Key Points
-
Targeted treatments are lacking for most autoimmune diseases. Many patients experience adverse effects with current therapies because they destroy or inactivate healthy cells along with pathological cells.
-
Newly targeted therapies aimed exclusively at the rare autoreactive T lymphocytes (T cells) causing autoimmune diseases have the greatest potential to avoid adverse effects, as well as to prevent, treat or reverse certain autoimmune diseases.
-
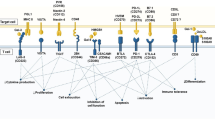
Tumour necrosis factor (TNF) is a master signalling cytokine used by the immune system and other bodily systems. Autoreactive T cells are extremely vulnerable to death from exogenous TNF. However, because TNF receptors are ubiquitous, TNF as a treatment is too toxic.
-
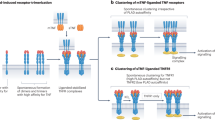
There are two TNF receptors. TNF receptor 1 (TNFR1) is found on almost all cells of the body but TNF receptor 2 (TNFR2) is more restrictively expressed. T cells, and certain other cell types, express TNFR2.
-
Research has demonstrated that agonists targeted at TNFR2 are successful at killing autoreactive T cells in an animal model of type 1 diabetes. The effect is dose-dependent.
-
This Review harnesses the rationale for drug discovery of exogenous agonists for TNFR2 as a less toxic approach to treatment, and it offers short-term and long-term strategies for their development.
Abstract
Although drug development has advanced for autoimmune diseases, many current therapies are hampered by adverse effects and the frequent destruction or inactivation of healthy cells in addition to pathological cells. Targeted autoimmune therapies capable of eradicating the rare autoreactive immune cells that are responsible for the attack on the body's own cells are yet to be identified. This Review presents a new emerging approach aimed at selectively destroying autoreactive immune cells by specific activation of tumour necrosis factor receptor 2 (TNFR2), which is found on autoreactive and normal T lymphocytes, with the potential of avoiding or reducing the toxicity observed with existing therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Croft, M. The role of TNF superfamily members in T-cell function and diseases. Nature Rev. Immunol. 9, 271–285 (2009).
Breedveld, F. C. et al. The PREMIER study: a multicenter, randomized, double-blind clinical trial of combination therapy with adalimumab plus methotrexate versus methotrexate alone or adalimumab alone in patients with early, aggressive rheumatoid arthritis who had not had previous methotrexate treatment. Arthritis Rheum. 54, 26–37 (2006).
Klareskog, L. et al. Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: double-blind randomised controlled trial. Lancet 363, 675–681 (2004).
Chaudhari, U. et al. Efficacy and safety of infliximab monotherapy for plaque-type psoriasis: a randomised trial. Lancet 357, 1842–1847 (2001).
Dereure, O. et al. Psoriatic lesions induced by antitumour necrosis factor-alpha treatment: two cases. Br. J. Dermatol. 151, 506–507 (2004).
Flendrie, M. et al. Dermatological conditions during TNF-alpha-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res. Ther. 7, R666–R676 (2005).
Kary, S. et al. New onset or exacerbation of psoriatic skin lesions in patients with definite rheumatoid arthritis receiving tumour necrosis factor alpha antagonists. Ann. Rheum. Dis. 65, 405–407 (2006).
Thurber, M., Feasel, A., Stroehlein, J. & Hymes, S. R. Pustular psoriasis induced by infliximab. J. Drugs Dermatol. 3, 439–440 (2004).
Verea, M. M., Del Pozo, J., Yebra-Pimentel, M. T., Porta, A. & Fonseca, E. Psoriasiform eruption induced by infliximab. Ann. Pharmacother. 38, 54–57 (2004).
Galaria, N. A., Werth, V. P. & Schumacher, H. R. Leukocytoclastic vasculitis due to etanercept. J. Rheumatol. 27, 2041–2044 (2000).
Jarrett, S. J. et al. Anti-tumor necrosis factor-alpha therapy-induced vasculitis: case series. J. Rheumatol. 30, 2287–2291 (2003).
Charles, P. J., Smeenk, R. J., De Jong, J., Feldmann, M. & Maini, R. N. Assessment of antibodies to double-stranded DNA induced in rheumatoid arthritis patients following treatment with infliximab, a monoclonal antibody to tumor necrosis factor alpha: findings in open-label and randomized placebo-controlled trials. Arthritis Rheum. 43, 2383–2390 (2000).
Feldmann, M., Brennan, F. M. & Maini, R. N. Role of cytokines in rheumatoid arthritis. Annu. Rev. Immunol. 14, 397–440 (1996).
US Food and Drug Administration. Update on the TNF-alpha blocking agents. US FDA website [online], (2006).
