Abstract
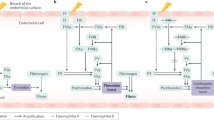
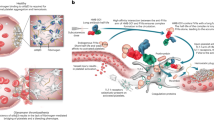
Haemophilia is a rare disease for which the approved therapeutic options have remained virtually unchanged for 50 years. In the past decade, however, there has been an explosion of innovation in the treatment options that are either in development or have been approved for haemophilia, including engineered clotting factors and an extensive pipeline of new approaches and modalities. Several of these new modalities, especially gene therapy, demonstrate proof of principle in haemophilia but could have broader applications. These advances, in combination with better diagnostics, are now enabling clinicians to improve the standard of care for people with haemophilia. The different mechanisms of action and modifications used in these therapies have implications for their safe and efficacious use, which must be balanced with their therapeutic utility. This Review focuses on the biological aspects of the most advanced and innovative approaches for haemophilia treatment and considers their future use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lee, C. A., Berntorp, E. & Hoots, K. Textbook of Hemophilia. 2nd edn (Wiley-Blackwell, 2010).
Doolittle, R. F. Step-by-step evolution of vertebrate blood coagulation. Cold Spring Harb. Symp. Quant. Biol. 74, 35–40 (2009).
Srivastava, A. et al. Guidelines for the management of hemophilia. Haemophilia 19, e1–e47 (2013).
Hay, C. R. & DiMichele, D. M. The principal results of the International Immune Tolerance Study: a randomized dose comparison. Blood 119, 1335–1344 (2012). This paper summarizes the results of the largest, prospective study comparing two dose regimens of ITI in patients with high anti-drug antibody titres and puts these data into the context of historical studies and reports.
Dargaud, Y. et al. Achievements, challenges and unmet needs for haemophilia patients with inhibitors: report from a symposium in Paris, France on 20 November 2014. Haemophilia 22 (Suppl. 1), 1–24 (2016).
Hoffman, M. & Monroe, D. M. 3rd. A cell-based model of hemostasis. Thromb. Haemost. 85, 958–965 (2001). This paper provides a major advance in the understanding of coagulation pathways as it recognizes the key roles of different clotting factors localized to different cell surfaces, which has implications for the multitude of therapeutic options in haemophilia treatment.
Smith, S. A. The cell-based model of coagulation. J. Vet. Emerg. Crit. Care 19, 3–10 (2009).
Ivanciu, L. & Stalker, T. J. Spatiotemporal regulation of coagulation and platelet activation during the hemostatic response in vivo. J. Thromb. Haemost. 13, 1949–1959 (2015).
Sehgal, A. et al. An RNAi therapeutic targeting antithrombin to rebalance the coagulation system and promote hemostasis in hemophilia. Nat. Med. 21, 492–497 (2015).
Chowdary, P. et al. Safety and pharmacokinetics of anti-TFPI antibody (concizumab) in healthy volunteers and patients with hemophilia: a randomized first human dose trial. J. Thromb. Haemost. 13, 743–754 (2015).
Polderdijk, S. G. et al. Design and characterization of an APC-specific serpin for the treatment of hemophilia. Blood 129, 105–113 (2017).
Turecek, P. L., Varadi, K., Gritsch, H. & Schwarz, H. P. FEIBA: mode of action. Haemophilia 10 (Suppl. 2), 3–9 (2004).
Agerso, H. et al. Recombinant human factor VIIa (rFVIIa) cleared principally by antithrombin following intravenous administration in hemophilia patients. J. Thromb. Haemost. 9, 333–338 (2011).
Wyseure, T., Mosnier, L. O. & von Drygalski, A. Advances and challenges in hemophilic arthropathy. Semin. Hematol. 53, 10–19 (2016). This paper describes the current understanding of haemophilic arthopathy, which can develop despite clotting factor replacement, and describes the gaps in our understanding of the causative pathobiology, as well as the novel strategies involving diagnostic biomarkers and point-of-care imaging techniques that are being explored.
Oldenburg, J. Optimal treatment strategies for hemophilia: achievements and limitations of current prophylactic regimens. Blood 125, 2038–2044 (2015). This paper reviews the approaches used in determining appropriate prophylaxis regimens, the dynamics of developing joint arthopathy and factors to consider in individualized therapy, including patient-to-patient variation, for joint bleeds at given factor levels.
