Key Points
-
Nuclear morphology is often altered in cancer. Irregularity in nuclear contours is a feature used by pathologists in diagnostic cytology.
-
The nuclear envelope provides a specialized microenvironment within the nucleus. It is also the site where a series of protein–protein interactions take place to connect the cytoskeleton to the interior of the nucleus.
-
Constituents of the nuclear envelope — lamins, nuclear pore complexes and lipid membranes — have key roles in several processes that affect tumour cell biology and response to therapy.
-
Changes in the abundance or the function of components of the nuclear envelope in tumour cells can cause the occurrence of dysmorphic nuclei and can deregulate cell migration, intracellular signalling, DNA repair, cell division and gene expression.
-
Tracking the abundance of molecular components of the nuclear envelope environment and enhanced methods to visualize changes at the nuclear envelope could potentially be used for the prognostic assessment of cancer patients.
Abstract
Because of the association between aberrant nuclear structure and tumour grade, nuclear morphology is an indispensible criterion in the current pathological assessment of cancer. Components of the nuclear envelope environment have central roles in many aspects of cell function that affect tumour development and progression. As the roles of the nuclear envelope components, including nuclear pore complexes and nuclear lamina, are being deciphered in molecular detail there are opportunities to harness this knowledge for cancer therapeutics and biomarker development. In this Review, we summarize the progress that has been made in our understanding of the nuclear envelope and the implications of changes in this environment for cancer biology.
Similar content being viewed by others
Main
Tumour cells are often distinguished by the occurrence of a lobulated 'polymorphic' nuclear envelope with a range of abnormal nuclear grooves and chromatin configurations1,2 (Fig. 1). There is growing appreciation that a wide range of cellular activities that are highly relevant to tumorigenesis are dependent on the composition and organization of the nuclear envelope. Certainly, changes at the nuclear envelope correspond to functional alterations that affect the behaviour and the phenotype of the tumour cell. Thus, changes in the morphology of the nuclear envelope in tumour cells may directly contribute to tumorigenesis.
Although tumours of different types show distinct morphological features, enlarged nuclei are characteristic of many cancer cells (compare the area indicated by square brackets in normal breast duct (part a) to invasive ductal carcinoma (part b)). At least in some cases, this increase in nuclear volume corresponds to aberrantly increased chromosome content with concomitant expansion of the nuclear periphery. Another hallmark of many cancer cells that is observed using haematoxylin and eosin staining is irregularity in nuclear contour, in the form of both indentations and protrusions. The invasive ductal breast carcinoma in part b shows an atypical invagination of the nucleus (indicated by an arrow). Changes in the appearance of chromatin, both at the nuclear periphery and in the nucleus interior — evident in part b — are also important criteria in diagnostic cytology. Nuclear grooves are characteristic of papillary thyroid cancer and their presence contributes to diagnosis (indicated by arrows in part c; this was imaged using a Papanicolaou stain of fine-needle aspirate). The enhancement of detection that is made possible with the use of immunofluorescence is illustrated in the comparison of haematoxylin and eosin staining (part d) versus immunofluorescence of lamin B (part e) in high-grade breast cancer. Changes to nuclear morphology indicate that cells may be neoplastic, but it is important to bear in mind that nuclear morphology is only one aspect of pathological assessment, which includes clinical context and often uses ancillary tests, such as immunohistochemistry, flow cytometry and molecular tests for the assessment of cellular aberrancies that are not detectable by light microscopy. Part d and part e are reproduced, with permission, from Ref. 153 © (2008) Romanian Academy Publishing House.
The main features of the nuclear envelope environment are the nuclear membrane, along with its range of integral membrane proteins and associated proteins — including, lamins (Box 1) and the nuclear pore complexes (NPCs)3,4 (Box 2). The nuclear membrane is comprised of two lipid bilayers, the inner nuclear membrane (INM) and the outer nuclear membrane (ONM), and it creates a barrier that allows genomic DNA to reside in surroundings that are dedicated to its protection, faithful replication and regulated transcription. The INM establishes further functional domains at the nuclear periphery by housing particular integral membrane proteins that in turn associate with lamins and a host of other proteins. NPCs are embedded within the nuclear membranes and provide a gateway for nucleocytoplasmic trafficking that is crucial to the unique composition of the nucleoplasm. NPCs, as well as other elements of the nuclear envelope, are dramatically remodelled during mitosis. The fidelity of this disassembly–assembly process is essential for the maintenance of proper nuclear architecture as cells proliferate. Furthermore, in many cases, components of the nuclear envelope and NPCs actively participate in mitotic events when released from their interphase organization5. A detailed knowledge of these mitotic roles is essential to understand the deregulated cell division that occurs in cancer and to provide additional insight into processes that are prone to defects together with cancer-associated alteration of nuclear envelope morphology.
Research into the nuclear envelope has recently been spurred on by the link that has been discovered between alterations in proteins of the nuclear envelope (including, lamins, emerin, nesprin 1 (also known as SYNE1), nesprin 2 (also known as SYNE2), lamin B receptor (LBR), the INM protein MAN1 (also known as LEMD3) and torsin 1A) and a variety of human diseases outside the realm of cancer6. Indeed, the phenotypes of such human diseases, ranging from rapid ageing to muscle dysfunction, have played a large part in revealing the far-reaching roles of the nuclear envelope and its components. Moreover, the tissue-specific nature of the disease phenotypes that are associated with mutation of ubiquitous nuclear envelope components has underscored the importance of cellular context, which is also a very important consideration in cancer. Although much of the current emphasis has been on deciphering the aetiology of these specific and often devastating diseases, this recent work also sheds new light on how cancer-associated changes in nuclear morphology may affect tumour phenotype. Thus, in this Review, we highlight relevant recent advances that have connected the nuclear envelope to cell migration, cell division, gene expression and the control of signalling. We also consider how the investigation of the constituents of the nuclear envelope in cancer lends insight into molecular changes in the nuclear envelope environment and reveals new strategies for tumour biomarkers.
The nuclear envelope and cell motility
In cancer, dynamics in cell migration and adhesion are particularly important to the process of metastasis, in which cancer cells escape from the primary tumour and enter the vasculature (intravasation); travel to distant sites; and extravasate to invade a new niche. A change to any key structural element of the nucleus has the potential to affect cell migration simply by increasing the plasticity of nuclear shape and by allowing changes to cell form that are required for squeezing the cell through restricted spaces7. This is illustrated by the case of neutrophils, in which low levels of lamins and other nuclear envelope structural components result in a lobulated nucleus that is adapted to further deformation that can be encountered as the neutrophil carries out its functions, such as during egress from blood vessels8.
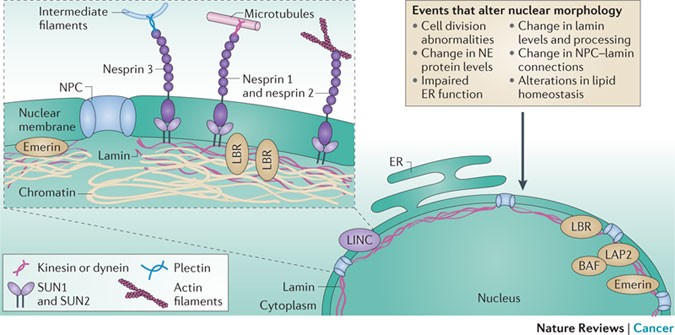
A growing appreciation for the existence of a protein complex that spans across the nuclear envelope, referred to as the linker of nucleoskeleton and cytoskeleton (LINC) complex9,10 (Fig. 2), has substantially added to the conceptual framework for understanding the role of nuclear envelope-associated proteins in cell migration7. This complex involves interactions between the nuclear lamina and integral membrane proteins of the INM, between luminal domains of INM and ONM proteins, and between regions of ONM proteins that face the cytoplasm and components of the cytoskeleton. Experimental manipulation of components of the LINC complex and associated proteins, such as emerin, affects perinuclear actin organization, nuclear shape, nuclear rigidity and positioning of the centrosome with respect to the nucleus7,11,12. Cancer-associated alterations in the expression of emerin13, lamin A and lamin B (Table 1) probably perturb a similar range of features. In turn, the concomitant alterations in nuclear plasticity, cell polarity cues and cell movement may contribute to the propensity for invasion and metastasis. The importance of cellular tension and tissue stiffness in tumorigenesis14 further highlights the importance of studying the function of the LINC complex, and its role in sensing and in mediating responses to extracellular force, in the context of cancer. Interestingly, nuclear deformation through the disruption of the LINC complex can be uncoupled from changes in the expression of genes, such as early growth response 1 (Egr1) and immediate early response 3 (Ier3; also known as Iex1), that are induced by mechanosensitive signalling in mouse embryonic fibroblasts15, which underscores the complexity of the coordinated response to changes in extracellular forces.
Two lipid bilayers surround the nucleus and are separated by a luminal domain that is contiguous with the endoplasmic reticulum (ER). The inner nuclear membrane (INM) and the outer nuclear membrane (ONM) are joined in a highly curved membrane domain at the site of nuclear pore complexes (NPCs)181 (Box 2). The NPCs create selective channels that function together with soluble receptors to guide protein and RNA trafficking between the nucleus and cytoplasm4. The nuclear lamina, a protein meshwork that is primarily comprised of lamin proteins (Box 1), underlies the INM. This area is a hub for protein–protein interactions that link chromatin to the nuclear periphery, lamina to nuclear pores and residents of the INM to the peripheral environment of the nuclear interior3,82. Just as important to nuclear structure and function is a series of interactions that create connectivity between the periphery of the nucleoplasm and the cytoskeleton. Specifically, the luminal domain of SUN domain-containing protein (SUN) family members residing in the INM interact with luminal domains of Klarsicht, ANC1, Syne homology (KASH) family members that are resident in the ONM10. In turn, the cytoplasmic domains of KASH proteins contain specific sites of contact for actin, microtubules and intermediate filaments. This network of interactions has been termed the linker of nucleoskeleton and cytoskeleton (LINC) complex. LINC complex proteins also have a role in tethering centrosomes to the near vicinity of the ONM. Invaginations of the nuclear membrane, which are often observed in tumour cells, have been characterized more broadly and are referred to as the nucleoplasmic reticulum. More recently, these were further classified into type I invaginations, which affect the INM only, and type II invaginations, which involve the INM and the ONM together182. Many processes and factors influence nuclear morphology (referred to in the box). BAF, barrier to autointegration factor; LAP2, lamina-associated polypeptide 2; LBR, lamin B receptor; NE, nuclear envelope.
