Abstract
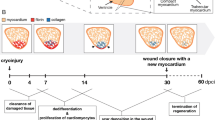
The introduction of injury models for neonatal mouse hearts has accelerated research on the mechanisms of cardiac regeneration in mammals. However, some existing models, such as apical resection and ligation of the left anterior descending artery, produce variable results, which may be due to technical difficulties associated with these methods. Here we present an alternative model for the study of cardiac regeneration in neonatal mice in which cryoinjury is used to induce heart injury. This model yields a reproducible injury size, does not induce known mechanisms of cardiac regeneration and leads to a sustained reduction of cardiac function. This protocol uses reusable cryoprobes that can be assembled in 5 min, with the entire procedure taking 15 min per pup. The subsequent heart collection and fixation takes 2 d to complete. Cryoinjury results in a myocardial scar, and the size of injury can be scaled by the use of different cryoprobes (0.5 and 1.5 mm). Cryoinjury models are medically relevant to diseases in human infants with heart disease. In summary, the myocardial cryoinjury model in neonatal mice described here is a useful tool for cardiac translational and regeneration research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Naqvi, N. et al. A proliferative burst during preadolescence establishes the final cardiomyocyte number. Cell 157, 795–807 (2014).
Wulfsohn, D., Nyengaard, J.R. & Tang, Y. Postnatal growth of cardiomyocytes in the left ventricle of the rat. Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 277, 236–247 (2004).
Li, F., Wang, X., Capasso, J.M. & Gerdes, A.M. Rapid transition of cardiac myocytes from hyperplasia to hypertrophy during postnatal development. J. Mol. Cell. Cardiol. 28, 1737–1746 (1996).
Mollova, M. et al. Cardiomyocyte proliferation contributes to heart growth in young humans. Proc. Natl. Acad. Sci. USA 110, 1446–1451 (2013).
Bergmann, O. et al. Evidence for cardiomyocyte renewal in humans. Science 324, 98–102 (2009).
Beinlich, C.J., Rissinger, C.J. & Morgan, H.E. Mechanisms of rapid growth in the neonatal pig heart. J. Mol. Cell. Cardiol. 27, 273–281 (1995).
Mahmoud, A.I., Porrello, E.R., Kimura, W., Olson, E.N. & Sadek, H.A. Surgical models for cardiac regeneration in neonatal mice. Nat. Protoc. 9, 305–311 (2014).
Porrello, E.R. et al. Transient regenerative potential of the neonatal mouse heart. Science 331, 1078–1080 (2011).
Porrello, E.R. et al. Regulation of neonatal and adult mammalian heart regeneration by the miR-15 family. Proc. Natl. Acad. Sci. USA 110, 187–192 (2013).
Haubner, B.J. et al. Complete cardiac regeneration in a mouse model of myocardial infarction. Aging 4, 966–977 (2012).
Jesty, S.A. et al. c-kit+ precursors support postinfarction myogenesis in the neonatal, but not adult, heart. Proc. Natl. Acad. Sci. USA 109, 13380–13385 (2012).
Burns, K.M. et al. New mechanistic and therapeutic targets for pediatric heart failure: report from a National Heart, Lung, and Blood Institute Working Group. Circulation 130, 79–86 (2014).
Bryant, D.M. et al. A systematic analysis of neonatal mouse heart regeneration after apical resection. J. Mol. Cell. Cardiol. 79, 315–318 (2015).
Andersen, D.C., Ganesalingam, S., Jensen, C.H. & Sheikh, S.P. Do neonatal mouse hearts regenerate following heart apex resection? Stem Cell Rep. 2, 406–413 (2014).
Konfino, T., Landa, N., Ben-Mordechai, T. & Leor, J. The type of injury dictates the mode of repair in neonatal and adult heart. J. Am. Heart Assoc. 4, e001320 (2015).
van den Bos, E.J., Mees, B.M., de Waard, M.C., de Crom, R. & Duncker, D.J. A novel model of cryoinjury-induced myocardial infarction in the mouse: a comparison with coronary artery ligation. Am. J. Physiol. Heart Circ. Physiol. 289, H1291–H1300 (2005).
Robey, T.E. & Murry, C.E. Absence of regeneration in the MRL/MpJ mouse heart following infarction or cryoinjury. Cardiovasc. Pathol. 17, 6–13 (2008).
van Amerongen, M.J., Harmsen, M.C., van Rooijen, N., Petersen, A.H. & van Luyn, M.J. Macrophage depletion impairs wound healing and increases left ventricular remodeling after myocardial injury in mice. Am. J. Pathol. 170, 818–829 (2007).
Polizzotti, B.D. et al. Neuregulin stimulation of cardiomyocyte regeneration in mice and human myocardium reveals a therapeutic window. Sci. Transl. Med. 7, 281ra245 (2015).
Darehzereshki, A. et al. Differential regenerative capacity of neonatal mouse hearts after cryoinjury. Dev. Biol. 399, 91–99 (2015).
Strungs, E.G. et al. Cryoinjury models of the adult and neonatal mouse heart for studies of scarring and regeneration. Methods Mol. Biol. 1037, 343–353 (2013).
Leferovich, J.M. et al. Heart regeneration in adult MRL mice. Proc. Natl. Acad. Sci. USA 98, 9830–9835 (2001).
Gonzalez-Rosa, J.M., Martin, V., Peralta, M., Torres, M. & Mercader, N. Extensive scar formation and regression during heart regeneration after cryoinjury in zebrafish. Development 138, 1663–1674 (2011).