Klinkhoff, A. Biological agents for rheumatoid arthritis: targeting both physical function and structural damage. Drugs 64, 1267–1283 (2004).
Stokes, M. B. et al. Development of glomerulonephritis during anti-TNF-alpha therapy for rheumatoid arthritis. Nephrol. Dial. Transplant. 20, 1400–1406 (2005).
Lipsky, P. E. et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-tumor necrosis factor trial in rheumatoid arthritis with concomitant therapy study group. N. Engl. J. Med. 343, 1594–1602 (2000).
Moreland, L. W. et al. Etanercept therapy in rheumatoid arthritis. A randomized, controlled trial. Ann. Intern. Med. 130, 478–486 (1999).
Vermeire, S. et al. Autoimmunity associated with anti-tumor necrosis factor alpha treatment in Crohn's disease: a prospective cohort study. Gastroenterology 125, 32–39 (2003).
Tack, C. J., Kleijwegt, F. S., Van Riel, P. L. & Roep, B. O. Development of type 1 diabetes in a patient treated with anti-TNF-alpha therapy for active rheumatoid arthritis. Diabetologia 52, 1442–1444 (2009).
Dorman, J. S. et al. The Pittsburgh insulin-dependent diabetes mellitus (IDDM) morbidity and mortality study. Mortality results. Diabetes 33, 271–276 (1984).
Mukai, Y. et al. Crystallization and preliminary X-ray analysis of the tumour necrosis factor alpha-tumour necrosis factor receptor type 2 complex. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 65, 295–298 (2009). This article is the first to use crystallography to find the structure of TNFR1.
Mukai, Y. et al. Structure–function relationship of tumor necrosis factor (TNF) and its receptor interaction based on 3D structural analysis of a fully active TNFR1-selective TNF mutant. J. Mol. Biol. 385, 1221–1229 (2009). This article is the first to use phage display techniques to produce receptor-specific TNF mutants to activate only one type of TNFR.
Aggarwal, B. B. Signalling pathways of the TNF superfamily: a double-edged sword. Nature Rev. Immunol. 3, 745–756 (2003).
Pimentel-Muinos, F. X. & Seed, B. Regulated commitment of TNF receptor signaling: a molecular switch for death or activation. Immunity 11, 783–793 (1999).
Kodama, S., Davis, M. & Faustman, D. L. The therapeutic potential of tumor necrosis factor for autoimmune disease: a mechanistically based hypothesis. Cell. Mol. Life Sci. 62, 1850–1862 (2005). This review article explains the evidence behind the counter-intuitive rationale to investigate TNF for its potential to selectively and effectively kill autoreactive T cells in several autoimmune disorders.
Abbott, D. W., Wilkins, A., Asara, J. M. & Cantley, L. C. The Crohn's disease protein, NOD2, requires RIP2 in order to induce ubiquitinylation of a novel site on NEMO. Curr. Biol. 14, 2217–2227 (2004).
Eckmann, L. & Karin, M. NOD2 and Crohn's disease: loss or gain of function? Immunity 22, 661–667 (2005).
Maeda, S. et al. Nod2 mutation in Crohn's disease potentiates NF-kappaB activity and IL-1beta processing. Science 307, 734–738 (2005).
Watanabe, T., Kitani, A., Murray, P. J. & Strober, W. NOD2 is a negative regulator of Toll-like receptor 2-mediated T helper type 1 responses. Nature Immunol. 5, 800–808 (2004).
Levine, A. et al. TNF promoter polymorphisms and modulation of growth retardation and disease severity in pediatric Crohn's disease. Am. J. Gastroenterol. 100, 1598–1604 (2005).
van Heel, D. A. et al. Inflammatory bowel disease is associated with a TNF polymorphism that affects an interaction between the OCT1 and NF-κB transcription factors. Hum. Mol. Genet. 11, 1281–1289 (2002).
Karban, A. S. et al. Functional annotation of a novel NFKB1 promoter polymorphism that increases risk for ulcerative colitis. Hum. Mol. Genet. 13, 35–45 (2004).
Kammer, G. M. & Tsokos, G. C. Abnormal T lymphocyte signal transduction in systemic lupus erythematosus. Curr. Dir. Autoimmun. 5, 131–150 (2002).
Hegazy, D. M. et al. NF-κB polymorphisms and susceptibility to type 1 diabetes. Genes Immun. 2, 304–308 (2001).
Deng, G. Y., Muir, A., Maclaren, N. K. & She, J. X. Association of LMP2 and LMP7 genes within the major histocompatibility complex with insulin-dependent diabetes mellitus: population and family studies. Am. J. Hum. Genet. 56, 528–534 (1995).
