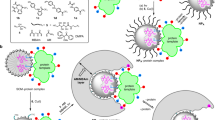
Abstract
Synthetic polymer nanoparticles (NPs) with intrinsic affinity for target biomacromolecules hold great promise in the development of novel tools for biological and biomedical research. We recently reported the design and synthesis of abiotic, synthetic polymer NPs with high intrinsic affinity for a peptide toxin melittin. The NP was selected by screening a small library of NPs (∼100 nm) composed of various ratios of monomers that contain functional groups complementary to the peptide melittin. The selected polymer NP, a co-polymer of acrylic acid (AAc), N-tert-butylacrylamide (TBAm), N-isopropylacrylamide (NIPAm) and N,N′-methylenebisacrylamide (BIS), effectively captures and neutralizes the toxicity of the peptide through a combination of electrostatic and hydrophobic interactions. This protocol describes a step-by-step procedure for the preparation and evaluation of synthetic polymer NPs for sequestration and neutralization of the target peptide toxin. The polymer NPs can be synthesized in a one-step polymerization reaction using commercially available reagents. The polymerization reaction for the synthesis of polymer NPs takes several hours, and the total protocol including subsequent purification and characterization by dynamic light scattering, NMR and toxicity neutralization assays takes 1–2 weeks in total.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kasturiratne, A. et al. The global burden of snakebite: a literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 5, 1591–1604 (2008).
Gutierrez, J., Williams, D., Fan, H. & Warrell, D. Snakebite envenoming from a global perspective: towards an integrated approach. Toxicon 56, 1223–1235 (2010).
Rainey, G. & Young, J. Antitoxins: novel strategies to target agents of bioterrorism. Nat. Rev. Microbiol. 2, 721–726 (2004).
Espino-Solis, G., Riano-Umbarila, L., Becerril, B. & Possani, L. Antidotes against venomous animals: state of the art and prospectives. J. Proteomics 72, 183–199 (2009).
Spiller, H., Bosse, G. & Ryan, M. Use of antivenom for snakebites reported to United States poison centers. Am. J. Emerg. Med. 28, 780–785 (2010).
Simpson, I. Time for an alternative perspective: the eternal problem of supply and quality of anti snake venom in the developing world—'It's the economy, stupid'. Wilderness Environ. Med. 19, 186–194 (2008).
Hoshino, Y., Kodama, T., Okahata, Y. & Shea, K.J. Peptide imprinted polymer nanoparticles: a plastic antibody. J. Am. Chem. Soc. 130, 15242–15243 (2008).
Hoshino, Y. et al. Design of synthetic polymer nanoparticles that capture and neutralize a toxic peptide. Small 5, 1562–1568 (2009).
Hoshino, Y. et al. The rational design of a synthetic polymer nanoparticle that neutralizes a toxic peptide in vivo. Proc. Natl. Acad. Sci. USA 109, 33–38 (2012).
Hu, C., Fang, R., Copp, J., Luk, B. & Zhang, L. A biomimetic nanosponge that absorbs pore-forming toxins. Nat. Nanotechnol. 8, 336–340 (2013).
Weisman, A., Chen, Y.A., Hoshino, Y., Zhang, H. & Shea, K.J. Engineering nanoparticle antitoxins utilizing aromatic interactions. Biomacromolecules 15, 3290–3295 (2014).
Yoshimatsu, K. et al. Epitope discovery for a synthetic polymer nanoparticle: a new strategy for developing a peptide tag. J. Am. Chem. Soc. 136, 1194–1197 (2014).
Mahon, C.S. & Fulton, D.A. Mimicking nature with synthetic macromolecules capable of recognition. Nat. Chem. 6, 665–672 (2014).
Hoshino, Y., Lee, H. & Miura, Y. Interaction between synthetic particles and biomacromolecules: fundamental study of nonspecific interaction and design of nanoparticles that recognize target molecules. Polym. J. 46, 537–545 (2014).
