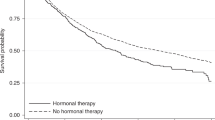
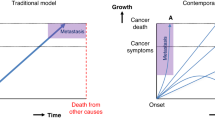
Abstract
The increase and diversity of clinical trial data has resulted in a greater reliance on statistical analyses to discern value. Assessing differences between two similar survival curves can pose a challenge for those without formal training in statistical interpretation; therefore, there has been an increased reliance on hazard ratios often to the exclusion of more-traditional survival measures. However, because a hazard ratio lacks dimensions it can only inform the reader about the reliability and uniformity of the data. It does not provide practitioners with quantitative values they can use, nor does it provide information they can discuss with patients. Motivated by a non-scientific poll of oncologists in training and those with board certification that suggested only a limited understanding of the derivation of hazard ratios we undertook this presentation of hazard ratios: a measure of treatment efficacy that is increasingly used and often misused.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hernán, M. A. The hazards of hazard ratios. Epidemiology 21, 13–15 (2010).
Kaplan, E. L. & Meier, P. Nonparametric estimation from incomplete observations. J. Am. Stat. Assoc. 53, 457–481 (1958).
Escudier, B. et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 356, 125–134 (2007).
Motzer, R. J. et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N. Engl. J. Med. 356, 115–124 (2007).
Armitage, P., Berry, G. & Matthews, J. N. S. Statistical Methods in Medical Research, 4th edn, 568–590 (Blackwell Science Ltd, Oxford, 2002).
Kattan, M. W. Evaluating a new marker's predictive contribution. Clin. Cancer Res. 10, 822–824 (2004).
Raymond, E. et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 501–513 (2011).
Yao, J. C. et al. Everolimus for advanced pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 514–523 (2011).
Baselga, J. et al. Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N. Engl. J. Med. http://dx.doi.org/10.1056/NEJMoa1109653.
Mantel, N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother. Rep. 50, 163–170 (1966).
Peto, R. & Peto, J. Asymptotically efficient rank invariant test procedures. J. Roy. Stat. Soc. Ser. A (General) 135, 185–207 (1972).
Cox, D. R. Regression models and life tables. J. Roy. Stat. Soc. Ser. B (Methogological) 34, 187–220 (1972).
Fan, J. & Li, R. Variable selection for Cox's proportional hazards model and frailty model. Ann. Stat. 30, 74–99 (2002).
Fisher, L. D. & Lin, D. Y. Time-dependent covariates in the Cox proportional-hazards regression model. Annu. Rev. Public Health 20, 145–157 (1999).
SAS/STAT® (SAS Institute Inc. Cary, NC, USA).
XLSTAT (Addinsoft, New York, NY, USA).
MedCalc version 12.0 (MedCalc Software, Mariakerke, Belgium).
Miettinen, O. S. Stratification by a multivariate confounder score. Am. J. Epidemiol. 104, 609–620 (1976).
Acknowledgements
K. B. Blagoev would like to acknowledge that this work was supported in part by the National Science Foundation. Any opinion, finding, conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Author information
Authors and Affiliations
Contributions
All authors contributed to researching the data for the article and had a substantial contribution to discussion of the content. K. B. Blagoev and T. Fojo wrote the manuscript and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Blagoev, K., Wilkerson, J. & Fojo, T. Hazard ratios in cancer clinical trials—a primer. Nat Rev Clin Oncol 9, 178–183 (2012). https://doi.org/10.1038/nrclinonc.2011.217
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2011.217
This article is cited by
-
Treatment effect of radiofrequency ablation versus liver transplantation and surgical resection for hepatocellular carcinoma within Milan criteria: a population-based study
European Radiology (2021)
-
Potential Bias Associated with Modeling the Effectiveness of Healthcare Interventions in Reducing Mortality Using an Overall Hazard Ratio
PharmacoEconomics (2020)
-
Assessment of proportional hazard assumption in aggregate data: a systematic review on statistical methodology in clinical trials using time-to-event endpoint
British Journal of Cancer (2018)
-
Early Benefit Assessments in Oncology in Germany: How Can a Clinically Relevant Endpoint Not Be Relevant to Patients?
Drugs in R&D (2015)