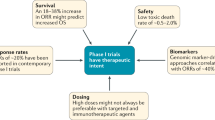
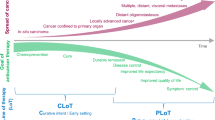
Abstract
The unprecedented pace of therapeutic development in oncology has created a climate in which the traditional methods of evaluating agent activity might no longer be adequate. How is the field transitioning to new endpoints in early drug development and what are the difficulties in this transition? Here, we will explore the historical context for the current criteria for tumour response evaluation and some of the pitfalls in using these standards when testing newer anticancer agents for activity. We will argue that the current drug development environment dictates different outcome measurements and therefore more imaginative and rigorous early-phase trial designs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Gilman, A. The initial clinical trial of nitrogen mustard. Am. J. Surg. 105, 574–578 (1963).
Farber, S., Diamond, L. K., Mercer, R. S. & Wolff, J. A. Temporary remission in acute Leukemia in children produced by folic acid antagonist, 4-Aminopteroyl-Glutamic Acid (Aminopterin). N. Engl. J. Med. 238, 787–793 (1948).
Bisel, H. F. Letter to the editor. Blood 11, 676 (1956).
Zubrod, C. G. et al. Appraisal of methods for the study of chemotherapy of cancer in man: Comparative therapeutic trial of nitrogen mustard and triethylene thiophosphoramide. J. Chronic Disease Dis. 11, 7–33 (1960).
World Health Organization. WHO Handbook for Reporting Results of Cancer Treatment Offset Publication No. 48. (WHO press, Geneva, 1979).
Gurland, J. & Johnson, R. O. How reliable are tumor measurements? JAMA 194, 973–978 (1965).
Moertel, C. G. & Hanley, J. A. The effect of measuring error on the results of therapeutic trials in advanced cancer. Cancer 38, 388–394 (1976).
Lavin, P. T. & Flowerdew, G. Studies in variation associated with the measurement of solid tumors. Cancer 46, 1286–1290 (1980).
Lavin, P. T. An alternative model for the evaluation of antitumor activity. Cancer Clin. Trials 4, 451–457 (1981).
Miller, A. B. et al. Reporting results of cancer treatment. Cancer 47, 207–214 (1981).
Tannock, I. & Murphy, K. Reflections on medical oncology: an appeal for better clinical trials and improved reporting of their results. J. Clin. Oncol. 1, 66–70 (1983).
Green, S. & Weiss, S. R. Southwest Oncology Group standard response criteria, endpoint definitions and toxicity criteria. Invest. New Drugs 10, 239–253 (1992).
James, K. et al. Measuring response in solid tumors: unidimensional versus bidimensional measurement. J. Natl Cancer Inst. 91, 523–528 (1999).
Therasse, P. et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl Cancer Inst. 92, 205–216 (2000).
Therasse, P., European Organisation for Research and Treatment of Cancer Data Center. Evaluation of response: new and standard criteria. Ann. Oncol. 13 (Suppl 4), 127–129 (2002).
Ratain, M. J., Mick R., Schilsky R. L & Siegler, M. Statistical and ethical issues in the design and conduct of phase I and II clinical trials of new anticancer agents. J. Natl Cancer Inst. 85, 1637–1643 (1993).
Von Hoff, D. D. There are no bad anticancer agents, only bad clinical trial designs — twenty-first Richard and Hinda Rosenthal Foundation Award Lecture. Clin. Cancer Res. 4, 1079–1086 (1998).
Therasse, P. et al. New guidelines to evaluate the design and conduct of phase I and II clinical trials of new anticancer agents. J. Natl Cancer Inst. 85, 205–216 (1993).
Parulekar, W. R. & Eisenhauer, E. A. Novel endpoints and design of early clinical trials. Ann. Oncol. 13 (Suppl. 4), 139–143 (2002).
Schilsky, R. L. End points in cancer clinical trials and the drug approval process. Clin. Cancer Res. 8, 935–938 (2002).
Ratain, M. J. & Eckhardt, S. J. Phase II studies of modern drugs directed against new targets: if you are fazed, too, then resist RECIST. J. Clin. Oncol. 22, 4442–4445 (2004).
Wilhelm S. & Chien D. S. Bay 43–9006: Preclinical Data. Curr. Pharm. Des. 8, 2255–2257 (2002).