Pelton, R. & Chibante, P. Preparation of aqueous lattices with N-isopropylacylamide. Colloids Surf. 20, 247–256 (1986).
Pelton, R. Temperature-sensitive aqueous microgels. Adv. Colloid Interface Sci. 85, 1–33 (2000).
Mcphee, W., Tam, K. & Pelton, R. Poly(N-isopropylacrylamide) lattices prepared with sodium dodecyl sulfate. J. Colloid Interface Sci. 156, 24–30 (1993).
Wu, X., Pelton, R., Hamielec, A., Woods, D. & Mcphee, W. The kinetics of poly(N-isopropylacrylamide) microgel latex formation. Colloid Polym. Sci. 272, 467–477 (1994).
Ito, S. et al. Preparation of thermosensitive submicrometer gel particles with anionic and cationic charges. Langmuir 15, 4289–4294 (1999).
Debord, J.D. & Lyon, L.A. Synthesis and characterization of pH-responsive copolymer microgels with tunable volume phase transition temperatures. Langmuir 19, 7662–7664 (2003).
Armarego, W.L.F. & Chai, C.L.L. Purification of Laboratory Chemicals 6th edn. (Butterworth Heinemann, Elsevier Inc., 2009).
Haberman, E. & Zeuner, G. Comparative studies of native and synthetic melittins. Naunyn-Schmiedeberg's Arch. 270, 1–9 (1971).
Son, D. et al. Melittin inhibits vascular smooth muscle cell proliferation through induction of apoptosis via suppression of nuclear factor-kappa B and Akt activation and enhancement of apoptotic protein expression. J. Pharmacol. Exp. Ther. 317, 627–634 (2006).
Li, B. et al. Growth arrest and apoptosis of the human hepatocellular carcinoma cell line Bel-7402 induced by melittin. Onkologie 29, 367–371 (2006).
Gajski, G. & Garaj-Vrhovac, V. Melittin: a lytic peptide with anticancer properties. Environ. Toxicol. Pharmacol. 36, 697–705 (2013).
Yoshimatsu, K. et al. Temperature-responsive “catch and release” of proteins by using multifunctional polymer-based nanoparticles. Angew. Chem. Int. Ed. Engl. 51, 2405–2408 (2012).
Lee, S.-H. Engineered synthetic polymer nanoparticles as IgG affinity ligands. J. Am. Chem. Soc. 134, 15765–15772 (2012).
Yonamine, Y. et al. Polymer nanoparticle-protein interface. Evaluation of the contribution of positively charged functional groups to protein affinity. ACS Appl. Mater. Interfaces 5, 374–379 (2013).
Nayak, S. & Lyon, L.A. Soft nanotechnology with soft nanoparticles. Angew. Chem. Int. Ed. Engl. 44, 7686–7708 (2005).
Kawaguchi, H. Thermoresponsive microhydrogels: preparation, properties and applications. Polym. Int. 63, 925–932 (2014).
Islam, M., Gao, Y., Li, X. & Serpe, M. Responsive polymers for biosensing and protein delivery. J. Mater. Chem. B 2, 2444–2451 (2014).
Cedervall, T. et al. Understanding the nanoparticle-protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl. Acad. Sci. USA 104, 2050–2055 (2007).
Lindman, S. et al. Systematic investigation of the thermodynamics of HSA adsorption to N-iso-propylacrylamide/N-tert-butylacrylamide copolymer nanoparticles. Effects of particle size and hydrophobicity. Nano Lett. 7, 914–920 (2007).
Linse, S. et al. Nucleation of protein fibrillation by nanoparticles. Proc. Natl. Acad. Sci. USA 104, 8691–8696 (2007).
Cabaleiro-Lago, C. et al. Inhibition of amyloid beta protein fibrillation by polymeric nanoparticles. J. Am. Chem. Soc. 130, 15437–15443 (2008).
Cabaleiro-Lago, C., Lynch, I., Dawson, K.A. & Linse, S. Inhibition of IAPP and IAPP((20-29)) fibrillation by polymeric nanoparticles. Langmuir 26, 3453–3461 (2010).
