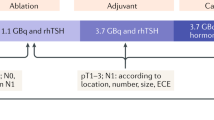
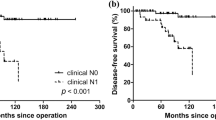
Abstract
Thyroid cancer is the most common malignancy of the endocrine system and its incidence has dramatically increased over the past three decades. Well-differentiated thyroid cancers (DTCs) are the main focus of this article, as they represent >90% of thyroid malignancies. This Review provides an overview of the controversies surrounding the optimal choice of surgery and extent of resection for patients with low-risk DTC or with papillary thyroid microcarcinoma, and the role of prophylactic central lymph node dissection. This Review also outlines the current surgical management of DTC and presents updated results for these techniques, along with important advances and current dilemmas in surgical approaches to treatment of these cancers. For example, endoscopic and robotic thyroidectomy are the two most recent innovations to present technical and other challenges to the endocrine surgeon; in addition, the risks as well as the advantages of same-day thyroid surgery, which has gained some acceptance, are detailed. Arguments for and against each approach are presented, along with supporting evidence. The authors' personal opinions are also provided for each topic.
Key Points
-
Thyroid cancer is the most common malignancy of the endocrine system and its incidence is increasing
-
The optimal therapy for papillary thyroid microcarcinoma is currently unclear, and is a controversial issue; suggested management strategies range from regular observation to total thyroidectomy
-
Prophylactic central lymph node dissection might identify high rates of lymph node involvement without providing long-term benefits; therefore, its routine use is currently unwarranted
-
Same-day thyroid surgery is gaining acceptance and can be safely performed in the majority of patients as long as an appropriate support system is present
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
06 December 2011
At the end of the second paragraph of the section 'Papillary thyroid microcarcinoma', it should read “In one study, tumor growth of 2 mm was detected in 30% of patients who underwent observation alone. More than one-third (56 of 162) of all patients assigned to observation underwent surgery after 19–56 months of observation, although for 47 of them this decision was not necessarily related to tumor progression. Lymph-node metastases were suspected by ultrasonography at the beginning of the study or during follow-up in 12.3% of all patients who underwent observation.” The error has been corrected for the HTML and PDF versions of the article.
References
Jemal, A. et al. Cancer statistics, 2007. CA Cancer J. Clin. 57, 43–66 (2007).
National Cancer Institute at the NIH. Cancer.gov [online], (2011).
Zarebczan, B. & Chen, H. Multi-targeted approach in the treatment of thyroid cancer. Minerva Chir. 65, 59–69 (2010).
Ito, Y. et al. Preoperative ultrasonographic examination for lymph node metastasis: usefulness when designing lymph node dissection for papillary microcarcinoma of the thyroid. World J. Surg. 28, 498–501 (2004).
Suliburk, J. & Delbridge, L. Surgical management of well-differentiated thyroid cancer: state of the art. Surg. Clin. North Am. 89, 1171–1191 (2009).
Cooper, D. S. et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19, 1167–1214 (2009).
Bilimoria, K. Y. et al. Extent of surgery affects survival for papillary thyroid cancer. Ann. Surg. 246, 375–381 (2007).
Hay, I. D., Bergstralh, E. J., Goellner, J. R., Ebersold, J. R. & Grant, C. S. Predicting outcome in papillary thyroid carcinoma: development of a reliable prognostic scoring system in a cohort of 1,779 patients surgically treated at one institution during 1940 through 1989. Surgery 114, 1050–1057 (1993).
Hay, I. D. et al. Papillary thyroid carcinoma managed at the Mayo Clinic during six decades (1940–1999): temporal trends in initial therapy and long-term outcome in 2,444 consecutively treated patients. World J. Surg. 26, 879–885 (2002).
Rubino, C. et al. Second primary malignancies in thyroid cancer patients. Br. J. Cancer 89, 1638–1644 (2003).
The American Association of Endocrine Surgeons Patient Education Site. The American Association of Endocrine Surgeons [online], (2011).
Kikuchi, S. et al. Complication of thyroidectomy in patients with radiation-induced thyroid neoplasms. Arch. Surg. 139, 1185–1188 (2004).
Sturniolo, G. et al. The recurrent laryngeal nerve related to thyroid surgery. Am. J. Surg. 177, 485–488 (1999).
Youngwirth, L., Benavidez, J., Sippel, R. & Chen, H. Parathyroid hormone deficiency after total thyroidectomy: incidence and time. J. Surg. Res. 163, 69–71 (2010).
Leyre, P. et al. Does the risk of compressive hematoma after thyroidectomy authorize 1-day surgery? Langenbecks Arch. Surg. 393, 733–737 (2008).