Components of the nuclear envelope affect cell migration, and its balance with adhesion, in additional ways beyond the paradigms of nuclear structure and physical connectivity between the nucleus and the cytoplasm. For example, the overexpression of lamin A, which increased the motility of a colon cancer cell line, resulted in the upregulation of the actin-binding protein T-plastin (also known as PLS3) and the downregulation of E-cadherin, and cytoskeletal and adhesion molecules that are involved in regulating cell migration16. The depletion of the nucleoporin NUP153 leads to alterations in the organization of A-type lamins and to alterations in the localization of the INM protein SUN domain-containing protein 1 (SUN1), and this is accompanied by impaired cell migration17. Determining whether a role for NUP153 in the integrity of the LINC complex underlies defective migration is worth further investigation.
However, NUP153 — as well as other nucleoporins — may contribute in additional ways to cell motility, as nucleoporins interact with important players in cell migration17,18,19,20,21,22,23. For example, NUP358 (also known as RANBP2) is reported to form a cooperative interaction with kinesin 2 to regulate the localization of adenomatous polyposis coli (APC) to the cell cortex19. This pore protein has also been implicated in regulating RAP family GTPases by binding a guanine nucleotide exchange factor, exchange protein directly activated by cyclic AMP 1 (EPAC1; also known as RAPGEF3). The depletion of NUP358 was found to enhance cAMP-induced cell adhesion in OVCAR3 ovarian cancer cells, which is consistent with biochemical characterization showing that NUP358 inhibits EPAC1 from activating RAP1B22. Collectively, these observations expand the functional horizon of nuclear pore proteins beyond serving as the gateway to the nucleus. Moreover, spatial organization is often a crucial aspect of the roles for nuclear envelope components in regulating cell migration and adhesion. For these reasons, it will be important to track not only changes in the expression of nuclear envelope components, but also changes in nuclear envelope organization, to better understand the relationship between the nuclear envelope and the capacity for tumour dissemination and invasiveness.
The nuclear envelope and fidelity of cell division
NPCs have important roles in cell division in the canonical context of nucleocytoplasmic trafficking by restricting the nuclear entry and exit of key mitotic regulators, such as p27 (also known as KIP1) and cyclin B24,25, as well as by directing selective export of mRNAs that encode cell cycle regulators26. The NPC-associated sentrin-specific protease 1 (SENP1)27 is also reported to influence cell cycle progression by regulating the expression of cyclin-dependent kinase (CDK) inhibitors28; and lamin A and lamina-associated polypeptide 2α (LAP2α) actively participate in cell cycle control by regulating RB-mediated transcriptional control of target genes3,29 (discussed below). Moreover, the nuclear envelope, including the underlying lamina and the NPCs, undergoes dramatic remodelling during each cell division: signalling in mitosis leads to nuclear envelope disassembly, and, following chromosome segregation, to coordinate reformation30. Errors in this process can contribute to various alterations ranging from aneuploidy to defective nuclear structure that have the potential to alter cellular function. There is now evidence for surveillance mechanisms of correct post-mitotic reformation of NPCs. Specifically, disrupting the assembly of the basket-like feature on the nuclear face of the NPC resulted in persistent aurora kinase B activation and delayed the completion of cytokinesis31. This role for aurora kinase B is probably deregulated in tumours in which aurora kinase B is overexpressed or, conversely, in which this kinase is inhibited by chemotherapy (for example, AZD1152, GSK1070916A and other compounds in clinical trials)32. Mechanisms that are involved in cell cycle-associated remodelling of the nuclear envelope are also important to consider for their potential to reveal new chemotherapeutic targets, as blocking a step in this process could provide a novel strategy to stop cell proliferation.
During interphase, emerin, nesprins and A-type lamins have roles in coordinating the anchorage of centrosomes to the ONM9. This contributes to microtubule organization and cellular orientation, which is important for migration, but probably also affects the central role of centrosomes in organizing and positioning the mitotic spindle. Notably, treatment with farnesyltransferase inhibitors (FTIs) results in defective centrosome separation, which causes profound aberrations in mitosis. Preventing lamin B1 farnesylation (Box 1) was shown to specifically contribute to this phenotype33. It is plausible that defects in targeting lamin B1 to nuclear membranes and consequently deficient integration of lamin B1 into the LINC complex affects centrosome separation, but this has not yet been investigated. Although FTIs are in clinical development for the treatment of cancer, the germane downstream targets have been elusive since it was discovered that oncogenic RAS mutants are not the targets, as originally intended34. A connection between FTIs, lamin B1 and impaired cell division may provide a crucial clue in the effort to understand the relevant targets and to further develop this therapeutic strategy.
At the G2/M cell cycle transition, two nucleoporins have roles in tethering centrosomes to the nuclear envelope35,36. During prophase, these interactions are thought to contribute to the role of microtubules in nuclear envelope breakdown30 and probably also to re-position sister centrosomes to opposite sides of the nucleus35,36. The importance of coordinating these events has been illustrated by a recent observation that cells in which centrosomes do not separate fully before nuclear envelope breakdown have a greater frequency of mis-segregated chromosomes37. The NPC additionally contains factors with important regulatory roles during mitosis that are released when the NPC disassembles at the end of prophase. For example, NPC-associated SUMO enzymes, such as the SUMO E3 ligase NUP358 and the SUMO proteases SENP1 and SENP2, are implicated in the sumoylation and the desumoylation of several mitotic factors38,39,40,41. The sumoylation of proteins such as topoisomerase IIα and CENPE is thought to regulate their localization at mitosis38,39. Targeting SENP2 to the NPC has been shown to restrain its catalytic activity42, which suggests a paradigm for the importance of this localization for both SENP1 and SENP2. This further implies that overexpression of these SENPs may override such spatial control of their activity. Interestingly, premature desumoylation of topoisomerase IIα by SENP1 or SENP2, which could either impair targeting of topoisomerase IIα to inner centromeres38 or upregulate its decatenation activity43, is proposed to lead to aneuploidy44, which hints at a possible pathogenic mechanism when SENP1 is overexpressed in cancers45,46.
A newly discovered complex between the mitotic regulator transforming acidic coiled-coil-containing protein 3 (TACC3) and the tumour suppressor tuberin (which is encoded by TSC2) was found to reside at the nuclear envelope partly through an interaction of TACC3–tuberin with NUP62 (Ref. 47). Deficiency of either TSC2 or TACC3 in mouse embryonic fibroblasts results in strikingly aberrant nuclear morphology; however, further investigation is needed to determine whether this is due to the disruption of a direct role at the nuclear envelope or whether it is a consequence of aberrancies that arise in mitosis47. If it is a result of aberrations that arise in mitosis, then perhaps NUP62 serves to sequester, rather than direct, the activity of TACC3–tuberin. Interaction with the nuclear pore can both restrict the spatial and temporal activity of mitotic regulators, and poise them for action. This is illustrated by knockdown of the nucleoporin translocated promoter region (TPR), which is a binding partner of the mitotic spindle assembly checkpoint proteins MAD1 and MAD2, which resulted in diminished recruitment of MAD1 tokinetochores and a compromised spindle assembly checkpoint in HeLa cells48.
The roles of nuclear envelope proteins also extend to the establishment of the mitotic spindle apparatus and chromosome segregation. Although these are not functions of the nuclear envelope per se, they are important to consider here, as a change in the nuclear periphery will often lead to a change in the fidelity of chromosome inheritance, which is a common feature of cancer cells that contributes to tumour phenotype and, at least in some cases, to tumorigenesis49. There are now many well-characterized examples of NPC components that have roles during mitosis. This includes roles for NUP358 at kinetochores and for NUP98 in regulating the APC/C (anaphase promoting complex; also known as the cyclosome)5 and in regulating mitotic microtubule dynamics to promote spindle assembly50. NUP88, which is frequently overexpressed in cancers, as well as TPR, which is found in oncogenic translocations51, have also been proposed to participate in proper mitotic spindle function5. Mice with reduced expression of NUP358 or of RNA export 1 homologue (RAE1), which is a binding partner of NUP98, have chromosome segregation defects, severe aneuploidy and are susceptible to carcinogen-induced tumour formation38,52,53. Although aneuploidy is likely to be a contributing factor, its relationship to tumorigenesis is complex, with reduced expression of NUP358, but not of RAE1, leading to spontaneous tumours in mice and the simultaneous reduction of NUP98 expression with RAE1 increasing aneuploidy but not tumour susceptibility52. The multifunctional role of these nucleoporins clearly creates individual profiles of deregulation when their expression levels are altered, culminating in different outcomes.
Interestingly, remnants of the nuclear membrane itself, along with associated proteins, are also emerging as active participants in mitosis54,55. A role in regulating spindle morphogenesis for the nuclear membrane and associated proteins may explain why the downregulation of emerin leads to cytokinesis failure and polyploidy13 or this may perhaps relate to the connection between the effects of FTIs and lamin B1 (Ref. 33). In either of these cases, decreased levels or mistargeting of nuclear membrane-associated proteins may impair a direct role in chromosome segregation, resulting in lagging chromosomes that in turn prevent proper progression through cytokinesis. Although attributing phenotype to a specific role is difficult — as is the case with many multifunctional proteins — it is evident that proteins of the nuclear periphery contribute at multiple levels to coordinating cell division. Indeed, a screen of transmembrane nuclear envelope proteins for roles in regulating the cell cycle has recently revealed eight novel candidates, reinforcing the importance of delving further into this link56. It is clear that cancer-associated changes in the composition and/or morphology of the nuclear periphery have the potential to create mitotic defects and that such errors can contribute to chromosome instability, which is often seen in tumours. Mis-coordinated chromosome segregation, however, can also lead to the formation of lobulated, malformed nuclei57. Therefore, even low-frequency errors in chromosome segregation may be further reinforced and augmented over time by altering nuclear envelope-dependent cell cycle events.
The nuclear envelope and gene expression
The nuclear envelope environment serves as a scaffold for numerous transcription factors (including, receptor-regulated SMADs (R-SMADS), sterol regulatory element-binding protein 1 (SREBP1), FOS and OCT1 (also known as POU2F1)), chromatin remodelers (for example, CREB-binding protein (CBP; also known as CREBBP) and histone deacetylase 3 (HDAC3)) and regulators of transcription (such as RB, inhibitor of growth protein 1 (ING1), germ cell-less (GCL; also known as GMCL1), protein phosphatase 1 (PP1), PP2, SENP1, SENP2 and ubiquitin-conjugating enzyme 9 (UBC9))3,27,58,59. Coordinated nuclear import, sequestration, post-translational modification and release of these proteins into the nucleoplasm are integrated at the nuclear envelope to regulate mRNA expression60. Similarly, the nuclear periphery — lamin B1 in particular — also modulates the expression of microRNAs (miRNAs)61. This additional layer of control is especially noteworthy as one of the most affected miRNAs in lamin B1 (Lmnb1)Δ/Δ cells, miR-31, is thought to have important roles in cancer. For example, miR-31 expression is elevated in colorectal cancer and its expression is correlated with more advanced stage of disease62. In the context of breast cancer, however, miR-31 expression was found to inversely correlate with metastasis, and experimentally inducing its expression in xenograft models of metastasis caused the regression of metastatic lesions63,64. The pleiotropic targets of this miRNA probably make the context of its expression crucial, but overall these observations highlight the importance of the connection between miRNA expression and lamin B1.