Ding, H., Cheng, H., Fu, Z., Yan, L. & Yang, G. Relationship of large multifunctional proteasome 7 gene polymorphism with susceptibility to type 1 diabetes mellitus and DR3 gene. Chin. Med. J. (Engl.) 114, 1263–1266 (2001).
Fu, Y., Yan, G., Shi, L. & Faustman, D. Antigen processing and autoimmunity. Evaluation of mRNA abundance and function of HLA-linked genes. Ann. NY Acad. Sci. 842, 138–155 (1998).
Hayashi, T. & Faustman, D. Essential role of HLA-encoded proteasome subunits in NF-κB activation and prevention of TNF-α induced apoptosis. J. Biol. Chem. 275, 5238–5247 (2000).
Hayashi, T. & Faustman, D. NOD mice are defective in proteasome production and activation of NF-κB. Mol. Cell Biol. 19, 8646–8659 (1999). This article was the first to demonstrate proteasomal defects prevent cleavage of NF-κB from its chaperone protein, IκBα, in a mouse model of type 1 diabetes.
Hayashi, T. & Faustman, D. L. Selected contribution: association of gender-related LMP2 inactivation with autoimmune pathogenesis. J. Appl. Physiol. 91, 2804–2815 (2001).
Yan, G., Fu, Y. & Faustman, D. L. Reduced expression of Ta p 1 and Lmp2 antigen processing genes in the nonobese diabetic (NOD) mouse due to a mutation in their shared bidirectional promoter. J. Immunol. 159, 3068–3080 (1997). This article was the first to demonstrate the LMP2 subunit of the proteasome was lacking in subpopulations of the immune system, thus influencing both MHC class I education and interrupting proper NF-κB activation in CD8 T cells, in a mouse model of autoimmunity.
Miterski, B. et al. Inhibitors in the NF-κB cascade comprise prime candidate genes predisposing to multiple sclerosis, especially in selected combinations. Genes Immun. 3, 211–219 (2002).
Chatzikyriakidou, A., Georgiou, I., Voulgari, P. V. & Drosos, A. A. The role of tumor necrosis factor (TNF)-alpha and TNF receptor polymorphisms in susceptibility to ankylosing spondylitis. Clin. Exp. Rheumatol. 27, 645–648 (2009).
Faustman, D. et al. Linkage of faulty major histocompatibility complex class I to autoimmune diabetes. Science 254, 1756–1761 (1991).
Faustman, D. L. & Davis, M. The primacy of CD8 T lymphocytes in type 1 diabetes and implications for therapies. J. Mol. Med. 87, 1173–1178 (2009).
Wong, F. S. et al. Identification of an MHC class I-restricted autoantigen in type 1 diabetes by screening an organ-specific cDNA library. Nature Med. 5, 1026–1031 (1999).
Christianson, S. W., Shultz, L. D. & Leiter, E. H. Adoptive transfer of diabetes into immunodeficient NOD-scid/scid mice. Relative contributions of CD4+ and CD8+ T-cells from diabetic versus prediabetic NOD.NON-Thy-1a donors. Diabetes 42, 44–55 (1993).
Yagi, H. et al. Analysis of the roles of CD4+ and CD8+ T cells in autoimmune diabetes of NOD mice using transfer to NOD athymic nude mice. Eur. J. Immunol. 22, 2387–2393 (1992).
Pinkse, G. G. et al. Autoreactive CD8 T cells associated with beta cell destruction in type 1 diabetes. Proc. Natl Acad. Sci. USA 102, 18425–18430 (2005).
Satoh, J., Seino, H. & Abo, T. Recombinant human tumor necrosis factor α suppresses autoimmune diabetes in nonobese diabetic mice. J. Clin. Invest. 84, 1345–1348 (1989).
Grewal, I. S. et al. Local expression of transgene encoded TNF alpha in islets prevents autoimmune diabetes in non-obese diabetic (NOD) mice by preventing the development of autoreactive islet specific T cells. J. Exp. Med. 184, 1963–1974 (1996).
Sadelain, M. W. et al. Prevention of diabetes in the BB rat by early immunotherapy using Freund's adjuvant. J. Autoimmun. 3, 671–680 (1990).
Ryu, S., Kodama, S., Ryu, K., Schoenfeld, D. A. & Faustman, D. L. Reversal of established autoimmune diabetes by restoration of endogenous beta cell function. J. Clin. Invest. 108, 63–72 (2001).
Nishio, J. et al. Islet recovery and reversal of murine type 1 diabetes in the absence of any infused spleen cell contribution. Science 311, 1775–1778 (2006).
Suri, A. et al. Immunological reversal of autoimmune diabetes without hematopoietic replacement of beta cells. Science 311, 1778–1780 (2006).