Shaha, A. R., Shah, J. P. & Loree, T. R. Differentiated thyroid cancer presenting initially with distant metastasis. Am. J. Surg. 174, 474–476 (1997).
Sanders, L. E. & Cady, B. Differentiated thyroid cancer: reexamination of risk groups and outcome of treatment. Arch. Surg. 133, 419–425 (1998).
Barney, B. M., Hitchcock, Y. J., Sharma, P., Shrieve, D. C. & Tward, J. D. Overall and cause-specific survival for patients undergoing lobectomy, near-total, or total thyroidectomy for differentiated thyroid cancer. Head Neck 33, 645–649 (2011).
Pitt, S. C., Sippel, R. S. & Chen, H. Contralateral papillary thyroid cancer: does size matter? Am. J. Surg. 197, 342–347 (2009).
Mazeh, H. et al. Multifocality in well-differentiated thyroid carcinomas calls for total thyroidectomy. Am. J. Surg. 201, 770–775 (2011).
Mazzaferri, E. L. What is the optimal initial treatment of low-risk papillary thyroid cancer (and why is it controversial)? Oncology (Williston Park) 23, 579–588 (2009).
Mendelsohn, A. H., Elashoff, D. A., Abemayor, E. & St. John, M. A. Surgery for papillary thyroid carcinoma: is lobectomy enough? Arch. Otolaryngol. Head Neck Surg. 136, 1055–1061 (2010).
Iyer, N. G. & Shaha, A. R. Management of thyroid nodules and surgery for differentiated thyroid cancer. Clin. Oncol. (R. Coll. Radiol.) 22, 405–412 (2010).
Cross, S., Wei, J. P., Kim, S. & Brams, D. M. Selective surgery and adjuvant therapy based on risk classifications of well-differentiated thyroid cancer. J. Surg. Oncol. 94, 678–682 (2006).
Shah, J. P. et al. Prognostic factors in differentiated carcinoma of the thyroid gland. Am. J. Surg. 164, 658–661 (1992).
Dean, D. S. & Hay, I. D. Prognostic indicators in differentiated thyroid carcinoma. Cancer Control 7, 229–239 (2000).
Pasieka, J. L. et al. Addition of nuclear DNA content to the AMES risk-group classification for papillary thyroid cancer. Surgery 112, 1154–1159 (1992).
Hedinger, C., Wiliams, E. D. & Sobin, L. H. in International Histological Classification of Tumors 1–18 (WHO, Geneva, 1988).
Davies, L., Ouellette, M., Hunter, M. & Welch, H. G. The increasing incidence of small thyroid cancers: where are the cases coming from? Laryngoscope 120, 2446–2451 (2010).
Hughes, D. T., Haymart, M. R., Miller, B. S., Gauger, P. G. & Doherty, G. M. The most commonly occurring papillary thyroid cancer in the United States is now a microcarcinoma in a patient older than 45 years. Thyroid 21, 231–236 (2011).
Hay, I. D. et al. Papillary thyroid microcarcinoma: a study of 900 cases observed in a 60-year period. Surgery 144, 980–987 (2008).
Chow, S. M. et al. Papillary microcarcinoma of the thyroid—prognostic significance of lymph node metastasis and multifocality. Cancer 98, 31–40 (2003).
Giordano, D., Gradoni, P., Oretti, G., Molina, E. & Ferri, T. Treatment and prognostic factors of papillary thyroid microcarcinoma. Clin. Otolaryngol. 35, 118–124 (2010).
Ito, Y. et al. An observation trial without surgical treatment in patients with papillary microcarcinoma of the thyroid. Thyroid 13, 381–387 (2003).
Mercante, G. et al. Prognostic factors affecting neck lymph node recurrence and distant metastasis in papillary microcarcinoma of the thyroid: results of a study in 445 patients. Thyroid 19, 707–716 (2009).
Ito, Y. & Miyauchi, A. Appropriate treatment for asymptomatic papillary microcarcinoma of the thyroid. Expert Opin. Pharmacother. 8, 3205–3215 (2007).
Ito, Y. et al. Clinical significance of metastasis to the central compartment from papillary microcarcinoma of the thyroid. World J. Surg. 30, 91–99 (2006).
Roti, E. et al. Clinical and histological characteristics of papillary thyroid microcarcinoma: results of a retrospective study in 243 patients. J. Clin. Endocrinol. Metab. 91, 2171–2178 (2006).
Arora, N. et al. Identification of borderline thyroid tumors by gene expression array analysis. Cancer 115, 5421–5431 (2009).