Nucleoporins take part in the regulated transport of transcription factors, sometimes in a direct manner, as has been described for the transforming growth factor-β (TGFβ)-responsive transcription factor SMAD2 (Ref. 65). Transcription factor export from the nucleus can also be a key step in the regulation of gene expression; thus, nucleoporins that interface with export machinery make important contributions in this regard. The cytoplasmic aggregates of NUP88 that are often seen when this nucleoporin is overexpressed in tumours could divert CRM1 (also known as EXP1) from its normal interactions at the pore, thereby impairing export-mediated downregulation of transiently activated transcription factors such as nuclear factor-κB (NF-κB)66,67.
Nucleoporins are also intimately connected to chromatin, as they take part in regulating transcription68,69,70. There is evidence for two classes of nucleoporin–chromatin interactions: those that take place at the NPC and those that occur in the nucleoplasm with a dynamic population of nucleoporins70. Although details of the determinants of nucleoporin–chromatin association and the exact roles of nucleoporins at this site are still unknown, one pathway that is emerging as a modulator of the connection between nucleoporins and chromatin in mammalian cells is protein acetylation. Histone deacetylase (HDAC) inhibitors were shown to alter NUP93-associated chromosome regions71, and, in cardiomyocytes, HDAC4 was reported to counteract the association of nucleoporins that are reactive with the monoclonal antibody mAb414 (which primarily recognizes NUP358, NUP214, NUP153 and NUP62) and certain target genes72. Moreover, NUP155 was discovered to be a partner of HDAC4, and this interaction was found to be important for the inhibitory effect of HDAC4 on the expression of these target genes. A deeper understanding of the interplay between nucleoporins and HDACs is relevant to the application of HDAC inhibitors as a cancer therapy73.
Although there are roles for mobile, nucleoplasmic nucleoporins in regulating transcriptional activity at interphase, the nuclear periphery remains an important environment for the regulation of gene expression74,75,76,77. In Drosophila melanogaster and human cells, A-type lamins and NPCs have been reported to possess boundary activity, which probably allows the coexistence of both transcriptionally permissive and repressive chromatin configurations at the nuclear periphery78,79. Experimental depletion of lamin B1 in mammalian cells results in blebs of the nuclear envelope that are enriched with A-type lamin and are, moreover, where gene-rich DNA tends to be concentrated. Notably, these lamin-defined microdomains are not, however, associated with active transcription, but rather they appear to contain stalled RNA polymerase II (Ref. 76). Nuclear blebs with similar features have recently been reported in prostate cancer (see below)80. These and other observations underscore that changes at the nuclear envelope and in its morphology are likely to be accompanied by alterations in gene expression. Such transcriptional deregulation may in some cases be a protective adaptation, but it also holds the potential of contributing to the progression from normal to tumorigenic cell growth.
The nuclear envelope and signalling pathways
Many signalling molecules associate with the nuclear envelope, which plays a part in coordinating multiple signalling pathways, including those downstream of TGFβ, insulin-like growth factor (IGF), epidermal growth factor (EGF), WNT and NOTCH3,81,82. These signalling pathways are often deregulated in cancer, leading to aberrant cell proliferation that is uncoupled from normal cues and control; changes at the nuclear envelope have the capacity to play a part in this deregulation.
Components of the nuclear periphery can differentially modulate signalling pathways. This point is exemplified by the canonical WNT signalling pathway, which leads to the stabilization and translocation of β-catenin to the nucleus, where it partners with members of the T cell factor (TCF)/lymphoid enhancer factor (LEF) family of transcription factors to activate specific genes. Whereas nesprin 2 promotes the nuclear localization of β-catenin, emerin seems to have the opposite effect83,84. The specific context may be important for these differing roles, but it is evident that perturbing the expression of these nuclear envelope proteins leads to defective WNT signalling and severely disrupts cellular proliferation and differentiation programmes60,83,85. The regulation of WNT signalling by the nuclear envelope is further highlighted by evidence that additionally implicates the sumoylation of transcription factor 4 (TCF4) — which is stimulated by NUP358 and counteracted by SENP2 — in promoting TCF4–β-catenin interaction and transcriptional activity86.
Extracellular signalling can also lead to functional alterations at the nuclear envelope. NPC density can change in response to hormonal stimulation87,88, according to the phase of the cell cycle89,90 and in response to cell signalling91. One pathway that illustrates the complex crosstalk between extracellular signals and NPCs is the MAPK–ERK pathway. Following mechanical stimulation that is induced by cell stretching, MAPK signalling is activated and leads to increased numbers of NPCs91. Activation of this pathway can also lead to functional modulation of nucleoporins. NUP50, NUP153 and NUP214 were found to be phosphorylated by ERK, and their interaction with the soluble transport receptor importin-β is decreased in the presence of active ERK, which is concomitant with a decreased ability of the receptor to accumulate in the nucleus92. This indicates that ERK negatively regulates importin-β-dependent import into the nucleus; exactly how this shift in homeostasis would affect tumour biology in the case in which ERK signalling is activated is not yet clear. In the case of TPR, phosphorylation by ERK2 stabilizes interaction with this kinase, which perhaps allows TPR to serve as a scaffold for further phosphorylation events93. In addition, direct interaction of activated ERK1–ERK2 with TPR, as well as NUP153, is thought to promote nuclear translocation of activated ERK1–ERK2, independently of soluble trafficking receptors93,94,95. Enhanced nuclear entry of ERK1–ERK2 in cancer cells versus cells cultured from normal tissue has been proposed to result from differences in nucleoporin levels. Experimentally decreasing levels of NUP153 in the ovarian cancer cell lines OVCAR-10 and SKOV3 attenuated the expression of FOS, which is a downstream target of the ERK pathway, without altering levels of activated ERK96.
Components of the INM are also implicated in ERK1–ERK2 regulation; for example, depletion of LEM domain-containing 2 (LEMD2; also known as NET25) was shown to result in elevated levels of phosphorylated ERK1 following EGF exposure97 — raising the issue of whether deregulation of LEMD2-dependent events at the nuclear envelope could also contribute to the hyperactivity of the EGF pathway in the context of cancer. Changes in the number of NPCs, in nucleoporin partnerships and in the global architecture of the nuclear envelope98, suggest that there is a shift in the landscape of the nuclear envelope in conjunction with MAPK–ERK activation. There remains much to understand in terms of variables, such as acute versus chronic signalling and cellular context. In addition to the phosphorylation of nuclear periphery proteins, ERK1 and ERK2 can also physically compete with RB for interaction with lamin A, which thereby displaces RB into the nucleoplasm for phosphorylation and thus promotes entry into the cell cycle99. Emerin and LAP2β, the phosphorylation of which may lead to functional modulation3, are additional components of the nuclear envelope that are targeted by multiple signalling cascades (such as EGF and fibroblast growth factor (FGF) pathways) and kinases (for example, SRC and ABL)3.
Whatever the mechanism by which a tumour cell acquires the hallmark folding and invagination of the nuclear envelope (Fig. 1), one consequence with respect to signalling may be increased sensitization to the key second-messenger molecule calcium. Involutions of the nuclear envelope that occur under non-pathogenic conditions have been suggested to enhance nuclear transduction of calcium signals98,100. Calcium-mediated signalling is altered in multiple cancers101 and is important in regulating key cell cycle factors, as well as survival and apoptosis pathways. Thus, this is a prime opportunity to apply approaches that are used in other fields to elucidate how cancer-associated changes in nuclear architecture affect calcium signalling. Our knowledge to date, however, already indicates that morphological and molecular changes at the nuclear envelope that are associated with cancer are likely to be intertwined with alterations in intracellular signalling.
The nuclear envelope and genome stability
Studies of laminopathies have highlighted the importance of lamins to DNA repair. Mutation or aberrant expression of lamin A compromises DNA repair pathways, with defective recruitment of DNA double-strand break (DSB) repair proteins, including RAD50 and RAD51, and aberrant recruitment of the nucleotide excision repair factor xeroderma pigmentosum complementation group A (XPA) to constitutive DSBs detected as γH2AX foci102. The expression of lamin A mutants that are associated with muscular dystrophy or progeria in cultured cells also results in the mislocalization of the DNA damage response kinase ataxia-telangiectasia and Rad3-related (ATR)103. Furthermore, response to DNA damage caused by cisplatin or ultraviolet (UV) irradiation is impaired in the presence of lamin A mutants103. Several layers of regulation in the DNA damage response intersect with lamin: loss of lamin A function reduces the transcription of RAD51 and BRCA1 (Ref. 104), and also results in the downregulation and mislocalization of the tumour suppressors RB105,106 and ING1 (Ref. 107), as well as the DNA repair factor p53-binding protein 1 (53BP1)108. Thus, changes in lamin levels (Table 1) or even lamin organization (as may be the case when lobulated polymorphic nuclei are observed in tumours) could reflect defects in the ability of a cell to respond to DNA damage and the accumulation of DNA lesions. This deregulation increases the potential of acquiring mutations that might contribute to tumorigenesis or further augment the aggressiveness of the tumour cells, but may also promote sensitivity to chemotherapeutic strategies that capitalize on an impaired ability for DNA repair.
Proteins of the NPC also aid in coordinating DNA repair. In yeast, many observations point to the NPC as a hub for SUMO- and ubiquitin-mediated regulation of DNA repair27,109. In addition, Nup1p, Nup2p and Nup60p are phosphorylated in response to DNA damage that is induced by treatment with the DNA-alkylating agent methyl methanesulphonate, and these proteins have been identified as direct targets of Rad53p (the yeast homologue of the human checkpoint kinase CHK2), which functions in triggering cell cycle checkpoint arrest in response to DNA damage110. Replication stress and inhibition of Chk2 were found to be lethal in zebrafish larvae bearing a mutation in the nucleoporin Elys111. In mice, tissue-specific disruption of ELYS in intestinal epithelium leads to elevated activation of the DNA damage response, along with crypt cell apoptosis and disruption of intestinal epithelium morphology112. Overexpression of the UBC9-binding motif of the mammalian nucleoporin NUP358 compromises RAD51 recruitment to DNA damage-induced foci113; whereas, NUP153 was recently identified in a screen for factors important for 53BP1 recruitment to DNA damage-induced foci following ionizing radiation114. Follow-up experiments showed that NUP153 is crucial to the nuclear import of 53BP1, although whether this is the full scope of its role remains to be seen. In a different study, lowering the level of NUP62 expression in cultured ovarian carcinoma cells was found to result in cisplatin resistance115. Although the molecular pathway underlying the role of NUP62 in this context has not yet been elucidated, these results point towards the importance of nucleoporins in cellular responses to DNA damage.