Chong, A. S. et al. Reversal of diabetes in non-obese diabetic mice without spleen cell-derived beta cell regeneration. Science 311, 1774–1775 (2006).
Kodama, S., Kuhtreiber, W., Fujimura, S., Dale, E. A. & Faustman, D. L. Islet regeneration during the reversal of autoimmune diabetes in NOD mice. Science 302, 1223–1227 (2003). This article was the first to show that treatment with a TNF inducer was capable of stimulating pancreatic islet cells to be restored and function normally by returning glycaemic levels back to healthy levels in a mouse model of advanced type 1 diabetes.
Sidhu, R. S. & Bollon, A. P. Tumor necrosis factor activities and cancer therapy — a perspective. Pharmacol. Ther. 57, 79–128 (1993).
Hieber, U. & Heim, M. E. Tumor necrosis factor for the treatment of malignancies. Oncology 51, 142–153 (1994).
Balkwill, F. Tumour necrosis factor and cancer. Nature Rev. Cancer 9, 361–371 (2009).
Qin, H. Y., Chaturvedi, P. & Singh, B. In vivo apoptosis of diabetogenic T cells in NOD mice by IFN-γ/TNF-α. Int. Immunol. 16, 1723–1732 (2004).
Christen, U. et al. A dual role for TNF-alpha in type 1 diabetes: islet-specific expression abrogates the ongoing autoimmune process when induced late but not early during pathogenesis. J. Immunol. 166, 7023–7032 (2001).
Tran, S. D. et al. Reversal of Sjogren's-like syndrome in non-obese diabetic mice. Ann. Rheum. Dis. 66, 812–814 (2007).
Ban, L. et al. Selective death of autoreactive T cells in human diabetes by TNF or TNF receptor 2 agonism. Proc. Natl Acad. Sci. USA 105, 13644–13649 (2008). This article was the first to demonstrate that a specific TNFR2 ligand was capable of selectively killing insulin-specific autoreactive T cells in type 1 diabetes.
Zuniga-Pflucker, J. C., Di, J. & Lenardo, M. J. Requirement for TNF-alpha and IL-1 alpha in fetal thymocyte commitment and differentiation. Science 268, 1906–1909 (1995).
Samira, S. et al. Tumor necrosis factor promotes human T-cell development in nonobese diabetic/severe combined immunodeficient mice. Stem Cells 22, 1085–1100 (2004).
Lenardo, M. J. Interleukin-2 programs mouse alpha beta T lymphocytes for apoptosis. Nature 353, 858–861 (1991).
Ware, C. F. et al. Tumor necrosis factor (TNF) receptor expression in T lymphocytes. Differential regulation of the type I TNF receptor during activation of resting and effector T cells. J. Immunol. 147, 4229–4238 (1991).
Yang, L., Lindholm, K., Konishi, Y., Li, R. & Shen, Y. Target depletion of distinct tumor necrosis factor receptor subtypes reveals hippocampal neuron death and survival through different signal transduction pathways. J. Neurosci. 22, 3025–3032 (2002).
McCoy, M. K. & Tansey, M. G. TNF signaling inhibition in the CNS: implications for normal brain function and neurodegenerative disease. J. Neuroinflammation 5, 45 (2008).
Arnett, H. A. et al. TNF alpha promotes proliferation of oligodendrocyte progenitors and remyelination. Nature Neurosci. 4, 1116–1122 (2001).
Dopp, J. M. et al. Expression of the p75 TNF receptor is linked to TNF-induced NF-κB translocation and oxyradical neutralization in glial cells. Neurochem. Res. 27, 1535–1542 (2002).
Irwin, M. W. et al. Tissue expression and immunolocalization of tumor necrosis factor-alpha in postinfarction dysfunctional myocardium. Circulation 99, 1492–1498 (1999).
Tartaglia, L. A. et al. The two different receptors for tumor necrosis factor mediate distinct cellular responses. Proc. Natl Acad. Sci. USA 88, 9292–9296 (1991).
Grell, M., Becke, F. M., Wajant, H., Mannel, D. N. & Scheurich, P. TNF receptor type 2 mediates thymocyte proliferation independently of TNF receptor type 1. Eur. J. Immunol. 28, 257–263 (1998).
Bocker, W. et al. IKK-2 is required for TNF-α-induced invasion and proliferation of human mesenchymal stem cells. J. Mol. Med. 86, 1183–1192 (2008).
Chen, L. B., Jiang, X. B. & Yang, L. Differentiation of rat marrow mesenchymal stem cells into pancreatic islet beta-cells. World J. Gastroenterol. 10, 3016–3020 (2004).
Carpentier, I., Coornaert, B. & Beyaert, R. Function and regulation of tumor necrosis factor type 2. Curr. Med. Chem. 11, 2205–2212 (2004). This review article provides the most comprehensive portrait and analysis of TNFR2.