Dunki-Jacobs, E., Grannan, K., McDonough, S. & Engel, A. M. Clinically unsuspected papillary microcarcinomas of the thyroid: a common finding with favorable biology? Am. J. Surg. doi: 10.1016/j.amjsurg.2010.12.008.
Pellegriti, G. et al. Clinical behavior and outcome of papillary thyroid cancers smaller than 1.5 cm in diameter: study of 299 cases. J. Clin. Endocrinol. Metab. 89, 3713–3720 (2004).
Yu, X. M., Wan, Y., Sippel, R. S. & Chen, H. Should all papillary thyroid microcarcinomas be aggressively treated? An analysis of 18,445 cases. Ann. Surg. doi: 10.1097/SLA.0b013e318230036d.
Roh, J. L., Kim, J. M. & Park, C. I. Central lymph node metastasis of unilateral papillary thyroid carcinoma: patterns and factors predictive of nodal metastasis, morbidity, and recurrence. Ann. Surg. Oncol. 18, 2245–2250 (2011).
Fritze, D. & Doherty, G. M. Surgical management of cervical lymph nodes in differentiated thyroid cancer. Otolaryngol. Clin. North Am. 43, 285–300 (2010).
Bardet, S. et al. Macroscopic lymph-node involvement and neck dissection predict lymph-node recurrence in papillary thyroid carcinoma. Eur. J. Endocrinol. 158, 551–560 (2008).
Mazzaferri, E. L., Doherty, G. M. & Steward, D. L. The pros and cons of prophylactic central compartment lymph node dissection for papillary thyroid carcinoma. Thyroid 19, 683–689 (2009).
Sywak, M. et al. Routine ipsilateral level VI lymphadenectomy reduces postoperative thyroglobulin levels in papillary thyroid cancer. Surgery 140, 1000–1005 (2006).
Pereira, J. A. et al. Nodal yield, morbidity, and recurrence after central neck dissection for papillary thyroid carcinoma. Surgery 138, 1095–1100 (2005).
Bozec, A. et al. Clinical impact of cervical lymph node involvement and central neck dissection in patients with papillary thyroid carcinoma: a retrospective analysis of 368 cases. Eur. Arch. Otorhinolaryngol. 268, 1205–1212 (2011).
So, Y. K., Seo, M. Y. & Son, Y. I. Prophylactic central lymph node dissection for clinically node-negative papillary thyroid microcarcinoma: Influence on serum thyroglobulin level, recurrence rate, and postoperative complications. Surgery doi: 10.1016/j.surg.2011.02.004.
Rosenbaum, M. A. & McHenry, C. R. Central neck dissection for papillary thyroid cancer. Arch. Otolaryngol. Head Neck Surg. 135, 1092–1097 (2009).
Ito, Y. et al. Risk factors for recurrence to the lymph node in papillary thyroid carcinoma patients without preoperatively detectable lateral node metastasis: validity of prophylactic modified radical neck dissection. World J. Surg. 31, 2085–2091 (2007).
White, M. L., Gauger, P. G. & Doherty, G. M. Central lymph node dissection in differentiated thyroid cancer. World J. Surg. 31, 895–904 (2007).
Eichhorn, W. et al. Prognostic factors determining long-term survival in well-differentiated thyroid cancer: an analysis of four hundred eighty-four patients undergoing therapy and aftercare at the same institution. Thyroid 13, 949–958 (2003).
Xing, M. Prognostic utility of BRAF mutation in papillary thyroid cancer. Mol. Cell Endocrinol. 321, 86–93 (2010).
Nikiforov, Y. E. Molecular diagnostics of thyroid tumors. Arch. Pathol. Lab. Med. 135, 569–577 (2011).
Melck, A. L., Yip, L. & Carty, S. E. The utility of BRAF testing in the management of papillary thyroid cancer. Oncologist 15, 1285–1293 (2010).
Scheumann, G. F., Gimm, O., Wegener, G., Hundeshagen, H. & Dralle, H. Prognostic significance and surgical management of locoregional lymph node metastases in papillary thyroid cancer. World J. Surg. 18, 559–567 (1994).
Pacini, F. et al. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur. J. Endocrinol. 154, 787–803 (2006).
Burkey, S. H. et al. Reexploration for symptomatic hematomas after cervical exploration. Surgery 130, 914–920 (2001).
Steckler, R. M. Outpatient thyroidectomy: a feasibility study. Am. J. Surg. 152, 417–419 (1986).
Inabnet, W. B., Shifrin, A., Ahmed, L. & Sinha, P. Safety of same day discharge in patients undergoing sutureless thyroidectomy: a comparison of local and general anesthesia. Thyroid 18, 57–61 (2008).
