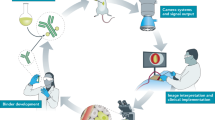
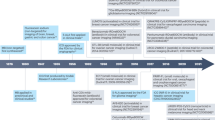
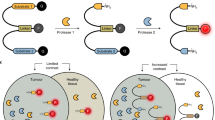
Abstract
A glowing new era in cancer surgery may be dawning. Using fluorescently labelled markers, surgical molecular navigation means that tumours and nerves can be displayed in real time intra-operatively in contrasting pseudocolours, which allows more complete tumour resection while preserving important structures. These advances can potentially cause a paradigm shift in cancer surgery, improving patient outcome and decreasing overall health-care costs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
National Cancer Institute. Previous version: SEER cancer statistics review, 1975–2005 [online], (2008).
Haque, R., Contreras, R., McNicoll, M. P., Eckberg, E. C. & Petitti, D. B. Surgical margins and survival after head and neck cancer surgery. BMC Ear Nose Throat Disord. 16, 2 (2006).
Singletary, S. Surgical margins in patients with early-stage breast cancer treated with breast conservation therapy. Am. J. Surg. 184, 383–393 (2002).
Meric, F. et al. Positive surgical margins and ipsilateral breast tumor recurrence predict disease-specific survival after breast-conserving therapy. Cancer 97, 926–933 (2003).
Snijder, R., de la Riviere, A., Elbers, H. & van den Bosch, J. Survival in resected stage I lung cancer with residual tumor at the bronchial resection margin. Ann. Thorac. Surg. 65, 212–216 (1998).
Nagtegaal, I. D. & Quirke, P. What is the role for the circumferential margin in the modern treatment of rectal cancer? J. Clin. Oncol. 26, 303–312 (2008).
Dotan, Z. et al. Positive surgical margins in soft tissue following radical cystectomy for bladder cancer and cancer specific survival. J. Urol. 178, 2308–2312 (2007).
Wieder, J. A. & Soloway, M. S. Incidence, etiology, location, prevention and treatment of positive surgical margins after radical prostatectomy for prostate cancer. J. Urol. 160, 299–315 (1998).
Kumar, A., Puri, R., Gadgil, P. V. & Jatoi, I. Sentinel lymph node biopsy in primary breast cancer: window to management of the axilla. World J. Surg. 36, 1453–1459 (2012).
Wong, S. L. et al. Sentinel lymph node biopsy for melanoma: American Society of Clinical Oncology and Society of Surgical Oncology joint clinical practice guideline. J. Clin. Oncol. 30, 2912–2918 (2012).
Hutteman, M. et al. Randomized, double-blind comparison of indocyanine green with or without albumin premixing for near-infrared fluorescence imaging of sentinel lymph nodes in breast cancer patients. Breast Cancer Res. Treat. 127, 163–170 (2011).
van der Vorst, J. R. et al. Randomized comparison of near-infrared fluorescence imaging using indocyanine green and 99m technetium with or without patent blue for the sentinel lymph node procedure in breast cancer patients. Ann. Surg. Oncol. 19, 4104–4111 (2012).
Emerson, D. K. et al. A receptor-targeted fluorescent radiopharmaceutical for multireporter sentinel lymph node imaging. Radiology 265, 186–193 (2012).
Joseph, M., Phillips, M., Farrell, T. M. & Rupp, C. C. Can residents safely and efficiently be taught single incision laparoscopic cholecystectomy? J. Surg. Educ. 69, 468–472 (2012).
Kandil, E. H., Noureldine, S. I., Yao, L. & Slakey, D. P. Robotic transaxillary thyroidectomy: an examination of the first one hundred cases. J. Am. Coll. Surg. 214, 558–564 (2012).
Lee, J. C., Jang, H. D. & Shin, B. J. Learning curve and clinical outcomes of minimally invasive transforaminal lumbar interbody fusion: our experience in 86 consecutive cases. Spine 37, 1548–1557 (2012).
Pappo, I. et al. Diagnostic performance of a novel device for real-time margin assessment in lumpectomy specimens. J. Surg. Res. 160, 277–281 (2010).