The nuclear envelope environment is also of particular importance to telomere maintenance. The fibroblasts from patients with Hutchinson–Gilford progeria syndrome (HGPS) (Box 3) have aberrantly short telomeres116. Interestingly, overexpression of either the mutant form of lamin A found in progeria, which accumulates at the INM with a persistently farnesylated tail, or wild-type lamin A, leads to telomere shortening117, indicating that the balanced expression of lamin A, as well as the integrity of the nuclear lamina, are important for telomere maintenance. Lamin A, together with its binding partners LAP2α and barrier to autointegration factor (BAF; also known as BANF1), are known to participate in tethering telomeres to the nuclear periphery118,119. Molecular details of the roles for components of the nuclear periphery in telomere maintenance are better characterized in yeast, in which the recruitment of telomeres by INM proteins is proposed to promote telomere addition by telomerase; whereas, the recruitment of telomeres to NPCs is implicated in telomerase-independent recombination mechanisms that are the last resort when telomeres are severely eroded120. A recent study has highlighted a role of sumoylation in the dynamics of telomere tethering to the nuclear periphery121. With many players now identified, this area of research is poised to yield further mechanistic information that is important for the spatiotemporal control of DNA repair. Given that genomic instability and telomere attrition are typical features of tumour cells from many cancer types, connections between the nuclear envelope and DNA repair and telomere maintenance are a highly relevant consideration in the assessment of phenotypes that accompany changes in nuclear morphology.
Applying knowledge of the nuclear envelope
As our knowledge of the molecular nature of the nuclear envelope environment grows, can this be applied to create more precise, objective measurements to classify tumours or to make prognostic predictions? Given the myriad determinants of nuclear morphology, a simple molecular signature that corresponds to altered shape is unlikely. Compensatory changes and cell type-specific context will influence how the alteration of a nuclear envelope component manifests. Nonetheless, the intimate connection between nuclear envelope morphology and cancer suggests that the expression levels of key proteins at the nuclear envelope may provide an informative parameter in tumour detection and characterization. Several components of the nuclear periphery — lamins, LAP2 and emerin — have already been assessed as biomarkers in a wide range of cancer types (for example, see Refs 13,122,123,124,125) (Table 1). NUP88 was serendipitously discovered as a cancer biomarker126,127, and has since been found in many studies to be overexpressed in malignant tissues127,128,129,130,131,132,133. More generally, lamins, certain nucleoporins and SENP1 have emerged in unbiased screens as biomarkers of interest for their change in levels found in cancer (for example, see Refs 45,134,135,136). Intriguingly, not only was lamin B1 found in a proteomic approach to be upregulated in hepatocellular tumours137, but mRNA encoding lamin B1 was also detected in the blood circulation, and its detection in plasma indicated early stage hepatocellular carcinoma with 76% sensitivity and 82% accuracy138.
Absolute levels, of course, do not always reflect the functional status of a particular protein. In this regard, it is interesting that in a comparative phosphoproteomic analysis using subclones of a breast tumour cell line with differing metastatic properties, phosphorylation of lamin A was found to be associated with metastatic potential139. Although the mechanistic consequence of this modification in tumour cells has not yet been pursued, one function ascribed to lamin A phosphorylation is in regulating the process of lamina assembly and disassembly during the cell cycle140. It is also interesting to note that certain viral infections cause the phosphorylation of lamins, which triggers local lamina reorganization that affects nuclear envelope integrity (for example, see Ref. 141).
The assessment of the components of the nuclear envelope as prognostic indicators has been limited (Table 1), and again does not reflect a simple pattern81,142. Elevated expression of various lamins was found to correlate with indicators of poor prognosis in liver (lamin B1)138, prostate (lamin A)136 and colorectal cancer (lamin A)16. By contrast, downregulation of A-type lamins correlates with poor prognosis in gastric carcinoma143 and large B cell lymphoma144. Changes in the levels of lamins at the nuclear envelope, either an increase or a decrease, may cause an imbalance that undermines their function. Consistent with this, comparative proteomic analysis in progression models of both fibrosarcoma and colorectal carcinoma point to an inverse correlation between lamin A expression and malignant progression145,146; whereas, lamin A levels increased in more advanced stages of ovarian cancer147. Collectively, these studies indicate that, when considering lamin expression, deviation from the norm, rather than an increase or a decrease, could be used as a marker of nuclear envelope deregulation. However, overexpression of a distinct marker of the nuclear envelope, NUP88, consistently correlates with an aggressive tumour phenotype and poor prognosis in numerous cancers128,129,131,132,148.
The long list of translocations that involve nucleoporins also underscores the link between NPC-associated alterations and tumorigenesis. Although many of the resulting fusion proteins have roles that are independent of the NPC (see Ref. 51 for a detailed review), altered function of the NPC may contribute to pathogenicity. In the case of NUP214–ABL, targeting of this fusion protein to the NPC proved important for its transforming activity149, and NUP98 fusion proteins have been found to divert CRM1 from crucial roles in nuclear export150. In addition to their ancillary use for diagnosis, detecting the presence of these nucleoporin fusions can have implications for therapeutic stratification, as has been shown for the t(6;9) DEK–NUP214 fusion, which is associated with aggressive acute myelogenous leukaemia151.
As with other biomarkers, a particular combination — which could include, but need not exclusively include, components of the nuclear envelope — might be most informative. This combination might be derived from unbiased analysis or could be directed towards markers that reflect a particular feature of the tumour or its environment that is relevant to consider in conjunction with alterations at the nuclear envelope. As an example, tracking both EGF receptor and lamin levels might be more predictive than tracking either factor alone. Another strategy is to combine the assessment of a nuclear envelope component with a functional attribute of the tumour. Meijer and colleagues152 have recently carried out such an analysis in colon cancer, looking at lamin A expression in conjunction with microsatellite stability, which indicates the functional status of the mismatch repair pathway152. Interestingly, they found that, in microsatellite-stable stage III tumours, if LMNA mRNA was low, 100% of patients who had not received adjuvant chemotherapy had disease recurrence versus 37.8% of patients with the same profile except for high LMNA expression (P < 0.01). This finding needs to be confirmed in an independent data set, but it points to additional variables that may help to resolve discrepant conclusions of biomarker studies that looked exclusively at LMNA in cohorts that were mixed with respect to microsatellite stability, tumour grade and status of adjuvant therapy.
The morphology of the nucleus is particularly informative in pathological assessment because it gives an integrated view of many layers of cellular regulation that affect cell growth and behaviour. Thus, to derive more benefit in the clinical arena from our growing knowledge of the nuclear envelope environment, another tactic is to image key features of the nuclear periphery with more precision. For example, the detection of nuclear envelope components can give a much more detailed view of the contours of the nucleus than can be obtained from standard histological stains, as has been shown by tracking the INM proteins emerin and lamin153. And, although immunohistochemistry reveals this pattern, the detection of specific nuclear envelope components by immunofluorescence gives a more detailed view153 (Fig. 1) and is becoming more readily applied in the clinical setting154. There are indications in the literature that higher NPC numbers are associated with more aggressive tumours155,156,157,158. The quantitative assessment of NPCs is now possible with high-resolution light microscopy (for example, see Ref. 90) and could be used to evaluate this more thoroughly and to determine its potential use as a prognostic tool. Beyond absolute levels of proteins in the nuclear envelope environment, their precise localization more fully reflects whether they are functioning normally. Consistent with this, the prevalence of lamin B-deficient microdomains of the nuclear envelope was found to increase with Gleason grade in prostate cancer80. Overall, the information we are gaining in terms of the composition and function of the nuclear envelope opens up several new strategies to make greater use of the long-standing connection between nuclear morphology and cancer phenotype.
Conclusions and future perspectives
Currently, our knowledge suggests that changes in the nuclear envelope environment can propagate to alterations in signalling, as well as the control of transcription, cell migration and polarity, genome stability and cell division (Fig. 3). The nuclear envelope environment is clearly tightly interconnected, both physically and functionally, with many aspects of cell biology. For example, nuclear shape is highly sensitive to the function of the endoplasmic reticulum (ER), including lipid production159; thus, cancer-associated changes in nuclear shape may additionally implicate an alteration in ER function, lipid homeostasis and/or lipid signalling. As the site of secretory and transmembrane protein synthesis, the ER is also crucial for post-translational protein glycosylation and for the folding of many proteins, including those located in the nuclear envelope. Hypoxia and nutrient depletion, which are characteristics of many tumour microenvironments, can compromise these functions of the ER and can lead to the accumulation of misfolded proteins, creating a condition generally termed ER stress160. Not only would this probably influence the range of nuclear envelope proteins (and, in turn, the structure and function of the nucleus), but physical continuity between the nuclear envelope and the ER also means that the lumen of the nuclear envelope would be directly subjected to stress-associated changes that should be evaluated for their effect on nuclear envelope function.
Myriad cellular activities are coordinated by a range of factors that are integral to or that are associated with the nuclear envelope. The permeability barrier and the scaffolding platform established by the nuclear envelope locally concentrate factors, permit regulated sequestration of their activity and allow spatially restricted post-translational modification. These events at the nuclear periphery control the activity, as well as the stability and the proper cellular localization of, components that are key to signalling, transcription, DNA repair, cell division, and cell shape and migration. Proteins of the nuclear periphery, such as nucleoporins (NUPs) and lamins, can also take on roles within the nucleoplasm and during mitosis. As a physical anchoring point for centrosomes and the cytoskeleton, proper function of the nuclear envelope is also crucial for various mechanical events, including those of cell division and cell migration. BAF, barrier to autointegration factor; cAMP, cyclic AMP; CBP, CREB-binding protein; CTCF, CCCTC-binding factor; EGF, epidermal growth factor; EPAC1, exchange protein directly activated by cAMP 1; GCL, germ cell-less; HDAC, histone deacetylase; HP1, heterochromatin protein 1; IGF, insulin-like growth factor; LAP2β, lamin-associated polypeptide 2β; LINC, linker of nucleoskeleton and cytoskeleton; NPC, nuclear pore complex; P, phosphorylation; SENP, sentrin-specific protease; SREBP1, sterol regulatory element-binding protein 1; TGFβ, transforming growth factor-β; UBC9, ubiquitin-conjugating enzyme 9.
