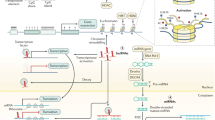
Abstract
Colorectal cancer (CRC) is a leading cause of cancer deaths worldwide. It results from an accumulation of genetic and epigenetic changes in colon epithelial cells, which transforms them into adenocarcinomas. Over the past decade, major advances have been made in understanding cancer epigenetics, particularly regarding aberrant DNA methylation. Assessment of the colon cancer epigenome has revealed that virtually all CRCs have aberrantly methylated genes and that the average CRC methylome has hundreds to thousands of abnormally methylated genes. As with gene mutations in the cancer genome, a subset of these methylated genes, called driver genes, is presumed to have a functional role in CRC. The assessment of methylated genes in CRCs has also revealed a unique molecular subgroup of CRCs called CpG island methylator phenotype (CIMP) cancers; these tumors have a particularly high frequency of methylated genes. These advances in our understanding of aberrant methylation in CRC have led to epigenetic alterations being developed as clinical biomarkers for diagnostic, prognostic and therapeutic applications. Progress in this field suggests that these epigenetic alterations will be commonly used in the near future to direct the prevention and treatment of CRC.
Key Points
-
The average colon cancer epigenome has hundreds to thousands of abnormally methylated genes, a subset of which is thought to drive colorectal cancer (CRC) pathogenesis and clinical behavior
-
Alterations in DNA methylation patterns in CRC contribute to the molecular heterogeneity of this cancer, as illustrated by the identification of a CpG island methylator phenotype (CIMP) that has a distinct epigenome
-
Clinical markers for diagnostic, prognostic and therapeutic applications are being developed using the epigenetic alterations and DNA methylation signatures in CRC
-
Although the mechanisms by which aberrant DNA methylation occurs are not yet fully understood, a mechanistic connection might exist between the altered methylation seen in CRC and the phenomena of age-related methylation and field cancerization
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Fearon, E. R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990).
Goldstein, N. S. Serrated pathway and APC (conventional)-type colorectal polyps: molecular-morphologic correlations, genetic pathways, and implications for classification. Am. J. Clin. Pathol. 125, 146–153 (2006).
Noffsinger, A. E. Serrated polyps and colorectal cancer: new pathway to malignancy. Annu. Rev. Pathol. 4, 343–364 (2009).
Ahnen, D. J. The American College of Gastroenterology Emily Couric Lecture—the adenoma–carcinoma sequence revisited: has the era of genetic tailoring finally arrived? Am. J. Gastroenterol. 106, 190–198 (2011).
Jemal, A. et al. Cancer statistics, 2006. CA Cancer J. Clin. 56, 106–130 (2006).
Pritchard, C. C. & Grady, W. M. Colorectal cancer molecular biology moves into clinical practice. Gut 60, 116–129 (2011).
Toyota, M. et al. CpG island methylator phenotype in colorectal cancer. Proc. Natl Acad. Sci. USA 96, 8681–8686 (1999).
Sjoblom, T. et al. The consensus coding sequences of human breast and colorectal cancers. Science 314, 268–274 (2006).
Jass, J. R. Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology 50, 113–130 (2007).
Ogino, S. & Goel, A. Molecular classification and correlates in colorectal cancer. J. Mol. Diagn. 10, 13–27 (2008).
Vogelstein, B. et al. Genetic alterations during colorectal-tumor development. N. Engl. J. Med. 319, 525–532 (1988).
Vazquez, A., Bond, E. E., Levine, A. J. & Bond, G. L. The genetics of the p53 pathway, apoptosis and cancer therapy. Nat. Rev. Drug Discov. 7, 979–987 (2008).
Grady, W. & Markowitz, S. D. TGF-β Signaling Pathway and Tumor Suppression in The TGF-β Family (eds Derynck, R. & Miyazano, K.) 889–938 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, 2008).
Grady, W. et al. Mutation of the type II transforming growth factor-β receptor is coincident with the transformation of human colon adenomas to malignant carcinomas. Cancer Res. 58, 3101–3104 (1998).