Rivera, R. J., Holmes, D. R. & Tafra, L. Analysis of the impact of intraoperative margin assessment with adjunctive use of MarginProbe versus standard of care on tissue volume removed. Int. J. Surg. Oncol. 2012, 868623 (2012).
Dotan, Z. A. et al. Detection of prostate cancer by radio-frequency near-field spectroscopy in radical prostatectomy ex vivo specimens. Prostate Cancer Prostatic Dis. 16, 73–78 (2012).
Pavlova, I. et al. Multiphoton microscopy and microspectroscopy for diagnostics of inflammatory and neoplastic lung. J. Biomed. Opt. 17, 036014 (2012).
Pierce, M. C. et al. Accuracy of in vivo multimodal optical imaging for detection of oral neoplasia. Cancer Prev. Res. 5, 801–809 (2012).
Gurtner, G. C. et al. Intraoperative laser angiography using the SPY system: review of the literature and recommendations for use. Ann. Surg. Innov. Res. 7, 1 (2013).
Kuroda, K. et al. Intra-arterial injection fluorescein videoangiography in aneurysm surgery. Neurosurgery 72, 141–150 (2012).
Shin, D., Vigneswaran, N., Gillenwater, A. & Richards-Kortum, R. Advances in fluorescence imaging techniques to detect oral cancer and its precursors. Future Oncol. 6, 1143–1154 (2010).
Foersch, S. et al. Confocal laser endomicroscopy for diagnosis and histomorphologic imaging of brain tumors in vivo. PLoS ONE 7, e41760 (2012).
Vila, P. M. et al. Discrimination of benign and neoplastic mucosa with a High-Resolution Microendoscope (HRME) in head and neck cancer. Ann. Surg. Oncol. 19, 3534–3539 (2012).
Barker, F. G. et al. Survival and functional status after resection of recurrent glioblastoma multiforme. Neurosurgery 42, 709–720 (1998).
Swindle, P. et al. Do margins matter? The prognostic significance of positive surgical margins in radical prostatectomy specimens. J. Urol. 179, S47–S51 (2008).
Warner, J. N. et al. Impact of margin status at 37 months after robot assisted radical prostatectomy. Can. J. Urol. 18, 6043–6049 (2011).
Chalfin, H. J. et al. Impact of surgical margin status on prostate-cancer-specific mortality. BJU Int. 110, 1684–1689 (2012).
Pleijhuis, R. G. et al. Obtaining adequate surgical margins in breast-conserving therapy for patients with early-stage breast cancer: current modalities and future directions. Ann. Surg. Oncol. 16, 2717–2730 (2009).
Atkins, J. et al. Positive margin rates following breast-conserving surgery for stage I-III breast cancer: palpable versus nonpalpable tumors. J. Surg. Res. 177, 109–115 (2012).
Cutter, J. L. et al. Topical application of activity-based probes for visualization of brain tumor tissue. PLoS ONE 7, e33060 (2012).
Verdoes, M. et al. A nonpeptidic cathepsin S activity-based probe for noninvasive optical imaging of tumor-associated macrophages. Chem. Biol. 19, 619–628 (2012).
Sheth, R. A. et al. Improved detection of ovarian cancer metastases by intraoperative quantitative fluorescence protease imaging in a pre-clinical model. Gynecol. Oncol. 112, 616–622 (2009).
Nguyen, Q. T. et al. Surgery with molecular fluorescence imaging using activatable cell-penetrating peptides decreases residual cancer and improves survival. Proc. Natl Acad. Sci. USA 107, 4317–4322 (2010).
Jiang, T. et al. Tumor imaging by means of proteolytic activation of cell-penetrating peptides. Proc. Natl Acad. Sci. USA 101, 17867–17872 (2004).
Olson, E. S. et al. In vivo characterization of activatable cell penetrating peptides for targeting protease activity in cancer. Integr. Biol. 1, 382–393 (2009).
Aguilera, T. A., Olson, E. S., Timmers, M. M., Jiang, T. & Tsien, R. Y. Systemic in vivo distribution of activatable cell penetrating peptides is superior to that of cell penetrating peptides. Integr. Biol. 1, 371–381 (2009).