Toole, J. J. et al. Molecular cloning of a cDNA encoding human antihaemophilic factor. Nature 312, 342–347 (1984).
Vehar, G. A. et al. Structure of human factor VIII. Nature 312, 337–342 (1984).
Jankowski, M. A. et al. Defining 'full-length' recombinant factor VIII: a comparative structural analysis. Haemophilia 13, 30–37 (2007).
Sandberg, H. et al. Structural and functional characterization of B-domain deleted recombinant factor VIII. Semin. Hematol. 38, 4–12 (2001).
Toole, J. J. et al. A large region (approximately equal to 95 kDa) of human factor VIII is dispensable for in vitro procoagulant activity. Proc. Natl Acad. Sci. USA 83, 5939–5942 (1986).
Peters, R. T. et al. Biochemical and functional characterization of a recombinant monomeric factor VIII-Fc fusion protein. J. Thromb. Haemost. 11, 132–141 (2013).
Dumont, J. A. et al. Prolonged activity of a recombinant factor VIII-Fc fusion protein in hemophilia A mice and dogs. Blood 119, 3024–3030 (2012).
Turecek, P. L. et al. BAX 855, a PEGylated rFVIII product with prolonged half-life. Development, functional and structural characterisation. Hamostaseologie 32 (Suppl. 1), 29–38 (2012).
Mei, B. et al. Rational design of a fully active, long-acting PEGylated factor VIII for hemophilia A treatment. Blood 116, 270–279 (2010).
Stennicke, H. R. et al. A novel B-domain O-glycoPEGylated FVIII (N8-GP) demonstrates full efficacy and prolonged effect in hemophilic mice models. Blood 121, 2108–2116 (2013).
Shen, B. W. et al. The tertiary structure and domain organization of coagulation factor VIII. Blood 111, 1240–1247 (2008).
Pipe, S. W., Montgomery, R. R., Pratt, K. P., Lenting, P. J. & Lillicrap, D. Life in the shadow of a dominant partner: the FVIII-VWF association and its clinical implications for hemophilia A. Blood 128, 2007–2016 (2016). This paper describes the biology of VWF and FVIII from biosynthesis to clearance and relates this to the limit on half-life extension of FVIII seen in clinical studies of EHL FVIII despite a variety of approaches.
Podust, V. N. et al. Extension of in vivo half-life of biologically active molecules by XTEN protein polymers. J. Control. Release 240, 52–66 (2016).
Mahlangu, J. et al. Phase 3 study of recombinant factor VIII Fc fusion protein in severe hemophilia A. Blood 123, 317–325 (2014).
Tiede, A. et al. Enhancing the pharmacokinetic properties of recombinant factor VIII: first-in-human trial of glycoPEGylated recombinant factor VIII in patients with hemophilia A. J. Thromb. Haemost. 11, 670–678 (2013).
Coyle, T. E. et al. Phase I study of BAY 94–9027, a PEGylated B-domain-deleted recombinant factor VIII with an extended half-life, in subjects with hemophilia A. J. Thromb. Haemost. 12, 488–496 (2014).
Konkle, B. A. et al. Pegylated, full-length, recombinant factor VIII for prophylactic and on-demand treatment of severe hemophilia A. Blood 126, 1078–1085 (2015).
Liu, T. et al. Prolonged efficacy in hemophilia A mouse bleeding models of a recombinant FVIII-XTEN/D'D3 heterodimer with four-fold extended half-life in circulation [abstract]. Haemophilia 20 (Suppl. 3), 76 (2014).
Chhabra, E. S. et al. Application of in silico antigenicity prediction methods to avoid neo-epitopes during the designing of BIIB073, a next generation long acting recombinant Factor VIII (FVIII) molecule [abstract]. Haemophilia 22 (Suppl. 4), 18 (2016).
Pestel, S. et al. Half-life extension of FVIII by coadministration of a recombinant D'D3 albumin fusion protein [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), OC 10.5 (2017).