Markowitz, S. et al. Inactivation of the type II TGF-β receptor in colon cancer cells with microsatellite instability. Science 268, 1336–1338 (1995).
Eppert, K. et al. Madr2 maps to 18q21 and encodes a TGF-β regulated Mad-related protein that is functionally mutated in colorectal carcinoma. Cell 86, 543–552 (1996).
Takaku, K. et al. Intestinal tumorigenesis in compound mutant mice of both Dpc4(Smad4) and Apc genes. Cell 92, 645–656 (1998).
Wood, L. D. et al. The genomic landscapes of human breast and colorectal cancers. Science 318, 1108–1113 (2007).
Fearon, E. R. Molecular genetics of colorectal cancer. Annu. Rev. Pathol. 6, 479–507 (2011).
Samowitz, W. S. et al. Evaluation of a large, population-based sample supports a CpG island methylator phenotype in colon cancer. Gastroenterology 129, 837–845 (2005).
Nosho, K. et al. Comprehensive biostatistical analysis of CpG island methylator phenotype in colorectal cancer using a large population-based sample. PLoS ONE 3, e3698 (2008).
Grady, W. M. & Carethers, J. M. Genomic and epigenetic instability in colorectal cancer pathogenesis. Gastroenterology 135, 1079–1099 (2008).
Sharma, S., Kelly, T. K. & Jones, P. A. Epigenetics in cancer. Carcinogenesis 31, 27–36 (2010).
Suzuki, M. M. & Bird, A. DNA methylation landscapes: provocative insights from epigenomics. Nat. Rev. Genet. 9, 465–476 (2008).
Feinberg, A. P. & Tycko, B. The history of cancer epigenetics. Nat. Rev. Cancer 4, 143–153 (2004).
Sawan, C. & Herceg, Z. Histone modifications and cancer. Adv. Genet. 70, 57–85 (2010).
Ballestar, E. & Esteller, M. Epigenetic gene regulation in cancer. Adv. Genet. 61, 247–267 (2008).
Ting, A. H., McGarvey, K. M. & Baylin, S. B. The cancer epigenome—components and functional correlates. Genes Dev. 20, 3215–3231 (2006).
van Engeland, M., Derks, S., Smits, K. M., Meijer, G. A. & Herman, J. G. Colorectal cancer epigenetics: complex simplicity. J. Clin. Oncol. 29, 1382–1391 (2011).
Bestor, T. H. The DNA methyltransferases of mammals. Hum. Mol. Genet. 9, 2395–2402 (2000).
McCabe, M. T., Lee, E. K. & Vertino, P. M. A multifactorial signature of DNA sequence and polycomb binding predicts aberrant CpG island methylation. Cancer Res. 69, 282–291 (2009).
Gardiner-Garden, M. & Frommer, M. CpG islands in vertebrate genomes. J. Mol. Biol. 196, 261–282 (1987).
Deaton, A. M. & Bird, A. CpG islands and the regulation of transcription. Genes Dev. 25, 1010–1022 (2011).
Caiafa, P. & Zampieri, M. DNA methylation and chromatin structure: the puzzling CpG islands. J. Cell Biochem. 94, 257–265 (2005).
Hinoue, T. et al. Genome-scale analysis of aberrant DNA methylation in colorectal cancer. Genome Res. doi:10.1101/gr.117523.110.
Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 16, 6–21 (2002).
Hellman, A. & Chess, A. Gene body-specific methylation on the active X chromosome. Science 315, 1141–1143 (2007).
Reik, W. & Lewis, A. Co-evolution of X-chromosome inactivation and imprinting in mammals. Nat. Rev. Genet. 6, 403–410 (2005).
Hermann, A., Gowher, H. & Jeltsch, A. Biochemistry and biology of mammalian DNA methyltransferases. Cell. Mol. Life Sci. 61, 2571–2587 (2004).
Okano, M., Bell, D., Haber, D. & Li, E. DNA methylatransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 29, 247–257 (1999).