Recent advances support the view that the nuclear envelope is a site where many aspects of cell function are coordinated. Nuclear envelope components have vital roles both in this environment and sometimes — through dynamic spatial and temporal regulation — at other sites in the cell. Changes that are associated with alterations of the nuclear envelope can undermine normal cell function and have the potential to augment the ability of tumour cells to adapt and evolve. With the availability of molecular tools for detection and analysis, systematic approaches can now be carried out to more directly address the ramifications of discrete changes of the nuclear envelope on tumour growth. For example, mice expressing different levels or mutant alleles of lamins could be crossed with genetically engineered mouse models of cancer161. The inducible expression of protein (or, similarly, the expression of mutants or loss of expression through genetic deletion) could be used to disrupt the normal organization of the nuclear envelope in order to probe the contribution of such changes to different stages in carcinogenesis (such as, initiation versus metastasis). Such models would also be of use in understanding how the response to DNA-damaging agents and other chemotherapeutics is altered by changes at the nuclear envelope. As pathologists have known for years, even subtle alterations in the morphology of the nuclear envelope can have profound implications on cellular function. The knowledge of underlying pathways and molecules has given us new insight into how such perturbations may influence tumour cell function and has laid the groundwork for future research to further decipher and capitalize on the connections between the nuclear envelope and cancer.
References
Dey, P. Cancer nucleus: morphology and beyond. Diagn. Cytopathol. 38, 382–390 (2010).
True, L. D. & Jordan, C. D. The cancer nuclear microenvironment: interface between light microscopic cytology and molecular phenotype. J. Cell. Biochem. 104, 1994–2003 (2008).
Wilson, K. L. & Berk, J. M. The nuclear envelope at a glance. J. Cell. Sci. 123, 1973–1978 (2010).
Wente, S. R. & Rout, M. P. The nuclear pore complex and nuclear transport. Cold Spring Harb. Perspect. Biol. 2, a000562 (2010).
Chatel, G. & Fahrenkrog, B. Nucleoporins: Leaving the nuclear pore complex for a successful mitosis. Cell Signal 13 Jun 2011 (doi:10.1016/j.cellsig.2011.05.023).
Dauer, W. T. & Worman, H. J. The nuclear envelope as a signaling node in development and disease. Dev. Cell 17, 626–638 (2009).
Friedl, P., Wolf, K. & Lammerding, J. Nuclear mechanics during cell migration. Curr. Opin. Cell Biol. 23, 55–64 (2011).
Olins, A. L. et al. The LINC-less granulocyte nucleus. Eur. J. Cell Biol. 88, 203–214 (2009).
Méjat, A. & Misteli, T. LINC complexes in health and disease. Nucleus 1, 40–52 (2010).
Starr, D. A. & Fridolfsson, H. N. Interactions between nuclei and the cytoskeleton are mediated by SUN-KASH nuclear-envelope bridges. Annu. Rev. Cell Dev. Biol. 26, 421–444 (2010).
Khatau, S. B. et al. A perinuclear actin cap regulates nuclear shape. Proc. Natl Acad. Sci. USA 106, 19017–19022 (2009).
Luxton, G. W. G., Gomes, E. R., Folker, E. S., Vintinner, E. & Gundersen, G. G. Linear arrays of nuclear envelope proteins harness retrograde actin flow for nuclear movement. Science 329, 956–959 (2010).
Capo-chichi, C. D., Cai, K. Q., Testa, J. R., Godwin, A. K. & Xu, X.-X. Loss of GATA6 leads to nuclear deformation and aneuploidy in ovarian cancer. Mol. Cell. Biol. 29, 4766–4777 (2009).
Butcher, D. T., Alliston, T. & Weaver, V. M. A tense situation: forcing tumour progression. Nature Rev. Cancer 9, 108–122 (2009).
Lombardi, M. L. et al. The interaction between nesprins and sun proteins at the nuclear envelope is critical for force transmission between the nucleus and cytoskeleton. J. Biol. Chem. 286, 26743–26753 (2011).
Willis, N. D. et al. Lamin A/C is a risk biomarker in colorectal cancer. PLoS ONE 3, e2988 (2008). The authors found that lamin A expression in colorectal cancer tissue is predictive of poor prognosis, and uncovered a role of lamin A in enhancing cell motility and invasiveness.
Zhou, L. & Panté, N. The nucleoporin Nup153 maintains nuclear envelope architecture and is required for cell migration in tumor cells. FEBS Lett. 584, 3013–3020 (2010).
Joseph, J. & Dasso, M. The nucleoporin Nup358 associates with and regulates interphase microtubules. FEBS Lett. 582, 190–196 (2008).
Murawala, P., Tripathi, M. M., Vyas, P., Salunke, A. & Joseph, J. Nup358 interacts with APC and plays a role in cell polarization. J. Cell. Sci. 122, 3113–3122 (2009).
Collin, L., Schlessinger, K. & Hall, A. APC nuclear membrane association and microtubule polarity. Biol. Cell 100, 243–252 (2008).
Hubert, T., Vandekerckhove, J. & Gettemans, J. Exo70-mediated recruitment of nucleoporin Nup62 at the leading edge of migrating cells is required for cell migration. Traffic 10, 1257–1271 (2009).
Gloerich, M. et al. The nucleoporin RanBP2 tethers the cAMP effector Epac1 and inhibits its catalytic activity. J. Cell Biol. 193, 1009–1020 (2011).
Liu, C. et al. The interaction of Epac1 and Ran promotes Rap1 activation at the nuclear envelope. Mol. Cell. Biol. 30, 3956–3969 (2010).
Susaki, E. & Nakayama, K. I. Multiple mechanisms for p27(Kip1) translocation and degradation. Cell Cycle 6, 3015–3020 (2007).
Gavet, O. & Pines, J. Activation of cyclin B1-Cdk1 synchronizes events in the nucleus and the cytoplasm at mitosis. J. Cell Biol. 189, 247–259 (2010).
Chakraborty, P. et al. Nucleoporin levels regulate cell cycle progression and phase-specific gene expression. Dev. Cell 15, 657–667 (2008).
Palancade, B. & Doye, V. Sumoylating and desumoylating enzymes at nuclear pores: underpinning their unexpected duties? Trends Cell Biol. 18, 174–183 (2008).
Xu, Y. et al. SUMO-specific protease 1 regulates the in vitro and in vivo growth of colon cancer cells with the upregulated expression of CDK inhibitors. Cancer Lett. 309, 78–84 (2011).
Dorner, D., Gotzmann, J. & Foisner, R. Nucleoplasmic lamins and their interaction partners, LAP2α, Rb, and BAF, in transcriptional regulation. FEBS J. 274, 1362–1373 (2007).
Burke, B. & Ellenberg, J. Remodelling the walls of the nucleus. Nature Rev. Mol. Cell Biol. 3, 487–497 (2002).
Mackay, D. R., Makise, M. & Ullman, K. S. Defects in nuclear pore assembly lead to activation of an Aurora B-mediated abscission checkpoint. J. Cell Biol. 191, 923–931 (2010).
Dar, A. A., Goff, L. W., Majid, S., Berlin, J. & El-Rifai, W. Aurora kinase inhibitors - rising stars in cancer therapeutics? Mol.Cancer Ther. 9, 268–278 (2010).
Verstraeten, V. L. R. M. et al. Protein farnesylation inhibitors cause donut-shaped cell nuclei attributable to a centrosome separation defect. Proc. Natl Acad. Sci. USA 108, 4997–5002 (2011). This study reports how a striking nuclear phenotype that results from inhibition of FNT is tied to the disruption of centrosome separation and a role for lamin B1, pointing to a novel mechanism that contributes to downstream consequences of this class of chemotherapy.
Sebti, S. M. & Der, C. J. Opinion: Searching for the elusive targets of farnesyltransferase inhibitors. Nature Rev. Cancer 3, 945–951 (2003).
Bolhy, S. et al. A Nup133-dependent NPC-anchored network tethers centrosomes to the nuclear envelope in prophase. J. Cell Biol. 192, 855–871 (2011).
Splinter, D. et al. Bicaudal D2, dynein, and kinesin-1 associate with nuclear pore complexes and regulate centrosome and nuclear positioning during mitotic entry. PLoS Biol. 8, e1000350 (2010).
Silkworth, W. T., Nardi, I. K., Paul, R., Mogilner, A. & Cimini, D. Timing of centrosome separation is important for accurate chromosome segregation. Mol. Biol. Cell 30 Nov 2011 (doi:10.1091/mbc.E11-02-0095). The authors observe that when centrosomes do not fully separate prior to nuclear envelope breakdown, there is a greater likelihood of defective kinetochore attachment to mitotic spindles and aberrant chromosome segregation.
Dawlaty, M. M. et al. Resolution of sister centromeres requires RanBP2-mediated SUMOylation of topoisomerase IIα. Cell 133, 103–115 (2008). This study reports a tumour-suppressive function of NUP358 that is attributed to the regulation of topoisomerase IIα sumoylation.
Zhang, X.-D. et al. SUMO-2/3 modification and binding regulate the association of CENP-E with kinetochores and progression through mitosis. Mol. Cell 29, 729–741 (2008).
Klein, U. R., Haindl, M., Nigg, E. A. & Muller, S. RanBP2 and SENP3 function in a mitotic SUMO2/3 conjugation-deconjugation cycle on Borealin. Mol. Biol. Cell 20, 410–418 (2009).
Ban, R., Nishida, T. & Urano, T. Mitotic kinase Aurora-B is regulated by SUMO-2/3 conjugation/deconjugation during mitosis. Genes Cells 16, 652–669 (2011).
Hang, J. & Dasso, M. Association of the human SUMO-1 protease SENP2 with the nuclear pore. J. Biol. Chem. 277, 19961–19966 (2002).
Ryu, H., Furuta, M., Kirkpatrick, D., Gygi, S. P. & Azuma, Y. PIASy-dependent SUMOylation regulates DNA topoisomerase IIα activity. J. Cell Biol. 191, 783–794 (2010).
Gardner, L., Malik, R., Shamizu, Y., Mullins, N. & Elshamy, W. M. Geminin overexpression prevents the completion of topoisomerase IIα chromosome decatenation leading to aneuploidy in human mammary epithelial cells. Breast Cancer Res. 13, R53 (2011).
Jacques, C. et al. Two-step differential expression analysis reveals a new set of genes involved in thyroid oncocytic tumors. J. Clin. Endocrinol. Metab. 90, 2314–2320 (2005).