Tartaglia, L. A., Ayres, T. M., Wong, G. H. & Goeddel, D. V. A novel domain within the 55 kd TNF receptor signals cell death. Cell 74, 845–853 (1993).
Hsu, H., Xiong, J. & Goeddel, D. V. The TNF receptor 1-associated protein TRADD signals cell death and NF-κB activation. Cell 81, 495–504 (1995).
Wilson, N. S., Dixit, V. & Ashkenazi, A. Death receptor signal transducers: nodes of coordination in immune signaling networks. Nature Immunol. 10, 348–355 (2009).
Rothe, M., Sarma, V., Dixit, V. M. & Goeddel, D. V. TRAF2-mediated activation of NF-kappaB by TNF receptor 2 and CD40. Science 269, 1424–1427 (1995).
Rothe, M., Wong, S. C., Henzel, W. J. & Goeddel, D. V. A novel family of putative signal transducers associated with the cytoplasmic domain of the 75 kDa tumor necrosis factor receptor. Cell 78, 681–692 (1994).
Gehr, G., Gentz, R., Brockhaus, M., Loetscher, H. & Lesslauer, W. Both tumor necrosis factor receptor types mediate proliferative signals in human mononuclear cell activation. J. Immunol. 149, 911–917 (1992).
Fontaine, V. et al. Neurodegenerative and neuroprotective effects of tumor necrosis factor (TNF) in retinal ischemia: opposite roles of TNF receptor 1 and TNF receptor 2. J. Neurosci. 22, RC216 (2002).
Shen, Y., Li, R. & Shiosaki, K. Inhibition of p75 tumor necrosis factor receptor by antisense oligonucleotides increases hypoxic injury and beta-amyloid toxicity in human neuronal cell line. J. Biol. Chem. 272, 3550–3553 (1997).
Shen, H. M. & Pervaiz, S. TNF receptor superfamily-induced cell death: redox-dependent execution. FASEB J. 20, 1589–1598 (2006).
Milani, D. et al. Tumour necrosis factor-related apoptosis-inducing ligand sequentially activates pro-survival and pro-apoptotic pathways in SK-N-MC neuronal cells. J. Neurochem. 86, 126–135 (2003).
Hoffmann, A. & Baltimore, D. Circuitry of nuclear factor kappaB signaling. Immunol. Rev. 210, 171–186 (2006).
Fotin-Mleczek, M. et al. Apoptotic crosstalk of TNF receptors: TNF-R2-induces depletion of TRAF2 and IAP proteins and accelerates TNF-R1-dependent activation of caspase-8. J. Cell Sci. 115, 2757–2770 (2002). This article was one of the first to demonstrate in vitro crosstalk between activation of either TNFR1 and TNFR2, and the mechanisms behind crosstalk.
Dembic, Z. et al. Two human TNF receptors have similar extracellular, but distinct intracellular, domain sequences. Cytokine 2, 231–237 (1990).
Meylan, E. & Tschopp, J. The RIP kinases: crucial integrators of cellular stress. Trends Biochem. Sci. 30, 151–159 (2005).
Vercammen, D. et al. Cytotoxicity in L929 murine fibrosarcoma cells after triggering of transfected human p75 tumour necrosis factor (TNF) receptor is mediated by endogenous murine TNF. Cytokine 7, 463–470 (1995).
Zheng, L. et al. Induction of apoptosis in mature T cells by tumour necrosis factor. Nature 377, 348–351 (1995). This article was the first to find that stimulation of TNFR2 induces cell death in mature T cells of mice.
Grell, M. et al. The transmembrane form of tumor necrosis factor is the prime activating ligand of the 80 kDa tumor necrosis factor receptor. Cell 83, 793–802 (1995).
Linseman, D. A. et al. Suppression of death receptor signaling in cerebellar Purkinje neurons protects neighboring granule neurons from apoptosis via an insulin-like growth factor I-dependent mechanism. J. Biol. Chem. 277, 24546–24553 (2002).
Krippner-Heidenreich, A. et al. Control of receptor-induced signaling complex formation by the kinetics of ligand/receptor interaction. J. Biol. Chem. 277, 44155–44163 (2002).
Weiss, T. et al. TNFR80-dependent enhancement of TNFR60-induced cell death is mediated by TNFR-associated factor 2 and is specific for TNFR60. J. Immunol. 161, 3136–3142 (1998).
Weiss, T. et al. Enhancement of TNF receptor p60-mediated cytotoxicity by TNF receptor p80: requirement of the TNF receptor-associated factor-2 binding site. J. Immunol. 158, 2398–2404 (1997).