Xu, G. L. et al. Chromosome instability and immunodeficiency syndrome caused by mutations in a DNA methyltransferase gene. Nature 402, 187–191 (1999).
Laird, P. et al. Suppression of intestinal neoplasia by DNA hypomethylation. Cell 81, 197–205 (1995).
Berdasco, M. & Esteller, M. Aberrant epigenetic landscape in cancer: how cellular identity goes awry. Dev. Cell 19, 698–711 (2010).
Comb, M. & Goodman, H. M. CpG methylation inhibits proenkephalin gene expression and binding of the transcription factor AP-2. Nucleic Acids Res. 18, 3975–3982 (1990).
Inamdar, N. M., Ehrlich, K. C. & Ehrlich, M. CpG methylation inhibits binding of several sequence-specific DNA-binding proteins from pea, wheat, soybean and cauliflower. Plant Mol. Biol. 17, 111–123 (1991).
Campanero, M. R., Armstrong, M. I. & Flemington, E. K. CpG methylation as a mechanism for the regulation of E2F activity. Proc. Natl Acad. Sci. USA 97, 6481–6486 (2000).
Deng, G., Chen, A., Pong, E. & Kim, Y. S. Methylation in hMLH1 promoter interferes with its binding to transcription factor CBF and inhibits gene expression. Oncogene 20, 7120–7127 (2001).
Bednarik, D. P. et al. DNA CpG methylation inhibits binding of NF-kappa B proteins to the HIV-1 long terminal repeat cognate DNA motifs. New Biol. 3, 969–976 (1991).
Tsai, H. C. & Baylin, S. B. Cancer epigenetics: linking basic biology to clinical medicine. Cell Res. 21, 502–517 (2011).
Kirmizis, A. et al. Silencing of human polycomb target genes is associated with methylation of histone H3 Lys 27. Genes Dev. 18, 1592–1605 (2004).
Cao, R. et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 298, 1039–1043 (2002).
Irizarry, R. A. et al. The human colon cancer methylome shows similar hypo- and hypermethylation at conserved tissue-specific CpG island shores. Nat. Genet. 41, 178–186 (2009).
Doi, A. et al. Differential methylation of tissue- and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nat. Genet. 41, 1350–1353 (2009).
Feinberg, A. & Vogelstein, B. Hypomethylation distinguishes genes of some human cancers from their normal counterparts. 301, 89–92 (1983).
Feinberg, A. P. The epigenetics of cancer etiology. Semin. Cancer Biol. 14, 427–432 (2004).
Ehrlich, M. DNA methylation in cancer: too much, but also too little. Oncogene 21, 5400–5413 (2002).
Suzuki, K. et al. Global DNA demethylation in gastrointestinal cancer is age dependent and precedes genomic damage. Cancer Cell 9, 199–207 (2006).
Sakatani, T. et al. Loss of imprinting of Igf2 alters intestinal maturation and tumorigenesis in mice. Science 307, 1976–1978 (2005).
Cui, H. et al. Loss of imprinting in colorectal cancer linked to hypomethylation of H19 and IGF2. Cancer Res. 62, 6442–6446 (2002).
Pfeifer, G. P. & Rauch, T. A. DNA methylation patterns in lung carcinomas. Semin. Cancer Biol. 19, 181–187 (2009).
Costello, J. F. et al. Aberrant CpG-island methylation has non-random and tumour-type-specific patterns. Nat. Genet. 24, 132–138 (2000).
Gonzalez-Zulueta, M. et al. Methylation of the 5′ CpG island of the p16/CDKN2 tumor suppressor gene in normal and transformed human tissues correlates with gene silencing. Cancer Res. 55, 4531–4535 (1995).
Graff, J. et al. E-cadherin expression is silenced by DNA hypermethylation in human breast and prostate carcinomas. Cancer Res. 55, 5195–5199 (1995).
Herman, J. et al. Silencing of the VHL tumor-suppressor gene by DNA methylation in renal carcinoma. Proc. Natl Acad. Sci. USA 91, 9700–9704 (1994).