Cheng, J., Bawa, T., Lee, P., Gong, L. & Yeh, E. T. H. Role of desumoylation in the development of prostate cancer. Neoplasia 8, 667–676 (2006).
Gómez-Baldó, L. et al. TACC3-TSC2 maintains nuclear envelope structure and controls cell division. Cell Cycle 9, 1143–1155 (2010).
Lee, S. H., Sterling, H., Burlingame, A. & McCormick, F. Tpr directly binds to Mad1 and Mad2 and is important for the Mad1-Mad2-mediated mitotic spindle checkpoint. Genes Dev. 22, 2926–2931 (2008).
Schvartzman, J.-M., Sotillo, R. & Benezra, R. Mitotic chromosomal instability and cancer: mouse modelling of the human disease. Nature Rev. Cancer 10, 102–115 (2010).
Cross, M. K. & Powers, M. A. Nup98 regulates bipolar spindle assembly through association with microtubules and opposition of MCAK. Mol. Biol. Cell 22, 661–672 (2011).
Xu, S. & Powers, M. A. Nuclear pore proteins and cancer. Semin. Cell Dev. Biol. 20, 620–630 (2009).
Jeganathan, K. B., Baker, D. J. & van Deursen, J. M. Securin associates with APCCdh1 in prometaphase but its destruction is delayed by Rae1 and Nup98 until the metaphase/anaphase transition. Cell Cycle 5, 366–370 (2006).
Jeganathan, K. B., Malureanu, L. & van Deursen, J. M. The Rae1-Nup98 complex prevents aneuploidy by inhibiting securin degradation. Nature 438, 1036–1039 (2005).
Tsai, M.-Y. et al. A mitotic lamin B matrix induced by RanGTP required for spindle assembly. Science 311, 1887–1893 (2006).
Civelekoglu-Scholey, G., Tao, L., Brust-Mascher, I., Wollman, R. & Scholey, J. M. Prometaphase spindle maintenance by an antagonistic motor-dependent force balance made robust by a disassembling lamin-B envelope. J. Cell Biol. 188, 49–68 (2010).
Korfali, N. et al. A flow cytometry-based screen of nuclear envelope transmembrane proteins identifies NET4/Tmem53 as involved in stress-dependent cell cycle withdrawal. PLoS ONE 6, e18762 (2011). The results in this paper highlight the importance of the nuclear envelope as a source for cell cycle regulatory proteins, many of which have not yet been characterized.
Draviam, V. M. et al. A functional genomic screen identifies a role for TAO1 kinase in spindle-checkpoint signalling. Nat. Cell Biol. 9, 556–564 (2007).
Ruault, M., Dubarry, M. & Taddei, A. Re-positioning genes to the nuclear envelope in mammalian cells: impact on transcription. Trends Genet. 24, 574–581 (2008).
Batrakou, D. G., Kerr, A. R. W. & Schirmer, E. C. Comparative proteomic analyses of the nuclear envelope and pore complex suggests a wide range of heretofore unexpected functions. J. Proteomics 72, 56–70 (2009).
Andrés, V. & González, J. M. Role of A-type lamins in signaling, transcription, and chromatin organization. J. Cell Biol. 187, 945–957 (2009).
Malhas, A., Saunders, N. J. & Vaux, D. J. The nuclear envelope can control gene expression and cell cycle progression via miRNA regulation. Cell Cycle 9, 531–539 (2010). This study underscores the effect of the nuclear lamina on regulating genes with key roles in cancer, including miRNAs.
Bandrés, E. et al. Identification by Real-time PCR of 13 mature microRNAs differentially expressed in colorectal cancer and non-tumoral tissues. Mol. Cancer 5, 29 (2006).
Valastyan, S., Chang, A., Benaich, N., Reinhardt, F. & Weinberg, R. A. Activation of miR-31 function in already-established metastases elicits metastatic regression. Genes Dev. 25, 646–659 (2011).
Valastyan, S. et al. A pleiotropically acting microRNA, miR-31, inhibits breast cancer metastasis. Cell 137, 1032–1046 (2009).
Xu, L., Kang, Y., Cöl, S. & Massagué, J. Smad2 nucleocytoplasmic shuttling by nucleoporins CAN/Nup214 and Nup153 feeds TGFβ signaling complexes in the cytoplasm and nucleus. Mol. Cell 10, 271–282 (2002).
Xylourgidis, N., Roth, P., Sabri, N., Tsarouhas, V. & Samakovlis, C. The nucleoporin Nup214 sequesters CRM1 at the nuclear rim and modulates NFkappaB activation in Drosophila. J. Cell. Sci. 119, 4409–4419 (2006).
Takahashi, N. et al. Tumor marker nucleoporin 88 kDa regulates nucleocytoplasmic transport of NF-kappaB. Biochem. Biophys. Res. Commun. 374, 424–430 (2008).
Capelson, M. et al. Chromatin-bound nuclear pore components regulate gene expression in higher eukaryotes. Cell 140, 372–383 (2010).
Vaquerizas, J. M. et al. Nuclear pore proteins nup153 and megator define transcriptionally active regions in the Drosophila genome. PLoS Genet. 6, e1000846 (2010).
Kalverda, B., Pickersgill, H., Shloma, V. V. & Fornerod, M. Nucleoporins directly stimulate expression of developmental and cell-cycle genes inside the nucleoplasm. Cell 140, 360–371 (2010).
Brown, C. R., Kennedy, C. J., Delmar, V. A., Forbes, D. J. & Silver, P. A. Global histone acetylation induces functional genomic reorganization at mammalian nuclear pore complexes. Genes Dev. 22, 627–639 (2008).
Kehat, I., Accornero, F., Aronow, B. J. & Molkentin, J. D. Modulation of chromatin position and gene expression by HDAC4 interaction with nucleoporins. J. Cell Biol. 193, 21–29 (2011).
Dickinson, M., Johnstone, R. W. & Prince, H. M. Histone deacetylase inhibitors: potential targets responsible for their anti-cancer effect. Invest. New Drugs 28 (Suppl. 1), 3–20 (2010).
Arib, G. & Akhtar, A. Multiple facets of nuclear periphery in gene expression control. Curr. Opin. Cell Biol. 23, 346–353 (2011).
Kind, J. & van Steensel, B. Genome-nuclear lamina interactions and gene regulation. Curr. Opin. Cell Biol. 22, 320–325 (2010).
Shimi, T. et al. The A- and B-type nuclear lamin networks: microdomains involved in chromatin organization and transcription. Genes Dev. 22, 3409–3421 (2008).
Mewborn, S. K. et al. Altered chromosomal positioning, compaction, and gene expression with a lamin A/C gene mutation. PLoS ONE 5, e14342 (2010).
Ottaviani, A., Schluth-Bolard, C., Gilson, E. & Magdinier, F. D4Z4 as a prototype of CTCF and lamins-dependent insulator in human cells. Nucleus 1, 30–36 (2010).
Kalverda, B. & Fornerod, M. Characterization of genome-nucleoporin interactions in Drosophila links chromatin insulators to the nuclear pore complex. Cell Cycle 9, 4812–4817 (2010).
Helfand, B. T. et al. Chromosomal regions associated with prostate cancer risk localize to lamin B deficient microdomains and exhibit reduced gene transcription. J. Pathol. 25 Oct 2011 (doi:10.1002/path.3033). The authors track the distribution of lamin proteins in prostate cancer and find that the frequency of nuclear blebs deficient in B-type lamins corresponds to the severity of Gleason grade.
Prokocimer, M. et al. Nuclear lamins: key regulators of nuclear structure and activities. J. Cell. Mol. Med. 13, 1059–1085 (2009).
Maraldi, N. M., Capanni, C., Cenni, V., Fini, M. & Lattanzi, G. Laminopathies and lamin-associated signaling pathways. J. Cell. Biochem. 112, 979–992 (2011).
Neumann, S. et al. Nesprin-2 interacts with α-catenin and regulates Wnt signaling at the nuclear envelope. J. Biol. Chem. 285, 34932–34938 (2010).
Markiewicz, E. et al. The inner nuclear membrane protein emerin regulates beta-catenin activity by restricting its accumulation in the nucleus. EMBO J. 25, 3275–3285 (2006).
Hernandez, L. et al. Functional coupling between the extracellular matrix and nuclear lamina by Wnt signaling in progeria. Dev. Cell 19, 413–425 (2010).
Shitashige, M. et al. Regulation of Wnt signaling by the nuclear pore complex. Gastroenterology 134, 1961–1971 (2008).
Carmo-fonseca, M. Testosterone-induced changes in nuclear pore complex number of prostatic nuclei from castrated rats. J. Ultrastruct. Res. 80, 243–251 (1982).
Ortiz, H. E. & Cavicchia, J. C. Androgen-induced changes in nuclear pore number and in tight junctions in rat seminal vesicle epithelium. Anat. Rec. 226, 129–134 (1990).
Maul, G. G. et al. Time sequence of nuclear pore formation in phytohemagglutinin-stimulated lymphocytes and in HeLa cells during the cell cycle. J. Cell Biol. 55, 433–447 (1972).
Maeshima, K. et al. Nuclear pore formation but not nuclear growth is governed by cyclin-dependent kinases (Cdks) during interphase. Nature Struct. Mol. Biol. 17, 1065–1071 (2010).
Richard, M. N., Deniset, J. F., Kneesh, A. L., Blackwood, D. & Pierce, G. N. Mechanical stretching stimulates smooth muscle cell growth, nuclear protein import, and nuclear pore expression through mitogen-activated protein kinase activation. J. Biol. Chem. 282, 23081–23088 (2007).
Kosako, H. et al. Phosphoproteomics reveals new ERK MAP kinase targets and links ERK to nucleoporin-mediated nuclear transport. Nature Struct. Mol. Biol. 16, 1026–1035 (2009).
Vomastek, T. et al. Extracellular signal-regulated kinase 2 (ERK2) phosphorylation sites and docking domain on the nuclear pore complex protein Tpr cooperatively regulate ERK2-Tpr interaction. Mol. Cell. Biol. 28, 6954–6966 (2008).
Whitehurst, A. W. et al. ERK2 enters the nucleus by a carrier-independent mechanism. Proc. Natl Acad. Sci. USA 99, 7496–7501 (2002).
Yazicioglu, M. N. et al. Mutations in ERK2 binding sites affect nuclear entry. J. Biol. Chem. 282, 28759–28767 (2007).
Smith, E. R. et al. Nuclear entry of activated MAPK is restricted in primary ovarian and mammary epithelial cells. PLoS ONE 5, e9295 (2010).
Huber, M. D., Guan, T. & Gerace, L. Overlapping functions of nuclear envelope proteins NET25 (Lem2) and emerin in regulation of extracellular signal-regulated kinase signaling in myoblast differentiation. Mol. Cell. Biol. 29, 5718–5728 (2009).