Li, X., Yang, Y. & Ashwell, J. D. TNF-RII and c-IAP1 mediate ubiquitination and degradation of TRAF2. Nature 416, 345–347 (2002).
Grech, A. P. et al. Tumor necrosis factor receptor 2 (TNFR2) signaling is negatively regulated by a novel, carboxyl-terminal TNFR-associated factor 2 (TRAF2)-binding site. J. Biol. Chem. 280, 31572–31581 (2005).
Micheau, O. & Tschopp, J. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114, 181–190 (2003).
Ihnatko, R. & Kubes, M. TNF signaling: early events and phosphorylation. Gen. Physiol. Biophys. 26, 159–167 (2007).
Wang, M., Crisostomo, P. R., Markel, T. A., Wang, Y. & Meldrum, D. R. Mechanisms of sex differences in TNFR2-mediated cardioprotection. Circulation 118, S38–S45 (2008).
Rodriguez, M. et al. Tumor necrosis factor alpha is reparative via TNFR1 in the hippocampus and via TNFR2 in the striatum after virus-induced encephalitis. Brain Pathol. 19, 12–26 (2009).
Marchetti, L., Klein, M., Schlett, K., Pfizenmaier, K. & Eisel, U. L. Tumor necrosis factor (TNF)-mediated neuroprotection against glutamate-induced excitotoxicity is enhanced by N-methyl-D-aspartate receptor activation. Essential role of a TNF receptor 2-mediated phosphatidylinositol 3-kinase-dependent NF-κB pathway. J. Biol. Chem. 279, 32869–32881 (2004).
Monden, Y. et al. Tumor necrosis factor-alpha is toxic via receptor 1 and protective via receptor 2 in a murine model of myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 293, H743–H753 (2007).
Barton, A., John, S., Ollier, W. E., Silman, A. & Worthington, J. Association between rheumatoid arthritis and polymorphism of tumor necrosis factor receptor II, but not tumor necrosis factor receptor I, in Caucasians. Arthritis Rheum. 44, 61–65 (2001).
Dieude, P. et al. Association between tumor necrosis factor receptor II and familial, but not sporadic, rheumatoid arthritis: evidence for genetic heterogeneity. Arthritis Rheum. 46, 2039–2044 (2002).
Orozco, G. et al. Study of functional variants of the BANK1 gene in rheumatoid arthritis. Arthritis Rheum. 60, 372–379 (2009).
Sashio, H. et al. Polymorphisms of the TNF gene and the TNF receptor superfamily member 1B gene are associated with susceptibility to ulcerative colitis and Crohn's disease, respectively. Immunogenetics 53, 1020–1027 (2002).
Komata, T., Tsuchiya, N., Matsushita, M., Hagiwara, K. & Tokunaga, K. Association of tumor necrosis factor receptor 2 (TNFR2) polymorphism with susceptibility to systemic lupus erythematosus. Tissue Antigens 53, 527–533 (1999).
Pierik, M. et al. Tumour necrosis factor-α receptor 1 and 2 polymorphisms in inflammatory bowel disease and their association with response to infliximab. Aliment. Pharmacol. Ther. 20, 303–310 (2004).
Ishikawa, Y. et al. Polymorphisms in TNFA and TNFR2 affect outcome of unrelated bone marrow transplantation. Bone Marrow Transplant. 29, 569–575 (2002).
Till, A. et al. The Met-196→ Arg variation of human tumor necrosis factor receptor 2 (TNFR2) affects TNF-α-induced apoptosis by impaired NFκB signaling and target gene expression. J. Biol. Chem. 280, 5994–6004 (2005).
Plenge, R. M. et al. TRAF1-C5 as a risk locus for rheumatoid arthritis — a genomewide study. N. Engl. J. Med. 357, 1199–1209 (2007).
Diez-Ruiz, A. et al. Soluble receptors for tumour necrosis factor in clinical laboratory diagnosis. Eur. J. Haematol. 54, 1–8 (1995).
Lantz, M., Malik, S., Slevin, M. L. & Olsson, I. Infusion of tumor necrosis factor (TNF) causes an increase in circulating TNF-binding protein in humans. Cytokine 2, 402–406 (1990).
Jansen, J. et al. Inhibition of the release of soluble tumor necrosis factor receptors in experimental endotoxemia by an anti-tumor necrosis factor-alpha antibody. J. Clin. Immunol. 15, 45–50 (1995).
Cope, A. P. et al. Increased levels of soluble tumor necrosis factor receptors in the sera and synovial fluid of patients with rheumatic diseases. Arthritis Rheum. 35, 1160–1169 (1992).
Gabay, C. et al. Circulating levels of tumor necrosis factor soluble receptors in systemic lupus erythematosus are significantly higher than in other rheumatic diseases and correlate with disease activity. J. Rheumatol 24, 303–308 (1997).