Cunningham, J. et al. Hypermethylation of the hMLH1 promoter in colon cancer with microsatellite instability. Cancer Res. 58, 3455–3460 (1998).
Grady, W. et al. Methylation of the CDH1 promoter as the second genetic hit in hereditary diffuse gastric cancer. Nat. Genet. 26, 16–17 (2000).
Baylin, S. B. & Bestor, T. H. Altered methylation patterns in cancer cell genomes: cause or consequence. Cancer Cell 1, 299–305 (2002).
Suzuki, H. et al. Epigenetic inactivation of SFRP genes allows constitutive WNT signaling in colorectal cancer. Nat. Genet. 36, 417–422 (2004).
Schuebel, K. E. et al. Comparing the DNA hypermethylome with gene mutations in human colorectal cancer. PLoS Genet. 3, 1709–1723 (2007).
Knudson, A. G. Jr & Strong, L. C. Mutation and cancer: a model for Wilms' tumor of the kidney. J. Natl Cancer Inst. 48, 313–324 (1972).
Issa, J. P. et al. Increased cytosine DNA-methyltransferase activity during colon cancer progression. J. Natl Cancer Inst. 85, 1235–1240 (1993).
Nosho, K. et al. DNMT3B expression might contribute to CpG island methylator phenotype in colorectal cancer. Clin. Cancer Res. 15, 3663–3671 (2009).
Schmidt, W. M. et al. Progressive up-regulation of genes encoding DNA methyltransferases in the colorectal adenoma-carcinoma sequence. Mol. Carcinog. 46, 766–772 (2007).
Ibrahim, A. E. et al. Sequential DNA methylation changes are associated with DNMT3B overexpression in colorectal neoplastic progression. Gut 60, 499–508 (2011).
Steine, E. J. et al. Genes methylated by DNA methyltransferase 3b are similar in mouse intestine and human colon cancer. J. Clin. Invest. 121, 1748–1752 (2011).
Ehrlich, M. et al. Quantitative analysis of associations between DNA hypermethylation, hypomethylation, and DNMT RNA levels in ovarian tumors. Oncogene 25, 2636–2645 (2006).
Schlesinger, Y. et al. Polycomb-mediated methylation on Lys27 of histone H3 pre-marks genes for de novo methylation in cancer. Nat. Genet. 39, 232–236 (2007).
Ohm, J. E. et al. A stem cell-like chromatin pattern may predispose tumor suppressor genes to DNA hypermethylation and heritable silencing. Nat. Genet. 39, 237–242 (2007).
Handa, V. & Jeltsch, A. Profound flanking sequence preference of Dnmt3a and Dnmt3b mammalian DNA methyltransferases shape the human epigenome. J. Mol. Biol. 348, 1103–1112 (2005).
Feltus, F. A., Lee, E. K., Costello, J. F., Plass, C. & Vertino, P. M. DNA motifs associated with aberrant CpG island methylation. Genomics 87, 572–579 (2006).
Xie, H. et al. Genome-wide quantitative assessment of variation in DNA methylation patterns. Nucleic Acids Res. 39, 4099–4108 (2011).
Dickson, J. et al. VEZF1 elements mediate protection from DNA methylation. PLoS Genet. 6, e1000804 (2010).
Frigola, J. et al. Epigenetic remodeling in colorectal cancer results in coordinate gene suppression across an entire chromosome band. Nat. Genet. 38, 540–549 (2006).
Valinluck, V., Liu, P., Kang, J. I. Jr, Burdzy, A. & Sowers, L. C. 5-halogenated pyrimidine lesions within a CpG sequence context mimic 5-methylcytosine by enhancing the binding of the methyl-CpG-binding domain of methyl-CpG-binding protein 2 (MeCP2). Nucleic Acids Res. 33, 3057–3064 (2005).
Valinluck, V. & Sowers, L. C. Endogenous cytosine damage products alter the site selectivity of human DNA maintenance methyltransferase DNMT1. Cancer Res. 67, 946–950 (2007).