Patarroyo-White, S. et al. A FVIII/VWF chimeric protein with VWF independent pharmacokinetic properties [abstract]. J. Thromb. Haemost. 13, 254 (2015).
Peters, R. T. et al. Prolonged activity of factor IX as a monomeric Fc fusion protein. Blood 115, 2057–2064 (2010).
Metzner, H. J., Weimer, T., Kronthaler, U., Lang, W. & Schulte, S. Genetic fusion to albumin improves the pharmacokinetic properties of factor IX. Thromb. Haemost. 102, 634–644 (2009).
Ostergaard, H. et al. Prolonged half-life and preserved enzymatic properties of factor IX selectively PEGylated on native N-glycans in the activation peptide. Blood 118, 2333–2341 (2011).
Bjorkman, S., Carlsson, M. & Berntorp, E. Pharmacokinetics of factor IX in patients with haemophilia B. Methodological aspects and physiological interpretation. Eur. J. Clin. Pharmacol. 46, 325–332 (1994). This early, pioneering paper relates the pharmacokinetics of FIX to the physiological implications of extravascular distribution, which has recently become appreciated to have therapeutic implications.
Cheung, W. F. et al. Identification of the endothelial cell binding site for factor IX. Proc. Natl Acad. Sci. USA 93, 11068–11073 (1996).
Stern, D. M., Knitter, G., Kisiel, W. & Nawroth, P. P. In vivo evidence of intravascular binding sites for coagulation factor IX. Br. J. Haematol. 66, 227–232 (1987).
Cooley, B. et al. Prophylactic efficacy of BeneFIX versus Alprolix in hemophilia B mice. Blood 128, 286–292 (2016).
Collins, P. W. et al. Recombinant long-acting glycoPEGylated factor IX in hemophilia B: a multinational randomized phase 3 trial. Blood 124, 3880–3886 (2014).
Levy, H., Nichols, T., Merricks, E., Raymer, R. & Hetherington, A. Pharmacokinetic and activity levels achieved with daily subcutaneously administered CB 2679d/ISU304 in hemophilia B dogs [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), OC 10.3 (2017).
van der Flier, A. et al. The pharmacokinetic profiles of intravenously and subcutaneously administered recombinant factor IX Fc-XTEN in cynomolgus monkeys [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), OC 40.2 (2017).
Patel-Hett, S. et al. A monoclonal antibody with TFPI neutralizing activity improves the coagulation parameters of hemostatic assays performed with hemophilic whole blood [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), PB 1124 (2017).
Yegneswaran, S. et al. BAY 1093884 binds to the kunitz 1 and 2 domain interface of tissue factor pathway inhibitor and inhibits its function [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), PB 892 (2017).
Pasi, K. J. et al. Fitusiran, an investigational RNAi therapeutic targeting antithrombin for the treatment of hemophilia: interim results from a phase 2 extension study in patients with hemophilia A or B with and without inhibitors [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), ASY 01.2 (2017).
Ellery, P. E. et al. A balance between TFPI and thrombin-mediated platelet activation is required for murine embryonic development. Blood 125, 4078–4084 (2015).
Jalbert, L. R. et al. Inactivation of the gene for anticoagulant protein C causes lethal perinatal consumptive coagulopathy in mice. J. Clin. Invest. 102, 1481–1488 (1998).
Ishiguro, K. et al. Complete antithrombin deficiency in mice results in embryonic lethality. J. Clin. Invest. 106, 873–878 (2000).
Hilden, I. et al. Hemostatic effect of a monoclonal antibody mAb 2021 blocking the interaction between FXa and TFPI in a rabbit hemophilia model. Blood 119, 5871–5878 (2012).
Kitazawa, T. et al. A bispecific antibody to factors IXa and X restores factor VIII hemostatic activity in a hemophilia A model. Nat. Med. 18, 1570–1574 (2012).
Lenting, P. J., Denis, C. V. & Christophe, O. D. Emicizumab, a bispecific antibody recognizing coagulation factors IX and X: how does it actually compare to factor VIII? Blood 130, 2463–2468 (2017).
Oldenburg, J. et al. Emicizumab prophylaxis in hemophilia A with inhibitors. N. Engl. J. Med. 377, 809–818 (2017).