Savariar, E. N. et al. Real-time in vivo molecular detection of primary tumors and metastases with ratiometric activatable cell-penetrating peptides. Cancer Res. 73, 855–864 (2013).
Nakajima, T. et al. Targeted, activatable, in vivo fluorescence imaging of prostate-specific membrane antigen (PSMA) positive tumors using the quenched humanized J591 antibody-indocyanine green (ICG) conjugate. Bioconjug. Chem. 22, 1700–1705 (2011).
Tran Cao, H. S. et al. Tumor-specific fluorescence antibody imaging enables accurate staging laparoscopy in an orthotopic model of pancreatic cancer. Hepatogastroenterology 59, 1994–1999 (2012).
McElroy, M. et al. Imaging of primary and metastatic pancreatic cancer using a fluorophore-conjugated anti-CA19-9 antibody for surgical navigation. World J. Surg. 32, 1057–1066 (2008).
Hall, M. A. et al. Comparison of mAbs targeting epithelial cell adhesion molecule for the detection of prostate cancer lymph node metastases with multimodal contrast agents: quantitative small-animal PET/CT and NIRF. J. Nucl. Med. 53, 1427–1437 (2012).
Rosbach, K. J., Williams, M. D., Gillenwater, A. M. & Richards-Kortum, R. R. Optical molecular imaging of multiple biomarkers of epithelial neoplasia: epidermal growth factor receptor expression and metabolic activity in oral mucosa. Transl Oncol. 5, 160–171 (2012).
Urano, Y. et al. Rapid cancer detection by topically spraying a γ-glutamyltranspeptidase-activated fluorescent probe. Sci. Transl Med. 3, 110ra119 (2011).
van Dam, G. M. et al. Intraoperative tumor-specific fluorescence imaging in ovarian cancer by folate receptor-α targeting: first in-human results. Nature Med. 17, 1315–1319 (2011).
Stummer, W. et al. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol. 7, 392–401 (2006).
Stummer, W. et al. Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J. Neurosurg. 93, 1003–1013 (2000).
Kalli, K. R. et al. Folate receptor α as a tumor target in epithelial ovarian cancer. Gynecol. Oncol. 108, 619–626 (2008).
Schafer, C. et al. γ-glutamyl transferase expression in higher-grade astrocytic glioma. Acta Oncol. 40, 529–535 (2001).
Floeth, F. W. et al. Comparison of 18F-FET PET and 5-ALA fluorescence in cerebral gliomas. Eur. J. Nucl. Med. Mol. Imaging 38, 731–741 (2011).
Sun, C. et al. Tumor-targeted drug delivery and MRI contrast enhancement by chlorotoxin-conjugated iron oxide nanoparticles. Nanomed. 3, 495–505 (2008).
Sun, C. et al. In vivo MRI detection of gliomas by chlorotoxin-conjugated superparamagnetic nanoprobes. Small 4, 372–379 (2008).
Olson, E. S. et al. Activatable cell penetrating peptides linked to nanoparticles as dual probes for in vivo fluorescence and MR imaging of proteases. Proc. Natl Acad. Sci. USA 107, 4311–4316 (2010).
Gibbs-Strauss, S. L. et al. Nerve-highlighting fluorescent contrast agents for image-guided surgery. Mol. Imaging 10, 91–101 (2011).
Whitney, M. et al. Fluorescent peptides highlight peripheral nerves during surgery in mice. Nature Biotech. 29, 352–356 (2011).
Sturm, M. B. et al. Targeted imaging of esophageal neoplasia with a fluorescently labeled peptide: first-in-human results. Sci. Transl Med. 5, 184ra61 (2013).
Blum, G., von Degenfeld, G., Merchant, M. J., Blau, H. M. & Bogyo, M. Noninvasive optical imaging of cysteine protease activity using fluorescently quenched activity-based probes. Nature Chem. Biol. 3, 668–677 (2007).
Figueiredo, J. L., Alencar, H., Weissleder, R. & Mahmood, U. Near infrared thoracoscopy of tumoral protease activity for improved detection of peripheral lung cancer. Int. J. Cancer 118, 2672–2677 (2006).