Valinluck, V. & Sowers, L. C. Inflammation-mediated cytosine damage: a mechanistic link between inflammation and the epigenetic alterations in human cancers. Cancer Res. 67, 5583–5586 (2007).
Huang, J., Wang, Y., Guo, Y. & Sun, S. Down-regulated microRNA-152 induces aberrant DNA methylation in hepatitis B virus-related hepatocellular carcinoma by targeting DNA methyltransferase 1. Hepatology 52, 60–70 (2010).
Fabbri, M. et al. MicroRNA-29 family reverts aberrant methylation in lung cancer by targeting DNA methyltransferases 3A and 3B. Proc. Natl Acad. Sci. USA 104, 15805–15810 (2007).
Garzon, R. et al. MicroRNA-29b induces global DNA hypomethylation and tumor suppressor gene reexpression in acute myeloid leukemia by targeting directly DNMT3A and 3B and indirectly DNMT1. Blood 113, 6411–6418 (2009).
Watanabe, T. et al. Role for piRNAs and noncoding RNA in de novo DNA methylation of the imprinted mouse Rasgrf1 locus. Science 332, 848–852 (2011).
Saito, Y. & Jones, P. A. Epigenetic activation of tumor suppressor microRNAs in human cancer cells. Cell Cycle 5, 2220–2222 (2006).
Ahuja, N. & Issa, J. P. Aging, methylation and cancer. Histol. Histopathol. 15, 835–842 (2000).
Toyota, M. & Issa, J. P. CpG island methylator phenotypes in aging and cancer. Semin. Cancer Biol. 9, 349–357 (1999).
Issa, J.-P. et al. Methylation of the oestrogen receptor CpG island links ageing and neoplasia in human colon. Nat. Genet. 7, 536–540 (1994).
Fink, D. et al. The role of DNA mismatch repair in platinum drug resistance. Cancer Res. 56, 4881–4886 (1996).
Wilson, V. L. & Jones, P. A. DNA methylation decreases in aging but not in immortal cells. Science 220, 1055–1057 (1983).
Fuke, C. et al. Age related changes in 5-methylcytosine content in human peripheral leukocytes and placentas: an HPLC-based study. Ann. Hum. Genet. 68, 196–204 (2004).
Fraga, M. F. & Esteller, M. Epigenetics and aging: the targets and the marks. Trends Genet. 23, 413–418 (2007).
Yates, P. A., Burman, R. W., Mummaneni, P., Krussel, S. & Turker, M. S. Tandem B1 elements located in a mouse methylation center provide a target for de novo DNA methylation. J. Biol. Chem. 274, 36357–36361 (1999).
Hazra, A. et al. Germline polymorphisms in the one-carbon metabolism pathway and DNA methylation in colorectal cancer. Cancer Causes Control 21, 331–345 (2010).
Curtin, K. et al. Genetic polymorphisms in one-carbon metabolism: associations with CpG island methylator phenotype (CIMP) in colon cancer and the modifying effects of diet. Carcinogenesis 28, 1672–1679 (2007).
Casillas, M. A. Jr, Lopatina, N., Andrews, L. G. & Tollefsbol, T. O. Transcriptional control of the DNA methyltransferases is altered in aging and neoplastically-transformed human fibroblasts. Mol. Cell. Biochem. 252, 33–43 (2003).
Alemayehu, A., Sebova, K. & Fridrichova, I. Redundant DNA methylation in colorectal cancers of Lynch-syndrome patients. Genes Chromosomes Cancer 47, 906–914 (2008).
Slaughter, D. P., Southwick, H. W. & Smejkal, W. Field cancerization in oral stratified squamous epithelium; clinical implications of multicentric origin. Cancer 6, 963–968 (1953).
Polley, A. C. et al. Proteomic analysis reveals field-wide changes in protein expression in the morphologically normal mucosa of patients with colorectal neoplasia. Cancer Res. 66, 6553–6562 (2006).