Sampei, Z. et al. Identification and multidimensional optimization of an asymmetric bispecific IgG antibody mimicking the function of factor VIII cofactor activity. PLoS ONE 8, e57479 (2013).
Kitazawa, T. et al. Factor VIIIa-mimetic cofactor activity of a bispecific antibody to factors IX/IXa and X/Xa, emicizumab, depends on its ability to bridge the antigens. Thromb. Haemost. 117, 1348–1357 (2017).
Leksa, N. et al. Identification of FIXa- and FX-specific antibodies for the generation of bispecific antibodies with FVIIIa-like activity [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), OC 47.5 (2017).
Dunbar, C. E. et al. Gene therapy comes of age. Science 359, eaan4672 (2018). This comprehensive Review summarizes the history of gene therapy over almost 30 years, noting key milestones, including the advances that led to the haemophilia therapies discussed in this Review.
Donsante, A. et al. AAV vector integration sites in mouse hepatocellular carcinoma. Science 317, 477 (2007).
Chandler, R. J. et al. Vector design influences hepatic genotoxicity after adeno-associated virus gene therapy. J. Clin. Invest. 125, 870–880 (2015).
Vandamme, C., Adjali, O. & Mingozzi, F. Unraveling the complex story of immune responses to AAV vectors trial after trial. Hum. Gene Ther. 28, 1061–1074 (2017).
George, L. A. et al. Hemophilia B gene therapy with a high-specific-activity factor IX variant. N. Engl. J. Med. 377, 2215–2227 (2017).
Simioni, P. et al. X-Linked thrombophilia with a mutant factor IX (factor IX Padua). N. Engl. J. Med. 361, 1671–1675 (2009).
Miesbach, W. et al. Updated results from a dose-escalation study in adults with severe or moderate-severe hemophilia B treated with AMT-060 (AAV5-hFIX) gene therapy: up to 1.5 years follow-up [abstract]. Res. Pract. Thromb. Haemost. 1 (Suppl. 1), OC 13.2 (2017).
Leebeek, F. et al. Stable elevations in FIX activity and reductions in annualized bleeding rate over up to 2 years of follow-up of adults with severe or moderate-severe hemophilia B treated with AMT-060 (AAV5-hFIX) gene therapy. Blood 130, 602 (2017).
Rangarajan, S. et al. AAV5-factor VIII gene transfer in severe hemophilia A. N. Engl. J. Med. 377, 2519–2530 (2017).
Pasi, K. J. et al. Achievement of normal circulating factor VIII activity following Bmn 270 AAV5-FVIII gene transfer: interim, long-term efficacy and safety results from a phase 1/2 study in patients with severe hemophilia A. Blood 130, 603 (2017).
George, L. A. et al. Spk-8011: preliminary results from a phase 1/2 dose escalation trial of an investigational AAV-mediated gene therapy for hemophilia A. Blood 130, 604 (2017).
Shi, Q. et al. Platelet gene therapy corrects the hemophilic phenotype in immunocompromised hemophilia A mice transplanted with genetically manipulated human cord blood stem cells. Blood 123, 395–403 (2014).
Liu, T. et al. Intravenously administered FVIII encoding lenti-viral vector mediate up to 5000% of normal and persisting FVIII expression in HemA neonatal mice [abstract]. Mol. Ther. 25(5S1), 351 (2017).
Cantore, A. et al. Liver-directed lentiviral gene therapy in a dog model of hemophilia B. Sci. Transl Med. 7, 277ra28 (2015).
Den Uijl, I. E. et al. Clinical severity of haemophilia A: does the classification of the 1950s still stand? Haemophilia 17, 849–853 (2011).
Nathwani, A. C. et al. Adenovirus-associated virus vector-mediated gene transfer in hemophilia B. N. Engl. J. Med. 365, 2357–2365 (2011). This seminal paper describes the first successful AAV gene therapy trial in haemophilia, identifies AAV-capsid-specific T cells as a critical issue and demonstrates the ability of short courses of glucocorticoid therapy to suppress this immune response without loss of transgene expression; this strategy has been implemented in some form in every AAV clinical trial since this paper.