Kelly, K., Alencar, H., Funovics, M., Mahmood, U. & Weissleder, R. Detection of invasive colon cancer using a novel, targeted, library-derived fluorescent peptide. Cancer Res. 64, 6247–6251 (2004).
Veiseh, M. et al. Tumor paint: a chlorotoxin:Cy5.5 bioconjugate for intraoperative visualization of cancer foci. Cancer Res. 67, 6882–6888 (2007).
Stroud, M. R., Hansen, S. J. & Olson, J. M. In vivo bio-imaging using chlorotoxin-based conjugates. Curr. Pharm. Des. 17, 4362–4371 (2011).
Ye, Y. et al. Near-infrared fluorescent divalent RGD ligand for integrin αvβ3-targeted optical imaging. Bioorg. Med. Chem. Lett. 22, 5405–5409 (2012).
Gao, J. et al. A novel clinically translatable fluorescent nanoparticle for targeted molecular imaging of tumors in living subjects. Nano Lett. 12, 281–286 (2012).
Zhu, L. et al. Dual-functional, receptor-targeted fluorogenic probe for in vivo imaging of extracellular protease expressions. Bioconjugate Chem. 22, 1001–1005 (2011).
Wu, A. P. et al. Improved facial nerve identification with novel fluorescently labeled probe. Laryngoscope 121, 805–810 (2011).
Cotero, V. E. et al. Intraoperative fluorescence imaging of peripheral and central nerves through a myelin-selective contrast agent. Mol. Imaging Biol. 14, 708–717 (2012).
Tsien, R. Y. & Harootunian, A. T. Practical design criteria for a dynamic ratio imaging system. Cell Calcium 11, 93–109 (1990).
Orosco, R. K., Tsien, R. Y. & Nguyen, Q. T. Fluorescence imaging in surgery. IEEE Rev. Biomed. Eng. 6, 178–187 (2013).
Taruttis, A. & Ntziachristos, V. Translational optical imaging. Am. J. Roentgenol. 199, 263–271 (2012).
Nunn, A. D. The cost of developing imaging agents for routine clinical use. Invest. Radiol. 41, 206–212 (2006).
Mitsunaga, M. et al. Fluorescence endoscopic detection of murine colitis-associated colon cancer by topically applied enzymatically rapid-activatable probe. Gut 62, 1179–1186 (2013).
Ashitate, Y. et al. Simultaneous assessment of luminal integrity and vascular perfusion of the gastrointestinal tract using dual-channel near-infrared fluorescence. Mol. Imaging 11, 301–308 (2012).
Acknowledgements
The authors would like to thank S. Howell and L. Simon for helpful discussions on the regulatory aspects discussed in this manuscript, and M. Whitney for helpful scientific discussions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Approved
-
An approved drug is a drug that has been authorized for a therapeutic use by a ruling authority. In Europe, the European Medicines Agency (EMA) is the ruling authority. In the United States, the Food and Drug Administration (FDA), an agency within the Department of Health and Human Services, provides this approval. A list of FDA-approved drugs is updated regularly on the FDA Orange Book website (see Further information).
- Autofluorescence
-
Fluorescence that is inherent to different tissues (such as muscle, fat or liver) in the absence of any exogenously applied compounds.
- Chlorotoxin
-
A peptide that is found in the venom of the scorpion Leiurus quinquestriatus that blocks small-conductance chloride channels. Chlorotoxin binds preferentially to gliomas and tumours of neuroectodermal origin.
- Indication
-
A drug is approved by the US Food and Drug Administration (FDA) or the European Medicines Agency (EMA) for certain uses or indications following approved Phase I, II and III clinical trials. These indications are supplied with the package inserts for the drugs.
- Infrared reflectivity
-
The fraction of incident infrared light that is reflected, which can vary between different tissues.
- Intratumour microheterogeneity
-
The idea that within a given neoplasm, different tumour cells may display differences in genetic mutations and/or protein expression that may confer advantages and disadvantages for metastatic potential or response to chemotherapy or radiotherapy.
