. Dev. Genes Evol. 216, 565–577 (2006).
Huang, N.F. et al. A rodent model of myocardial infarction for testing the efficacy of cells and polymers for myocardial reconstruction. Nat. Protoc. 1, 1596–1609 (2006).
McCall, F.C. et al. Myocardial infarction and intramyocardial injection models in swine. Nat. Protoc. 7, 1479–1496 (2012).
van Laake, L.W. et al. Monitoring of cell therapy and assessment of cardiac function using magnetic resonance imaging in a mouse model of myocardial infarction. Nat. Protoc. 2, 2551–2567 (2007).
Christensen, G., Minamisawa, S., Gruber, P.J., Wang, Y. & Chien, K.R. High-efficiency, long-term cardiac expression of foreign genes in living mouse embryos and neonates. Circulation 101, 178–184 (2000).
Phifer, C.B. & Terry, L.M. Use of hypothermia for general anesthesia in preweanling rodents. Physiol. Behav. 38, 887–890 (1986).
Kulandavelu, S. et al. Embryonic and neonatal phenotyping of genetically engineered mice. ILAR J. 47, 103–117 (2006).
Sohal, D.S. et al. Temporally regulated and tissue-specific gene manipulations in the adult and embryonic heart using a tamoxifen-inducible Cre protein. Circulation Res. 89, 20–25 (2001).
Soriano, P Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat. Genet. 21, 70–71 (1999).
Acknowledgements
We thank J. Cabrera for graphical assistance. This work was supported by the National Health and Medical Research Council and National Heart Foundation of Australia (E.R.P.), a cardiovascular research scholar award from Gilead Sciences, a Grant-in-Aid award from the American Heart Association and an NIH R01 grant (1R01HL115275-01; H.A.S.).
Author information
Authors and Affiliations
Contributions
A.I.M., E.R.P., E.N.O. and H.A.S. designed the experiments. A.I.M. and E.R.P. performed the experiments. A.I.M., E.R.P., E.N.O. and H.A.S. analyzed the data. A.I.M., W.K. and H.A.S. made the figures. A.I.M., E.R.P. and H.A.S. wrote the manuscript. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mahmoud, A., Porrello, E., Kimura, W. et al. Surgical models for cardiac regeneration in neonatal mice. Nat Protoc 9, 305–311 (2014). https://doi.org/10.1038/nprot.2014.021
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.021
This article is cited by
-
The PD-1–PD-L1 pathway maintains an immunosuppressive environment essential for neonatal heart regeneration
Nature Cardiovascular Research (2024)
-
Altered acylcarnitine metabolism and inflexible mitochondrial fuel utilization characterize the loss of neonatal myocardial regeneration capacity
Experimental & Molecular Medicine (2023)
-
A modified apical resection model with high accuracy and reproducibility in neonatal mouse and rat hearts
npj Regenerative Medicine (2023)
-
LRRC10 regulates mammalian cardiomyocyte cell cycle during heart regeneration
npj Regenerative Medicine (2023)
-
N6-methyladenosine modulates long non-coding RNA in the developing mouse heart
Cell Death Discovery (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.