Holtmann, M. H. et al. Tumor necrosis factor-receptor 2 is up-regulated on lamina propria T cells in Crohn's disease and promotes experimental colitis in vivo. Eur. J. Immunol. 32, 3142–3151 (2002).
Xanthoulea, S. et al. Tumor necrosis factor (TNF) receptor shedding controls thresholds of innate immune activation that balance opposing TNF functions in infectious and inflammatory diseases. J. Exp. Med. 200, 367–376 (2004).
Engelmann, H. et al. Antibodies to a soluble form of a tumor necrosis factor (TNF) receptor have TNF-like activity. J. Biol. Chem. 265, 14497–14504 (1990).
Li, F., Guo, J., Fu, Y., Yan, G. & Faustman, D. Abnormal class I assembly and peptide presentation in the diabetic NOD mouse. Proc. Natl Acad. Sci. USA 91, 11128–11132 (1994).
Krause, S. et al. Immunoproteasome subunit LMP2 expression is deregulated in Sjogren's syndrome but not in other autoimmune disorders. Ann. Rheum. Dis. 65, 1021–1027 (2006). This article was the first to show in the human autoimmune disease Sjogren's syndrome, that a near-identical defect to a murine model, a defect in the LMP2 subunit of the proteasome, was a contributor to this disease.
Hayashi, T., Kodama, S. & Faustman, D. L. Reply to 'LMP2 expression and proteasome activity in NOD mice'. Nature Med. 6, 1065–1066 (2000).
Kessel, A. et al. Increased CD8+ T cell apoptosis in scleroderma is associated with low levels of NF-κB. J. Clin. Immunol. 24, 30–36 (2004).
Welborn, M. B. III . et al. A human tumor necrosis factor p75 receptor agonist stimulates in vitro T cell proliferation but does not produce inflammation or shock in the baboon. J. Exp. Med. 184, 165–171 (1996). This article showed, that an agonist for TNFR2 is non-toxic to nonhuman primates in a dose–response manner.
Chen, X. et al. Cutting edge: expression of TNFR2 defines a maximally suppressive subset of mouse CD4+CD25+FoxP3+ T regulatory cells: applicability to tumor-infiltrating T regulatory cells. J. Immunol. 180, 6467–6471 (2008).
Nagar, M. et al. TNF/TNFR2 signaling in T regulatory (Treg) cells induces a distinctive NF-kappaB-dependent cellular program that modulates their suppressive capacity. J. Immunol. 184, 3570–3581 (2010).
Askenasy, N., Kaminitz, A. & Yarkoni, S. Mechanisms of T regulatory cell function. Autoimmun. Rev. 7, 370–375 (2008).
Douni, E. & Kollias, G. A critical role of the p75 tumor necrosis factor receptor (p75TNF-R.) in organ inflammation independent of TNF, lymphotoxin alpha, or the p55TNF-R. J. Exp. Med. 188, 1343–1352 (1998).
Li, S., Wang, L. & Dorf, M. E. PKC phosphorylation of TRAF2 mediates IKKα/β recruitment and K63-linked polyubiquitination. Mol. Cell 33, 30–42 (2009).
He, M. M. et al. Small-molecule inhibition of TNF-alpha. Science 310, 1022–1025 (2005).
Sheehan, K. C. et al. Monoclonal antibodies specific for murine p55 and p75 tumor necrosis factor receptors: identification of a novel in vivo role for p75. J. Exp. Med. 181, 607–617 (1995).
Loetscher, H., Stueber, D., Banner, D., Mackay, F. & Lesslauer, W. Human tumor necrosis factor-α (TNF-α) mutants with exclusive specificity for the 55-kDa or 75-kDa TNF receptors. J. Biol. Chem. 268, 26350–26357 (1993).
Kretschmer, K., Apostolou, I., Jaeckel, E., Khazaie, K. & von Boehmer, H. Making regulatory T cells with defined antigen specificity: role in autoimmunity and cancer. Immunol. Rev. 212, 163–169 (2006).
Fournel, S. et al. C3-symmetric peptide scaffolds are functional mimetics of trimeric CD40L. Nature Chem. Biol. 1, 377–382 (2005).
Herold, K. C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002).
Keymeulen, B. et al. Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N. Engl. J. Med. 352, 2598–2608 (2005).
Herold, K. C. et al. A single course of anti-CD3 monoclonal antibody hOKT3γ1(Ala-Ala) results in improvement in C-peptide responses and clinical parameters for at least 2 years after onset of type 1 diabetes. Diabetes 54, 1763–1769 (2005).