- Lymphoedema
-
A condition of localized fluid retention and tissue swelling caused by a compromised lymphatic system. Lymphoedema is a common consequence of lymph node removal (for example, during neck dissection or axillary lymph node dissection) in the surgical treatment of cancer.
- Medical device
-
An instrument that is used to diagnose or treat disease or other medical conditions and that does not achieve its purpose through chemical action within or on the body. In the United States, the Food and Drug Administration (FDA) is responsible for medical device evaluation, through premarket evaluation (510(k)) to show similarity with a previously approved device or through premarket approval (PMA) applications. PMA approval for a new device is based on a determination by the FDA that the PMA contains sufficient valid scientific evidence to ensure that the device is safe and effective for its intended use or uses.
- Micro-anatomical cyto-architecture
-
The shapes and sizes of cells, nuclei, organelles and extracellular structures, which are used to distinguish tumour from normal tissue at microscopic resolution.
- Nonspecific hydrophobic stickiness
-
Nonspecific binding to tissue of hydrophobic molecules such as most near-infrared dyes (which emit at wavelengths >700nm), thus typically resulting in increased background fluorescence.
- Off-label
-
The clinical practice of prescribing pharmaceuticals for an unapproved indication or in an unapproved age group, at an unapproved dose or through an unapproved form of administration. After a drug has been approved for one purpose, licensed physicians are able to prescribe it for other purposes that in their professional judgment are both safe and effective, and are not limited to the official, approved indications.
- Open-field surgery
-
A type of surgery in which the surgeon's view of the patient's tissue is through direct visualization through a large skin incision. This is in contrast to minimally invasive surgery (that is, laparoscopic surgery, endoscopic surgery or robotic surgery) in which the surgeon views the patient's tissues through fibre-optic probes or cameras, which are inserted through natural orifices or small skin incisions and the images are displayed on a computer screen.
- Quantum dots
-
Inorganic nanocrystals that typically have a diameter of 2–8 nm. Most are composed of an inner semiconductor core of CdSe, an outer shell of ZnS, and an organic coating to make the quantum dot biocompatible. Quantum dots can be engineered to emit fluorescent light in the ultraviolet to infrared spectrum by varying their size.
- Raman scattering
-
A spectroscopic technique that differentiates different tissue types depending on the infrared spectra of different molecules (such as fat and collagen) within that tissue.
- Ratiometric imaging
-
Measurement of the ratio of fluorescence intensities at two wavelengths. When the response of the fluorescent probe to the desired signal (for example, protease activity) is markedly different at two wavelengths, calculating the ratio of the fluorescence at those wavelengths isolates the desired signal from confounding factors such as the absolute concentration of the fluorescent probe, the thickness of tissue, the intensity of illumination and the sensitivity of detection, which all affect intensities at both wavelengths by equal proportions. Ratiometric activatable cell-penetrating peptides (RACPPs)40 use pairs of dyes such as Cy5 and Cy7 as donor and acceptor dyes for fluorescence resonance energy transfer, thus enabling emission ratiometric discrimination of protease activity.
- Sentinel lymph node
-
(SLN). The first draining lymph node for a given anatomical site. For breast cancer, the SLNs are located in the axilla.
Rights and permissions
About this article
Cite this article
Nguyen, Q., Tsien, R. Fluorescence-guided surgery with live molecular navigation — a new cutting edge. Nat Rev Cancer 13, 653–662 (2013). https://doi.org/10.1038/nrc3566
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3566
This article is cited by
-
“All in one” nanoprobe Au-TTF-1 for target FL/CT bioimaging, machine learning technology and imaging-guided photothermal therapy against lung adenocarcinoma
Journal of Nanobiotechnology (2024)
-
Deep learning for tumor margin identification in electromagnetic imaging
Scientific Reports (2023)
-
Probes and nano-delivery systems targeting NAD(P)H:quinone oxidoreductase 1: a mini-review
Frontiers of Chemical Science and Engineering (2023)
-
The Evolution of Fluorescence-Guided Surgery
Molecular Imaging and Biology (2023)
-
Identification of a Suitable Untargeted Agent for the Clinical Translation of ABY-029 Paired-Agent Imaging in Fluorescence-Guided Surgery
Molecular Imaging and Biology (2023)