Wittmann, M. et al. Synaptic activity induces dramatic changes in the geometry of the cell nucleus: interplay between nuclear structure, histone H3 phosphorylation, and nuclear calcium signaling. J. Neurosci. 29, 14687–14700 (2009).
Rodríguez, J. et al. ERK1/2 MAP kinases promote cell cycle entry by rapid, kinase-independent disruption of retinoblastoma-lamin A complexes. J. Cell Biol. 191, 967–979 (2010). This study reports that ERK1–ERK2 can displace RB from lamin A, independently of ERK1–ERK2 kinase activity, to promote cell cycle entry.
Collado-Hilly, M., Shirvani, H., Jaillard, D. & Mauger, J.-P. Differential redistribution of Ca2+-handling proteins during polarisation of MDCK cells: Effects on Ca2+ signalling. Cell Calcium 48, 215–224 (2010).
Parkash, J. & Asotra, K. Calcium wave signaling in cancer cells. Life Sci. 87, 587–595 (2010).
Gonzalez-Suarez, I. & Gonzalo, S. Nurturing the genome: A-type lamins preserve genomic stability. Nucleus 1, 129–135 (2010).
Manju, K., Muralikrishna, B. & Parnaik, V. K. Expression of disease-causing lamin A mutants impairs the formation of DNA repair foci. J. Cell. Sci. 119, 2704–2714 (2006).
Redwood, A. B. et al. A dual role for A-type lamins in DNA double-strand break repair. Cell Cycle 10, 2549–2560 (2011).
Johnson, B. R. et al. A-type lamins regulate retinoblastoma protein function by promoting subnuclear localization and preventing proteasomal degradation. Proc. Natl Acad. Sci. USA 101, 9677–9682 (2004).
Nitta, R. T., Jameson, S. A., Kudlow, B. A., Conlan, L. A. & Kennedy, B. K. Stabilization of the retinoblastoma protein by A-type nuclear lamins is required for INK4A-mediated cell cycle arrest. Mol. Cell. Biol. 26, 5360–5372 (2006).
Han, X. et al. Tethering by lamin A stabilizes and targets the ING1 tumour suppressor. Nat. Cell Biol. 10, 1333–1340 (2008).
Gonzalez-Suarez, I. et al. Novel roles for A-type lamins in telomere biology and the DNA damage response pathway. EMBO J. 28, 2414–2427 (2009).
Nagai, S. et al. Functional targeting of DNA damage to a nuclear pore-associated SUMO-dependent ubiquitin ligase. Science 322, 597–602 (2008).
Smolka, M. B., Albuquerque, C. P., Chen, S-hong & Zhou, H. Proteome-wide identification of in vivo targets of DNA damage checkpoint kinases. Proc. Natl Acad. Sci. USA 104, 10364–10369 (2007).
Davuluri, G. et al. Mutation of the zebrafish nucleoporin elys sensitizes tissue progenitors to replication stress. PLoS Genet. 4, e1000240 (2008).
Gao, N. et al. The nuclear pore complex protein Elys is required for genome stability in mouse intestinal epithelial progenitor cells. Gastroenterology 140, 1547–1555 (2011). This paper shows that removal of the mammalian nucleoporin ELYS from the developing intestinal epithelium results in activation of a DNA damage response without noticeably affecting the presence of nuclear pores.
Saitoh, H., Pizzi, M. D. & Wang, J. Perturbation of SUMOlation enzyme Ubc9 by distinct domain within nucleoporin RanBP2/Nup358. J. Biol. Chem. 277, 4755–4763 (2002).
Moudry, P. et al. Nucleoporin NUP153 guards genome integrity by promoting nuclear import of 53BP1. Cell Death Differ. 11 Nov 2011 (doi: 10.1038/cdd.2011.150). This study reports the results of a screen that involved knocking down expression of more than 1,000 genes to identify factors required for 53BP1 recruitment to ionizing radiation-induced DNA damage foci.
Kinoshita, Y., Kalir, T., Rahaman, J., Dottino, P. & Kohtz, D. S. Alterations in nuclear pore architecture allow cancer cell entry into or exit from drug-resistant dormancy. Am. J. Pathol. 180, 375–389 (2012).
Allsopp, R. C. et al. Telomere length predicts replicative capacity of human fibroblasts. Proc. Natl Acad. Sci. USA 89, 10114–10118 (1992).
Huang, S., Risques, R. A., Martin, G. M., Rabinovitch, P. S. & Oshima, J. Accelerated telomere shortening and replicative senescence in human fibroblasts overexpressing mutant and wild-type lamin A. Exp. Cell Res. 314, 82–91 (2008).
Raz, V. et al. The nuclear lamina promotes telomere aggregation and centromere peripheral localization during senescence of human mesenchymal stem cells. J. Cell. Sci. 121, 4018–4028 (2008).
Dechat, T. et al. LAP2α and BAF transiently localize to telomeres and specific regions on chromatin during nuclear assembly. J. Cell. Sci. 117, 6117–6128 (2004).
Lisby, M., Teixeira, T., Gilson, E. & Géli, V. The fate of irreparable DNA double-strand breaks and eroded telomeres at the nuclear periphery. Nucleus 1, 158–161 (2010).
Ferreira, H. C. et al. The PIAS homologue Siz2 regulates perinuclear telomere position and telomerase activity in budding yeast. Nat. Cell Biol. 13, 867–874 (2011).
Broers, J. L. et al. Nuclear A-type lamins are differentially expressed in human lung cancer subtypes. Am. J. Pathol. 143, 211–220 (1993).
Moss, S. F. et al. Decreased and aberrant nuclear lamin expression in gastrointestinal tract neoplasms. Gut 45, 723–729 (1999).
Capo-chichi, C. D. et al. Nuclear envelope structural defects cause chromosomal numerical instability and aneuploidy in ovarian cancer. BMC Med. 9, 28 (2011).
Somech, R. et al. Enhanced expression of the nuclear envelope LAP2 transcriptional repressors in normal and malignant activated lymphocytes. Ann. Hematol. 86, 393–401 (2007).
Schneider, J. et al. Cross-reactivity between Candida albicans and human ovarian carcinoma as revealed by monoclonal antibodies PA10F and C6. Br. J. Cancer 77, 1015–1020 (1998).
Martínez, N., Alonso, A., Moragues, M. D., Pontón, J. & Schneider, J. The nuclear pore complex protein Nup88 is overexpressed in tumor cells. Cancer Res. 59, 5408–5411 (1999).
Gould, V. E. et al. Nup88 (karyoporin) in human malignant neoplasms and dysplasias: correlations of immunostaining of tissue sections, cytologic smears, and immunoblot analysis. Hum. Pathol. 33, 536–544 (2002).
Agudo, D. et al. Nup88 mRNA overexpression is associated with high aggressiveness of breast cancer. Int. J. Cancer 109, 717–720 (2004).
Knoess, M. et al. Nucleoporin 88 expression in hepatitis B and C virus-related liver diseases. World J. Gastroenterol. 12, 5870–5874 (2006).
Zhang, Z.-Y. et al. Nup88 expression in normal mucosa, adenoma, primary adenocarcinoma and lymph node metastasis in the colorectum. Tumour Biol. 28, 93–99 (2007).
Brustmann, H. & Hager, M. Nucleoporin 88 expression in normal and neoplastic squamous epithelia of the uterine cervix. Ann. Diagn. Pathol. 13, 303–307 (2009).
Schneider, J., Martínez-Arribas, F. & Torrejón, R. Nup88 expression is associated with myometrial invasion in endometrial carcinoma. Int. J. Gynecol. Cancer 20, 804–808 (2010).
Hernández, P. et al. Integrative analysis of a cancer somatic mutome. Mol. Cancer 6, 13 (2007).
Alfonso, P., Cañamero, M., Fernández-Carbonié, F., Núñez, A. & Casal, J. I. Proteome analysis of membrane fractions in colorectal carcinomas by using 2D-DIGE saturation labeling. J. Proteome Res. 7, 4247–4255 (2008).
Skvortsov, S. et al. Proteomics profiling of microdissected low- and high-grade prostate tumors identifies Lamin A as a discriminatory biomarker. J. Proteome Res. 10, 259–268 (2011).
Lim, S. O. et al. Proteome analysis of hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 291, 1031–1037 (2002).
Sun, S., Xu, M. Z., Poon, R. T., Day, P. J. & Luk, J. M. Circulating Lamin B1 (LMNB1) biomarker detects early stages of liver cancer in patients. J. Proteome Res. 9, 70–78 (2010).
Xie, X. et al. A comparative phosphoproteomic analysis of a human tumor metastasis model using a label-free quantitative approach. Electrophoresis 31, 1842–1852 (2010).
Dechat, T., Adam, S. A., Taimen, P., Shimi, T. & Goldman, R. D. Nuclear lamins. Cold Spring Harb. Perspect. Biol. 2, a000547 (2010).
Ballester, M. et al. Disruption of nuclear organization during the initial phase of African swine fever virus infection. J. Virol. 85, 8263–8269 (2011).
Foster, C. R., Przyborski, S. A., Wilson, R. G. & Hutchison, C. J. Lamins as cancer biomarkers. Biochem. Soc. Trans. 38, 297–300 (2010).
Wu, Z. et al. Reduced expression of lamin A/C correlates with poor histological differentiation and prognosis in primary gastric carcinoma. J. Exp. Clin. Cancer Res. 28, 8 (2009).
Agrelo, R. et al. Inactivation of the lamin A/C gene by CpG island promoter hypermethylation in hematologic malignancies, and its association with poor survival in nodal diffuse large B-cell lymphoma. J. Clin. Oncol. 23, 3940–3947 (2005).
Kuramitsu, Y. et al. Proteomic analysis for nuclear proteins related to tumour malignant progression: a comparative proteomic study between malignant progressive cells and regressive cells. Anticancer Res. 30, 2093–2099 (2010).
Roth, U. et al. Differential expression proteomics of human colorectal cancer based on a syngeneic cellular model for the progression of adenoma to carcinoma. Proteomics 10, 194–202 (2010).
Wang, Y. et al. Differential protein mapping of ovarian serous adenocarcinomas: identification of potential markers for distinct tumor stage. J. Proteome Res. 8, 1452–1463 (2009).
Emterling, A. et al. Clinicopathological significance of Nup88 expression in patients with colorectal cancer. Oncology 64, 361–369 (2003).
De Keersmaecker, K. et al. Kinase activation and transformation by NUP214-ABL1 is dependent on the context of the nuclear pore. Mol. Cell 31, 134–142 (2008). The translocation and resulting fusion protein NUP214–ABL, found in ∼6% of patients with T cell acute lymphoblastic leukaemia, is shown to require localization to the NPC for its transformation activity.