Tuggle, C. T., Roman, S., Udelsman, R. & Sosa, J. A. Same-day thyroidectomy: a review of practice patterns and outcomes for 1,168 procedures in New York State. Ann. Surg. Oncol. 18, 1035–1040 (2011).
Snyder, S. K. et al. Outpatient thyroidectomy is safe and reasonable: experience with more than 1,000 planned outpatient procedures. J. Am. Coll. Surg. 210, 575–582 (2010).
Sitges-Serra, A. et al. Outcome of protracted hypoparathyroidism after total thyroidectomy. Br. J. Surg. 97, 1687–1695 (2010).
Sippel, R. S., Ozgul, O., Hartig, G. K., Mack, E. A. & Chen, H. Risks and consequences of incidental parathyroidectomy during thyroid resection. ANZ J. Surg. 77, 33–36 (2007).
Brunaud, L. et al. Incision length for standard thyroidectomy and parathyroidectomy: when is it minimally invasive? Arch. Surg. 138, 1140–1143 (2003).
Huscher, C. S., Chiodini, S., Napolitano, C. & Recher, A. Endoscopic right thyroid lobectomy. Surg. Endosc. 11, 877 (1997).
Miccoli, P., Berti, P., Conte, M., Bendinelli, C. & Marcocci, C. Minimally invasive surgery for thyroid small nodules: preliminary report. J. Endocrinol. Invest. 22, 849–851 (1999).
Terris, D. J., Angelos, P., Steward, D. L. & Simental, A. A. Minimally invasive video-assisted thyroidectomy: a multi-institutional North American experience. Arch. Otolaryngol. Head Neck Surg. 134, 81–84 (2008).
Miccoli, P. et al. Surgical treatment of low- and intermediate-risk papillary thyroid cancer with minimally invasive video-assisted thyroidectomy. J. Clin. Endocrinol. Metab. 94, 1618–1622 (2009).
Wilhelm, T. & Metzig, A. Endoscopic minimally invasive thyroidectomy (eMIT): a prospective proof-of-concept study in humans. World J. Surg. 35, 543–551 (2011).
Kang, S. W. et al. Robot-assisted endoscopic surgery for thyroid cancer: experience with the first 100 patients. Surg. Endosc. 23, 2399–2406 (2009).
Henry, J. F. Minimally invasive thyroid and parathyroid surgery is not a question of length of the incision. Langenbecks Arch. Surg. 393, 621–626 (2008).
Ryu, H. R. et al. Feasibility and safety of a new robotic thyroidectomy through a gasless, transaxillary single-incision approach. J. Am. Coll. Surg. 211, e13–e19 (2010).
Berber, E. et al. Robotic transaxillary thyroidectomy: report of 2 cases and description of the technique. Surg. Laparosc. Endosc. Percutan. Tech. 20, e60–e63 (2010).
Landry, C. S., Grubbs, E. G. & Perrier, N. D. Bilateral robotic-assisted transaxillary surgery. Arch. Surg. 145, 717–720 (2010).
Kuppersmith, R. B. & Holsinger, F. C. Robotic thyroid surgery: An initial experience with North American patients. Laryngoscope 121, 521–526 (2011).
Perrier, N. D. et al. Robotic thyroidectomy: a framework for new technology assessment and safe implementation. Thyroid 20, 1327–1332 (2010).
Dhiman, S. V. & Inabnet, W. B. Minimally invasive surgery for thyroid diseases and thyroid cancer. J. Surg. Oncol. 97, 665–668 (2008).
Author information
Authors and Affiliations
Contributions
H. Mazeh researched the data for the article. Both authors wrote the article, and provided substantial contributions to discussions of the content, as well as to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mazeh, H., Chen, H. Advances in surgical therapy for thyroid cancer. Nat Rev Endocrinol 7, 581–588 (2011). https://doi.org/10.1038/nrendo.2011.140
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.140
This article is cited by
-
Comparison of endoscopic bilateral areolar and robotic-assisted bilateral axillo-breast approach thyroidectomy in differentiated thyroid carcinoma: a propensity-matched retrospective cohort study
BMC Surgery (2023)
-
Integrative metabolomic characterization identifies plasma metabolomic signature in the diagnosis of papillary thyroid cancer
Oncogene (2022)
-
Thyroidectomy for thyroid cancer via transareola single-site endoscopic approach: results of a case-match study with large-scale population
Surgical Endoscopy (2022)
-
Crosstalk between circRNAs and the PI3K/AKT signaling pathway in cancer progression
Signal Transduction and Targeted Therapy (2021)
-
Emerging integrated nanoclay-facilitated drug delivery system for papillary thyroid cancer therapy
Scientific Reports (2016)