Herold, K. C. et al. Treatment of patients with new onset Type 1 diabetes with a single course of anti-CD3 mAb Teplizumab preserves insulin production for up to 5 years. Clin. Immunol. 132, 166–173 (2009).
Ablamunits, V. & Herold, K. C. Generation and function of human regulatory CD8+ T cells induced by a humanized OKT3 monoclonal antibody hOKT3γ1 (Ala-Ala). Hum. Immunol. 69, 732–736 (2008).
Carswell, E. A. et al. An endotoxin-induced serum factor that causes necrosis of tumors. Proc. Natl Acad. Sci. USA 72, 3666–3670 (1975).
Choi, J. W. et al. AIMP2 promotes TNFα-dependent apoptosis via ubiquitin-mediated degradation of TRAF2. J. Cell Sci. 122, 2710–2715 (2009).
Acknowledgements
We would like to thank the Iacocca Foundation for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
OMIM
FURTHER INFORMATION
FDA's MedWatch Safety Alerts — September 2009: Stronger Warnings for TNF Blockers:
Glossary
- Cytokine
-
A small protein that is responsible for signalling between immune cells or cells of other types. Once the cytokine is released from an immune cell, it migrates to a target cell with a matching receptor. On activation by the cytokine, the receptor triggers a signal through an internal signal induction pathway that, on reaching the nucleus, causes specific genes to be transcribed.
- Tumour necrosis factor
-
(TNF). A ubiquitous cytokine, it is a first responder to fight disease or infection. TNF acts on its target cell to regulate expression of up to 500 genes. Although its actions are simplified as being either pro-inflammatory or anti-inflammatory, TNF carries out far more complex actions, including cell death, survival, proliferation, differentiation and expression of other cytokines. TNF is described as a 'regulatory' cytokine because it controls the release of other cytokines necessary to wage the fight against disease or infection.
- Type 1 diabetes
-
The autoimmune form of diabetes, as opposed to the more common type 2 diabetes. With type 1, the immune system attacks and kills insulin-producing islet cells of the pancreas. Type 1 diabetes is a chronic disease beginning most commonly in childhood (hence the synonym, juvenile diabetes).
- Nuclear factor-κB
-
(NF-κB). One of the most prevalent and important transcription factors. It is an intracellular protein that, when activated, directly stimulates genes to induce expression. T cells rely on NF-κB for their survival.
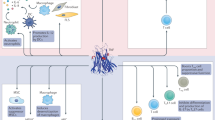
- Autoreactive T cell
-
A defective subset of T lymphocytes, or T cells, that attacks and kills the bodys own regular T-cell target proteins, for example, insulin, instead of attacking the same external targets. Although autoreactive T cells are supposed to be destroyed early in life, this rare type of T cell survives to mount autoimmune diseases.
- CD8+ cytotoxic T cell
-
A subset of T cells that directly kills its target proteins on cells. CD8 refers to a particular type of protein on the surface of these killer cells. When CD8 T cells are autoreactive, they kill the T cell's natural target proteins on cells.
- T-cell education
-
Describes the removal of newly formed autoreactive T cells and is the process by which antigen presenting cells properly present self peptides in the exterior facing human leukocyte antigen class I grooves. For autoreactive T cells this binding to self peptides results in death of these potentially bad cells prior to their release into the circulation.
- CD4+ T cell
-
A subset of T cells not directly responsible for killing target cells. Instead this type recruits other types of immune cells to assist in the battle. These cells bear CD4 proteins on their cellular surface. Some subpopulations of CD4 T cells can assist CD8+ cytotoxic T cells. Other subsets of CD4+ cells, called regulatory T cells function to turn off CD8+ cytotoxic T cells.
Rights and permissions
About this article
Cite this article
Faustman, D., Davis, M. TNF receptor 2 pathway: drug target for autoimmune diseases. Nat Rev Drug Discov 9, 482–493 (2010). https://doi.org/10.1038/nrd3030
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3030
This article is cited by
-
Development of a 1:1-binding biparatopic anti-TNFR2 antagonist by reducing signaling activity through epitope selection
Communications Biology (2023)
-
Bivalent structure of a TNFR2-selective and agonistic TNF-α mutein Fc-fusion protein enhances the expansion activity of regulatory T cells
Scientific Reports (2023)
-
Integrin β1, PDGFRβ, and type II collagen are essential for meniscus regeneration by synovial mesenchymal stem cells in rats
Scientific Reports (2022)
-
sTNFRII-Fc modification protects human UC-MSCs against apoptosis/autophagy induced by TNF-α and enhances their efficacy in alleviating inflammatory arthritis
Stem Cell Research & Therapy (2021)
-
Insights into the biology and therapeutic implications of TNF and regulatory T cells
Nature Reviews Rheumatology (2021)