Jothy, S. et al. Field effect of human colon carcinoma on normal mucosa: relevance of carcinoembryonic antigen expression. Tumour Biol. 17, 58–64 (1996).
Ramirez, N. et al. Epigenetic events in normal colonic mucosa surrounding colorectal cancer lesions. Eur. J. Cancer 44, 2689–2695 (2008).
Worthley, D. L. et al. DNA methylation within the normal colorectal mucosa is associated with pathway-specific predisposition to cancer. Oncogene 29, 1653–1662 (2010).
Novak, P., Jensen, T. J., Garbe, J. C., Stampfer, M. R. & Futscher, B. W. Stepwise DNA methylation changes are linked to escape from defined proliferation barriers and mammary epithelial cell immortalization. Cancer Res. 69, 5251–5258 (2009).
Widschwendter, M. et al. Epigenetic stem cell signature in cancer. Nat. Genet. 39, 157–158 (2007).
Shen, L. et al. MGMT promoter methylation and field defect in sporadic colorectal cancer. J. Natl Cancer Inst. 97, 1330–1338 (2005).
Milicic, A. et al. Ectopic expression of P-cadherin correlates with promoter hypomethylation early in colorectal carcinogenesis and enhanced intestinal crypt fission in vivo. Cancer Res. 68, 7760–7768 (2008).
Shen, L., Kondo, Y., Hamilton, S. R., Rashid, A. & Issa, J. P. P14 methylation in human colon cancer is associated with microsatellite instability and wild-type p53. Gastroenterology 124, 626–633 (2003).
Grady, W. M. et al. Epigenetic silencing of the intronic microRNA hsa-miR-342 and its host gene EVL in colorectal cancer. Oncogene 27, 3880–3888 (2008).
Belshaw, N. J. et al. Profiling CpG island field methylation in both morphologically normal and neoplastic human colonic mucosa. Br. J. Cancer 99, 136–142 (2008).
Konishi, K. et al. Concordant DNA methylation in synchronous colorectal carcinomas. Cancer Prev. Res. (Phila.) 2, 814–822 (2009).
Johnson, I. T. & Belshaw, N. J. Environment, diet and CpG island methylation: epigenetic signals in gastrointestinal neoplasia. Food Chem. Toxicol. 46, 1346–1359 (2008).
Hiraoka, S. et al. Methylation status of normal background mucosa is correlated with occurrence and development of neoplasia in the distal colon. Hum. Pathol. 41, 38–47 (2010).
Wallace, K. et al. Effect of calcium supplementation on the risk of large bowel polyps. J. Natl Cancer Inst. 96, 921–925 (2004).
Limsui, D. et al. Cigarette smoking and colorectal cancer risk by molecularly defined subtypes. J. Natl Cancer Inst. 102, 1012–1022 (2010).
Worthley, D. L. et al. DNA methylation in the rectal mucosa is associated with crypt proliferation and fecal short-chain fatty acids. Dig. Dis. Sci. 56, 387–396 (2011).
Shannon, B., Gnanasampanthan, S., Beilby, J. & Iacopetta, B. A polymorphism in the methylenetetrahydrofolate reductase gene predisposes to colorectal cancers with microsatellite instability. Gut 50, 520–524 (2002).
Rashid, A., Shen, L., Morris, J. S., Issa, J. P. & Hamilton, S. R. CpG island methylation in colorectal adenomas. Am. J. Pathol. 159, 1129–1135 (2001).
Chan, A. O. et al. CpG island methylation in aberrant crypt foci of the colorectum. Am. J. Pathol. 160, 1823–1830 (2002).
Li, H. et al. SLC5A8, a sodium transporter, is a tumor suppressor gene silenced by methylation in human colon aberrant crypt foci and cancers. Proc. Natl Acad. Sci. USA 100, 8412–8417 (2003).
Qi, J., Zhu, Y. Q., Luo, J. & Tao, W. H. Hypermethylation and expression regulation of secreted frizzled-related protein genes in colorectal tumor. World J. Gastroenterol. 12, 7113–7117 (2006).