Takeda, A., Sarma, N. J., Abdul-Nabi, A. M. & Yaseen, N. R. Inhibition of CRM1-mediated nuclear export of transcription factors by leukemogenic NUP98 fusion proteins. J. Biol. Chem. 285, 16248–16257 (2010).
Garçon, L. et al. DEK-CAN molecular monitoring of myeloid malignancies could aid therapeutic stratification. Leukemia 19, 1338–1344 (2005).
Belt, E. J. T. et al. Loss of lamin A/C expression in stage II and III colon cancer is associated with disease recurrence. Eur. J. Cancer 47, 1837–1845 (2011). This study combines the analysis of A-type lamins in colon tumours with two other parameters that have been shown to have prognostic value, pointing to the potential of combining prognostic indictors for more informative stratification of patients.
Bussolati, G. Proper detection of the nuclear shape: ways and significance. Rom J. Morphol. Embryol. 49, 435–439 (2008).
Moeder, C. B., Giltnane, J. M., Moulis, S. P. & Rimm, D. L. Quantitative, fluorescence-based in-situ assessment of protein expression. Methods Mol. Biol. 520, 163–175 (2009).
Johannessen, J. V., Sobrinho-Simões, M., Finseth, I. & Pilström, L. Papillary carcinomas of the thyroid have pore-deficient nuclei. Int. J. Cancer 30, 409–411 (1982).
Czerniak, B., Koss, L. G. & Sherman, A. Nuclear pores and DNA ploidy in human bladder carcinomas. Cancer Res. 44, 3752–3756 (1984).
Sugie, S., Yoshimi, N., Tanaka, T., Mori, H. & Williams, G. M. Alterations of nuclear pores in preneoplastic and neoplastic rat liver lesions induced by 2-acetylaminofluorene. Carcinogenesis 15, 95–98 (1994).
Lewin, J. M., Lwaleed, B. A., Cooper, A. J. & Birch, B. R. The direct effect of nuclear pores on nuclear chemotherapeutic concentration in multidrug resistant bladder cancer: the nuclear sparing phenomenon. J. Urol. 177, 1526–1530 (2007).
Webster, M., Witkin, K. L. & Cohen-Fix, O. Sizing up the nucleus: nuclear shape, size and nuclear-envelope assembly. J. Cell. Sci. 122, 1477–1486 (2009).
Lee, A. S. & Hendershot, L. M. ER stress and cancer. Cancer Biol. Ther. 5, 721–722 (2006).
Politi, K. & Pao, W. How genetically engineered mouse tumor models provide insights into human cancers. J. Clin. Oncol. 29, 2273–2281 (2011).
Goldberg, M. W., Huttenlauch, I., Hutchison, C. J. & Stick, R. Filaments made from A- and B-type lamins differ in structure and organization. J. Cell. Sci. 121, 215–225 (2008).
Sullivan, T. et al. Loss of A-type lamin expression compromises nuclear envelope integrity leading to muscular dystrophy. J. Cell Biol. 147, 913–920 (1999).
Ralle, T., Grund, C., Franke, W. W. & Stick, R. Intranuclear membrane structure formations by CaaX-containing nuclear proteins. J. Cell. Sci. 117, 6095–6104 (2004).
Polychronidou, M., Hellwig, A. & Grosshans, J. Farnesylated nuclear proteins Kugelkern and lamin Dm0 affect nuclear morphology by directly interacting with the nuclear membrane. Mol. Biol. Cell 21, 3409–3420 (2010).
Simon, D. N., Zastrow, M. S. & Wilson, K. L. Direct actin binding to A- and B-type lamin tails and actin filament bundling by the lamin A tail. Nucleus 1, 264–272 (2010).
Holaska, J. M., Kowalski, A. K. & Wilson, K. L. Emerin caps the pointed end of actin filaments: evidence for an actin cortical network at the nuclear inner membrane. PLoS Biol. 2, E231 (2004).
Antonin, W., Ungricht, R. & Kutay, U. Traversing the NPC along the pore membrane: targeting of membrane proteins to the INM. Nucleus 2, 87–91 (2011).
Liu, Q. et al. Functional association of Sun1 with nuclear pore complexes. J. Cell Biol. 178, 785–798 (2007).
Hawryluk-Gara, L. A., Shibuya, E. K. & Wozniak, R. W. Vertebrate Nup53 interacts with the nuclear lamina and is required for the assembly of a Nup93-containing complex. Mol. Biol. Cell 16, 2382–2394 (2005).
Lussi, Y. C., Hügi, I., Laurell, E., Kutay, U. & Fahrenkrog, B. The nucleoporin Nup88 is interacting with nuclear lamin A. Mol. Biol. Cell 22, 1080–1090 (2011).
Smythe, C., Jenkins, H. E. & Hutchison, C. J. Incorporation of the nuclear pore basket protein nup153 into nuclear pore structures is dependent upon lamina assembly: evidence from cell-free extracts of Xenopus eggs. EMBO J. 19, 3918–3931 (2000).
Mackay, D. R., Elgort, S. W. & Ullman, K. S. The nucleoporin Nup153 has separable roles in both early mitotic progression and the resolution of mitosis. Mol. Biol. Cell 20, 1652–1660 (2009).
D'Angelo, M. A., Raices, M., Panowski, S. H. & Hetzer, M. W. Age-dependent deterioration of nuclear pore complexes causes a loss of nuclear integrity in postmitotic cells. Cell 136, 284–295 (2009).
Scaffidi, P. & Misteli, T. Lamin A-dependent nuclear defects in human aging. Science 312, 1059–1063 (2006).
Tang, Y., Chen, Y., Jiang, H. & Nie, D. Promotion of tumor development in prostate cancer by progerin. Cancer Cell. Int. 10, 47 (2010).
Espada, J. et al. Nuclear envelope defects cause stem cell dysfunction in premature-aging mice. J. Cell Biol. 181, 27–35 (2008).
Mason, M. J., Fan, G., Plath, K., Zhou, Q. & Horvath, S. Signed weighted gene co-expression network analysis of transcriptional regulation in murine embryonic stem cells. BMC Genomics 10, 327 (2009).
Lupu, F., Alves, A., Anderson, K., Doye, V. & Lacy, E. Nuclear pore composition regulates neural stem/progenitor cell differentiation in the mouse embryo. Dev. Cell 14, 831–842 (2008).
Scaffidi, P. & Misteli, T. Lamin A-dependent misregulation of adult stem cells associated with accelerated ageing. Nat. Cell Biol. 10, 452–459 (2008).
Doucet, C. M. & Hetzer, M. W. Nuclear pore biogenesis into an intact nuclear envelope. Chromosoma 119, 469–477 (2010).
Malhas, A., Goulbourne, C. & Vaux, D. J. The nucleoplasmic reticulum: form and function. Trends Cell Biol. 21, 362–373 (2011).
Venables, R. S. et al. Expression of individual lamins in basal cell carcinomas of the skin. Br. J. Cancer 84, 512–519 (2001).
Tilli, C. M. L. J., Ramaekers, F. C. S., Broers, J. L. V., Hutchison, C. J. & Neumann, H. A. M. Lamin expression in normal human skin, actinic keratosis, squamous cell carcinoma and basal cell carcinoma. Br. J. Dermatol. 148, 102–109 (2003).
Bengtsson, S. et al. Large-scale proteomics analysis of human ovarian cancer for biomarkers. J. Proteome Res. 6, 1440–1450 (2007).
Coradeghini, R. et al. Differential expression of nuclear lamins in normal and cancerous prostate tissues. Oncol. Rep. 15, 609–613 (2006).
Doherty, J. A. et al. ESR1/SYNE1 polymorphism and invasive epithelial ovarian cancer risk: an ovarian cancer association consortium study. Cancer Epidemiol. Biomarkers Prev. 19, 245–250 (2010).
Sjöblom, T. et al. The consensus coding sequences of human breast and colorectal cancers. Science 314, 268–274 (2006).
Zhao, Z.-R. et al. Increased serum level of Nup88 protein is associated with the development of colorectal cancer. Med. Oncol. 24 Aug 2011 (doi:10.1007/s12032-011-0047-1).
Acknowledgements
This work was supported by the US National Institutes of Heath (R01GM61275) and the Huntsman Cancer Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
DATABASES
FURTHER INFORMATION
Glossary
- Aneuploidy
-
An alteration in the number of chromosomes within a cell.
- Farnesyltransferase inhibitors
-
Drugs that block the post-translational attachment of farnesyl isoprenoid lipid to the cysteine residues of a CAAX motif on proteins, thereby preventing farnesyl-mediated localization of these proteins to cell membranes.
- Spindle assembly checkpoint
-
A quality control mechanism that blocks chromosome segregation until all chromosomes are properly anchored to the microtubule spindle apparatus at kinetochores.
- Kinetochores
-
Proteinaceous structures assembled at the centromere to bridge the connection between chromosomes and mitotic microtubule spindle apparatus.
- APC/C
-
Anaphase-promoting complex (also known as the cyclosome). A protein complex that catalyses the ubiquitylation of mitotic factors such as separase and cyclins to target them for degradation, allowing regulated cell cycle progression.
- Chromosome instability
-
Changing chromosome number or structure (translocations, deletions and duplications) resulting in aberrant DNA content.
- Boundary activity
-
The ability to block either enhancer activity or repressive effects of heterochromatin.
- Progeria
-
A rare genetic disease that is characterized by the early onset of ageing symptoms in children.
- DNA damage-induced foci
-
Transiently formed sites in the nucleus where cellular DNA damage response machinery is concentrated and recruited to DNA lesions.
Rights and permissions
About this article
Cite this article
Chow, KH., Factor, R. & Ullman, K. The nuclear envelope environment and its cancer connections. Nat Rev Cancer 12, 196–209 (2012). https://doi.org/10.1038/nrc3219
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3219
This article is cited by
-
Intracellular remodeling associated with endoplasmic reticulum stress modifies biomechanical compliance of bladder cells
Cell Communication and Signaling (2023)
-
Deformation of the nucleus by TGFβ1 via the remodeling of nuclear envelope and histone isoforms
Epigenetics & Chromatin (2022)
-
Inner nuclear membrane protein TMEM201 promotes breast cancer metastasis by positive regulating TGFβ signaling
Oncogene (2022)
-
Weight-bearing activity impairs nuclear membrane and genome integrity via YAP activation in plantar melanoma
Nature Communications (2022)
-
A novel hierarchical targeting and controllable smart nanoparticles for enhanced in situ nuclear photodynamic therapy
Nano Research (2022)