Strohl, W. R. Fusion proteins for half-life extension of biologics as a strategy to make biobetters. BioDrugs 29, 215–239 (2015).
Turecek, P. L., Bossard, M. J., Schoetens, F. & Ivens, I. A. PEGylation of biopharmaceuticals: a review of chemistry and nonclinical safety information of approved drugs. J. Pharm. Sci. 105, 460–475 (2016).
DiMasi, J. A., Grabowski, H. G. & Hansen, R. W. Innovation in the pharmaceutical industry: new estimates of R&D costs. J. Health Econ. 47, 20–33 (2016).
Collins, P. et al. First-line immune tolerance induction for children with severe haemophilia A: a protocol from the UK Haemophilia Centre Doctors' Organisation Inhibitor and Paediatric Working Parties. Haemophilia 23, 654–659 (2017).
Krishnamoorthy, S. et al. Recombinant factor VIII Fc (rFVIIIFc) fusion protein reduces immunogenicity and induces tolerance in hemophilia A mice. Cell. Immunol. 301, 30–39 (2016).
Carcao, M. et al. Recombinant factor VIII Fc fusion protein for immune tolerance induction in patients with severe haemophilia A with inhibitors-A retrospective analysis. Haemophilia 24, 245–252 (2018).
Rath, T. et al. Fc-fusion proteins and FcRn: structural insights for longer-lasting and more effective therapeutics. Crit. Rev. Biotechnol. 35, 235–254 (2015).
van der Flier, A. et al. FcRn rescues recombinant factor VIII Fc fusion protein from a VWF independent FVIII clearance pathway in mouse hepatocytes. PLoS ONE 10, e0124930 (2015).
Rasmussen, C. E. et al. Evaluation of nonacog beta pegol long-term safety in the immune-deficient Rowett nude rat (Crl:NIH-Foxn1rnu). Toxicol. Pathol. 44, 726–737 (2016).
Rasmussen, C. E. et al. Long-term safety of PEGylated coagulation factor VIII in the immune-deficient Rowett nude rat. J. Toxicol. 2017, 8496246 (2017).
Viuff, D., Barrowcliffe, T., Saugstrup, T., Ezban, M. & Lillicrap, D. International comparative field study of N8 evaluating factor VIII assay performance. Haemophilia 17, 695–702 (2011).
Sommer, J. M. et al. Comparative field study: impact of laboratory assay variability on the assessment of recombinant factor IX Fc fusion protein (rFIXFc) activity. Thromb. Haemost. 112, 932–940 (2014).
Sommer, J. M. et al. Comparative field study evaluating the activity of recombinant factor VIII Fc fusion protein in plasma samples at clinical haemostasis laboratories. Haemophilia 20, 294–300 (2014).
Bowyer, A. E., Hillarp, A., Ezban, M., Persson, P. & Kitchen, S. Measuring factor IX activity of nonacog beta pegol with commercially available one-stage clotting and chromogenic assay kits: a two-center study. J. Thromb. Haemost. 14, 1428–1435 (2016).
Tarantino, M. D. et al. Clinical evaluation of an advanced category antihaemophilic factor prepared using a plasma/albumin-free method: pharmacokinetics, efficacy, and safety in previously treated patients with haemophilia A. Haemophilia 10, 428–437 (2004).
Saxena, K. et al. Efficacy and safety of BAY 81–8973, a full-length recombinant factor VIII: results from the LEOPOLD I trial. Haemophilia 22, 706–712 (2016).
Recht, M. et al. Clinical evaluation of moroctocog alfa (AF-CC), a new generation of B-domain deleted recombinant factor VIII (BDDrFVIII) for treatment of haemophilia A: demonstration of safety, efficacy, and pharmacokinetic equivalence to full-length recombinant factor VIII. Haemophilia 15, 869–880 (2009).
Martinowitz, U. et al. Bioequivalence between two serum-free recombinant factor VIII preparations (N8 and ADVATE(R))—an open-label, sequential dosing pharmacokinetic study in patients with severe haemophilia A. Haemophilia 17, 854–859 (2011).
Sandberg, H. et al. Functional characteristics of the novel, human-derived recombinant FVIII protein product, human-cl rhFVIII. Thromb. Res. 130, 808–817 (2012).