Gonzalez-Rosa, J.M. & Mercader, N. Cryoinjury as a myocardial infarction model for the study of cardiac regeneration in the zebrafish. Nat. Protoc. 7, 782–788 (2012).
Schnabel, K., Wu, C.C., Kurth, T. & Weidinger, G. Regeneration of cryoinjury induced necrotic heart lesions in zebrafish is associated with epicardial activation and cardiomyocyte proliferation. PLoS ONE 6, e18503 (2011).
Chablais, F., Veit, J., Rainer, G. & Jazwinska, A. The zebrafish heart regenerates after cryoinjury-induced myocardial infarction. BMC Dev. Biol. 11, 21 (2011).
Poss, K.D., Wilson, L.G. & Keating, M.T. Heart regeneration in zebrafish. Science 298, 2188–2190 (2002).
Sadek, H.A. et al. Multi-investigator letter on reproducibility of neonatal heart regeneration following apical resection. Stem Cell Rep. 3, 1 (2014).
Sen, S. & Sadek, H.A. Neonatal heart regeneration: mounting support and need for technical standards. J. Am. Heart Assoc. 4, e001727 (2015).
Wald, R.M. et al. Effects of regional dysfunction and late gadolinium enhancement on global right ventricular function and exercise capacity in patients with repaired tetralogy of Fallot. Circulation 119, 1370–1377 (2009).
Babu-Narayan, S.V. et al. Late gadolinium enhancement cardiovascular magnetic resonance of the systemic right ventricle in adults with previous atrial redirection surgery for transposition of the great arteries. Circulation 111, 2091–2098 (2005).
Babu-Narayan, S.V. et al. Ventricular fibrosis suggested by cardiovascular magnetic resonance in adults with repaired tetralogy of Fallot and its relationship to adverse markers of clinical outcome. Circulation 113, 405–413 (2006).
Xin, M., Olson, E.N. & Bassel-Duby, R. Mending broken hearts: cardiac development as a basis for adult heart regeneration and repair. Nat. Rev. Mol. Cell Biol. 14, 529–541 (2013).
Mahmoud, A.I. et al. Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature 497, 249–253 (2013).
Xin, M. et al. Hippo pathway effector Yap promotes cardiac regeneration. Proc. Natl. Acad. Sci. USA 110, 13839–13844 (2013).
Acknowledgements
We thank H. Sadek and M. Ahmad (University of Texas Southwestern Medical Center) for training in mouse surgery, and members of the Kühn laboratory for helpful suggestions and discussions. We thank M.A. Missinato, University of Pittsburgh, for sharing her cryoinjury learning curve experience. We apologize to researchers whose relevant work could not be discussed or referenced because of the limitations of the scope of this paper. This research was supported by the Department of Cardiology and the Translational Research Program at Boston Children's Hospital and US National Institutes of Health (NIH) grants R01HL106302 and K08HL085143 (to B.K.). B.D.P. was supported by the Office of Faculty Development (Boston Children's Hospital) and by grant no. T32HL007572 from the NIH. B.G. and B.K. are supported by the Richard King Mellon Institute for Pediatric Research (Children's Hospital of Pittsburgh of UPMC). B.G., B.J.H., J.M.P. and B.K. were supported by Transatlantic Network of Excellence grants by the Fondation Leducq (no. 15CVD03 to B.G. and B.K.).
Author information
Authors and Affiliations
Contributions
B.D.P., B.G., B.J.H. and B.K. designed the research. B.D.P., B.G. and B.J.H. performed and analyzed the experiments. B.J.H. and J.M.P. provided data for Figures 4a,c and 5. B.D.P., B.G. and B.K. wrote the manuscript, and all authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Validation of the presented protocol with examination of learning curve.
The graphs show the survival at 2 hr (left panel) and 24 hr (right panel) of batches of surgery. A batch of mice operated by one researcher in a day usually consisted of 1-3 litters of ICR mice. Researcher 1 developed and optimized the protocol, while researchers 2 and 3 (with no prior mouse surgical experience) followed the suggested sequence of steps (Table 3) to learn the cardiac cryoinjury technique.
Supplementary Figure 2 Cryoinjury induces scar formation in neonatal mice.
Mice underwent sham surgery or cryoinjury on day of life 1 with a 0.5 mm probe. Scar size at 7 and 30 dpi was quantified by thresholding in Metamorph. Statistical significance was tested with analysis of variance (ANOVA) followed by Bonferroni’s multiple comparison test. *P< 0.05, **P< 0.01. Cryo, Cryoinjury.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2 (PDF 309 kb)
Rights and permissions
About this article
Cite this article
Polizzotti, B., Ganapathy, B., Haubner, B. et al. A cryoinjury model in neonatal mice for cardiac translational and regeneration research. Nat Protoc 11, 542–552 (2016). https://doi.org/10.1038/nprot.2016.031
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2016.031
This article is cited by
-
Neonatal injury models: integral tools to decipher the molecular basis of cardiac regeneration
Basic Research in Cardiology (2022)
-
A surgical mouse model of neonatal pressure overload by transverse aortic constriction
Nature Protocols (2021)
-
Parallels between vertebrate cardiac and cutaneous wound healing and regeneration
npj Regenerative Medicine (2018)
-
Relevance of mouse models of cardiac fibrosis and hypertrophy in cardiac research
Molecular and Cellular Biochemistry (2017)
-
Mechanisms of Cardiomyocyte Proliferation and Differentiation in Development and Regeneration
Current Cardiology Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.