Luchini, A. et al. Smart hydrogel particles: Biomarker harvesting: one-step affinity purification, size exclusion, and protection against degradation. Nano Lett. 8, 350–361 (2008).
Smith, M.H. & Lyon, L.A. Tunable encapsulation of proteins within charged microgels. Macromolecules 44, 8154–8160 (2011).
Smith, M.H. & Lyon, L.A. Multifunctional nanogels for siRNA delivery. Acc. Chem. Res. 45, 985–993 (2012).
Yonamine, Y., Hoshino, Y. & Shea, K.J. ELISA-mimic screen for synthetic polymer nanoparticles with high affinity to target proteins. Biomacromolecules 13, 2952–2957 (2012).
Zeng, Z. et al. Synthetic polymer nanoparticle-polysaccharide interactions: a systematic study. J. Am. Chem. Soc. 134, 2681–2690 (2012).
Beierle, J.M. et al. Polymer nanoparticle hydrogels with autonomous affinity switching for the protection of proteins from thermal stress. Angew. Chem. Int. Ed. Engl. 53, 9275–9279 (2014).
Meunier, F., Elaissari, A. & Pichot, C. Preparation and characterization of cationic poly(N-isopropylacrylamide) copolymer latexes. Polym. Adv. Technol. 6, 489–496 (1995).
Hu, X., Tong, Z. & Lyon, L. Synthesis and physicochemical properties of cationic microgels based on poly(N-isopropylmethacrylamide). Colloid Polym. Sci. 289, 333–339 (2011).
Hoshino, Y., Imamura, K., Yue, M., Inoue, G. & Miura, Y. Reversible absorption of CO2 triggered by phase transition of amine-containing micro- and nanogel particles. J. Am. Chem. Soc. 134, 18177–18180 (2012).
Hu, X., Tong, Z. & Lyon, L. Control of poly(N-isopropylacrylamide) microgel network structure by precipitation polymerization near the lower critical solution temperature. Langmuir 27, 4142–4148 (2011).
Candau, F., Leong, Y., Pouyet, G. & Candau, S. Inverse microemulsion polymerization of acrylamide—characterization of the water-in-oil microemulsions and the final microlatexes. J. Colloid Interface Sci. 101, 167–183 (1984).
Antonietti, M., Basten, R. & Lohmann, S. Polymerization in microemulsions—a new approach to ultrafine, highly functionalized polymer dispersions. Macromol. Chem. Phys. 196, 441–466 (1995).
Acknowledgements
This research was supported by the US National Science Foundation (DMR-1308363). H.K. is a recipient of the Japan Society for the Promotion of Science (JSPS) Fellowship.
Author information
Authors and Affiliations
Contributions
K.Y., H.K., Y.H. and K.J.S. developed protocols. K.Y. and H.K. contributed all the data. K.Y., H.K. and K.J.S. wrote the paper. All authors have discussed the results and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1 (PDF 88 kb)
Supplementary Data 1
1H NMR spectrum (500 MHz, CD3OD) of polymer NP synthesized by the copolymerization of 5 mol% AAc, 40 mol% TBAm, 53 mol% NIPAm, and 2 mol% BIS (values are in monomer feed ratio). (ZIP 88 kb)
Supplementary Data 2
13C NMR spectrum (126 MHz, CD3OD) of polymer NP synthesized by the copolymerization of 5 mol% AAc, 40 mol% TBAm, 53 mol% NIPAm, and 2 mol% BIS (values are in monomer feed ratio). (ZIP 225 kb)
Rights and permissions
About this article
Cite this article
Yoshimatsu, K., Koide, H., Hoshino, Y. et al. Preparation of abiotic polymer nanoparticles for sequestration and neutralization of a target peptide toxin. Nat Protoc 10, 595–604 (2015). https://doi.org/10.1038/nprot.2015.032
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2015.032
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.