Lissitchkov, T. et al. Pharmacokinetics and efficacy of on-demand treatment with human-cl rhFVIII in previously treated patients with severe haemophilia A [abstract]. Haemophilia 18 (Suppl. 3), PO-TU-170 (2012).
Klamroth, R. et al. Comparative pharmacokinetics of rVIII-SingleChain and octocog alfa (Advate((R))) in patients with severe haemophilia A. Haemophilia 22, 730–738 (2016).
Paik, S. H. et al. Comparability studies of new 3rd generation recombinant human factor VIII GreenGene F after improvement of formulation and viral inactivation/removal process. Biologicals 40, 405–414 (2012).
White, G. et al. Clinical evaluation of recombinant factor IX. Semin. Hematol. 35, 33–38 (1998).
Windyga, J. et al. Pharmacokinetics, efficacy and safety of BAX326, a novel recombinant factor IX: a prospective, controlled, multicentre phase I/III study in previously treated patients with severe (FIX level <1%) or moderately severe (FIX level < = 2%) haemophilia B. Haemophilia 20, 15–24 (2014).
Martinowitz, U. et al. Pharmacokinetic properties of IB1001, an investigational recombinant factor IX, in patients with haemophilia B: repeat pharmacokinetic evaluation and sialylation analysis. Haemophilia 18, 881–887 (2012).
Powell, J. S. et al. Phase 3 study of recombinant factor IX Fc fusion protein in hemophilia B. N. Engl. J. Med. 369, 2313–2323 (2013).
Santagostino, E. et al. Safety and pharmacokinetics of a novel recombinant fusion protein linking coagulation factor IX with albumin (rIX-FP) in hemophilia B patients. Blood 120, 2405–2411 (2012).
Negrier, C., Knobe, K., Tiede, A., Giangrande, P. & Moss, J. Enhanced pharmacokinetic properties of a glycoPEGylated recombinant factor IX: a first human dose trial in patients with hemophilia B. Blood 118, 2695–2701 (2011).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
T.H. and R.P. are employees of Bioverativ, a Sanofi company.
Related links
RELATED LINKS
Glossary
- Haemostasis
-
Literally means halting of the blood. In this context, haemostasis is the means by which the body maintains the balance between normal blood flow and the clotting process to stop bleeding events.
- Thrombosis
-
The pathological formation of a blood clot in a blood vessel.
- Haemophilic arthropathy
-
Permanent joint disease occurring in patients with haemophilia owing to long-term bleeding into the joint (haemarthrosis) and resulting inflammation.
- Cryoprecipitates
-
Insoluble precipitates prepared by slowly thawing fresh-frozen plasma; cryoprecipitates are used as a source of factor VIII but also contain considerable amounts of fibrinogen, von Willebrand factor, FXIII and fibronectin.
- B domain
-
A large protein-coding domain in the middle of the F8 gene that is largely dispensable for the full activity of the protein.
- PEGylated
-
PEGylation is the chemical or enzymatic addition of polyethylene glycol (PEG) molecules to proteins. PEG molecules are synthetic, highly water-soluble polymers with a large hydrodynamic radius and are not degraded by endogenous enzymes. PEG molecules used for half-life extension typically range from 5 to 40 kDa in size.
Rights and permissions
About this article
Cite this article
Peters, R., Harris, T. Advances and innovations in haemophilia treatment. Nat Rev Drug Discov 17, 493–508 (2018). https://doi.org/10.1038/nrd.2018.70
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2018.70
This article is cited by
-
Vagus nerve stimulation primes platelets and reduces bleeding in hemophilia A male mice
Nature Communications (2023)
-
Liver-directed lentiviral gene therapy corrects hemophilia A mice and achieves normal-range factor VIII activity in non-human primates
Nature Communications (2022)
-
Prediction of hemophilia A severity using a small-input machine-learning framework
npj Systems Biology and Applications (2021)
-
Coagulation factor IX analysis in bioreactor cell culture supernatant predicts quality of the purified product
Communications Biology (2021)
-
Recent advances in lentiviral vectors for gene therapy
Science China Life Sciences (2021)