Strumberg, D. et al. Phase I clinical and pharmacokinetic study of the Novel Raf kinase and vascular endothelial growth factor receptor inhibitor BAY 43–9006 in patients with advanced refractory solid tumors. J. Clin. Oncol. 23, 965–972 (2005).
Rosner, G. L., Stadler, W. & Ratain, M. J. Randomized discontinuation design: application to cytostatic antineoplastic agents. J. Clin. Oncol. 20, 4478–4484 (2002).
Ratain M. J. et al. A phase II, placebo-controlled, randomized discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. J. Clin. Oncol. (in the press).
Ratain, M. J. Phase II oncology trials: let's be positive. Clin. Cancer Res. 11, 5661–5662 (2005).
Escudier, B. et al. Randomized phase III trial of the Raf kinase and VEGFR inhibitor sorafenib (BAY 43–9006) in patients with advanced renal cell carcinoma (RCC). J. Clin. Oncol. 23, 381s (2005).
Yang, J. C. et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N. Engl. J. Med. 349, 427–434 (2003).
George, S. L. Selection bias, phase II trials, and the FDA accelerated approval process. J. Natl Cancer Inst. 95, 1351–1352 (2003).
Roberts, T. G., Lynch, T. J. & Chabner, B. A. The phase III trial in the era of targeted therapy: unraveling the 'go or no go' decision. J. Clin. Oncol. 21, 3683–3695 (2003).
Stadler, W. New trial designs to assess antitumor and antiproliferative agents in prostate cancer. Invest. New Drugs 20, 201–208 (2002).
Gandara D. R. & Gumerlock, P. H. Epidermal growth factor receptor tyrosine kinase inhibitors plus chemotherapy: Case closed or is the jury still out? J. Clin. Oncol. 23, 5856–5858 (2005).
Zia, M. I. et al. Comparison of outcomes of phase II studies and subsequent randomized control studies using identical chemotherapeutic regimens. J. Clin. Oncol. 23, 6982–6991 (2005).
Parulekar, W. R. & Eisenhauer E. A. Novel endpoints and design of early clinical trials. Ann. Oncol. 13 (Suppl. 4), 139–143 (2002).
Schilsky, R. L., End points in cancer clinical trials and the drug approval process. Clin. Cancer Res. 8, 935–938 (2002).
Mack, G. S. Genta sued over NDA withdrawal. Nature Biotechnol. 22, 788–789 (2004).
Rubinstein, L. V. et al. Design issues of randomized phase II trials and a proposal for phase II screening trials. J. Clin. Oncol. 23, 7199–7206 (2005).
Stadler, W. New trial designs to assess antitumor and antiproliferative agents in prostate cancer. Invest. New Drugs 20, 201–208 (2002).
Stadler, W. M. et al. Successful implementation of the randomized discontinuation trial design: an application to the study of the putative antiangiogenic agent carboxyaminoimidazole in renal cell carcinoma–CALGB 69901. J. Clin. Oncol. 23, 3726–3732 (2005).
Center for Drug Evaluation and Research, Guidance for Industry. General Considerations for the Clinical Evaluation of Drugs p6 [online], http://www.fda.gov/cder/guidance/old034fn.pdf.(1997).
Food and Drug Administration. Challenge and Opportunity on the Critical Path to New Medical Products [online], http://www.fda.gov/oc/initiatives/criticalpath/whitepaper.html#execsummary (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
National Cancer Institute
Rights and permissions
About this article
Cite this article
Michaelis, L., Ratain, M. Measuring response in a post-RECIST world: from black and white to shades of grey. Nat Rev Cancer 6, 409–414 (2006). https://doi.org/10.1038/nrc1883
Issue Date:
DOI: https://doi.org/10.1038/nrc1883
This article is cited by
-
Support to early clinical decisions in drug development and personalised medicine with checkpoint inhibitors using dynamic biomarker-overall survival models
British Journal of Cancer (2023)
-
Comparison of [18F]FIMP, [11C]MET, and [18F]FDG PET for early-phase assessment of radiotherapy response
Scientific Reports (2023)
-
Vaccination with poly(IC:LC) and peptide-pulsed autologous dendritic cells in patients with pancreatic cancer
Journal of Hematology & Oncology (2017)
-
Second-line pazopanib in patients with relapsed and refractory small-cell lung cancer: a multicentre phase II study of the Hellenic Oncology Research Group
British Journal of Cancer (2017)
-
Perspective on the interpretation of research and translation to clinical care with therapy-associated metastatic breast cancer progression as an example
Clinical & Experimental Metastasis (2017)