Ausch, C. et al. Comparative analysis of PCR-based biomarker assay methods for colorectal polyp detection from fecal DNA. Clin. Chem. 55, 1559–1563 (2009).
Kim, Y. H. et al. CpG island methylation of genes accumulates during the adenoma progression step of the multistep pathogenesis of colorectal cancer. Genes Chromosomes Cancer 45, 781–789 (2006).
Oster, B. et al. Identification and validation of highly frequent CpG island hypermethylation in colorectal adenomas and carcinomas. Int. J. Cancer doi:10.1002/ijc.25951.
Sunami, E., de Maat, M., Vu, A., Turner, R. R. & Hoon, D. S. LINE-1 hypomethylation during primary colon cancer progression. PLoS ONE 6, e18884 (2011).
Esteller, M. et al. Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N. Engl. J. Med. 343, 1350–1354 (2000).
Nosho, K. et al. PIK3CA mutation in colorectal cancer: relationship with genetic and epigenetic alterations. Neoplasia 10, 534–541 (2008).
Yano, Y. et al. Clinicopathological and molecular features of colorectal serrated neoplasias with different mucosal crypt patterns. Am. J. Gastroenterol. 106, 1351–1358 (2011).
Dhir, M. et al. Sessile serrated adenomas and classical adenomas: An epigenetic perspective on premalignant neoplastic lesions of the gastrointestinal tract. Int. J. Cancer doi:10.1002/ijc.25847.
Wendt, M. K. et al. Silencing of epithelial CXCL12 expression by DNA hypermethylation promotes colonic carcinoma metastasis. Oncogene 25, 4986–4997 (2006).
Kim, M. S., Lee, J. & Sidransky, D. DNA methylation markers in colorectal cancer. Cancer Metastasis Rev. 29, 181–206 (2010).
Ogino, S. et al. LINE-1 hypomethylation is inversely associated with microsatellite instability and CpG island methylator phenotype in colorectal cancer. Int. J. Cancer 122, 2767–2773 (2008).
Estecio, M. R. et al. LINE-1 hypomethylation in cancer is highly variable and inversely correlated with microsatellite instability. PLoS ONE 2, e399 (2007).
Ogino, S. et al. CpG island methylator phenotype (CIMP) of colorectal cancer is best characterised by quantitative DNA methylation analysis and prospective cohort studies. Gut 55, 1000–1006 (2006).
Weisenberger, D. J. et al. CpG island methylator phenotype underlies sporadic microsatellite instability and is tightly associated with BRAF mutation in colorectal cancer. Nat. Genet. 38, 787–793 (2006).
Ogino, S. et al. Evaluation of markers for CpG island methylator phenotype (CIMP) in colorectal cancer by a large population-based sample. J. Mol. Diagn. 9, 305–314 (2007).
Kaneda, A. & Yagi, K. Two groups of DNA methylation markers to classify colorectal cancer into three epigenotypes. Cancer Sci. 102, 18–24 (2011).
Weisenberger, D. J. et al. DNA methylation analysis by digital bisulfite genomic sequencing and digital MethyLight. Nucleic Acids Res. 36, 4689–4698 (2008).
Curtin, K., Slattery, M. L. & Samowitz, W. S. CpG island methylation in colorectal cancer: past, present and future. Patholog. Res. Int. 2011, 902674 (2011).
Samowitz, W. S. et al. Association of smoking, CpG island methylator phenotype, and V600E BRAF mutations in colon cancer. J. Natl Cancer Inst. 98, 1731–1738 (2006).
Hughes, L. A. et al. Early life exposure to famine and colorectal cancer risk: a role for epigenetic mechanisms. PLoS ONE 4, e7951 (2009).
Hughes, L. A. et al. Body size, physical activity and risk of colorectal cancer with or without the CpG island methylator phenotype (CIMP). PLoS ONE 6, e18571 (2011).
Schernhammer, E. S. et al. Dietary folate, alcohol and B vitamins in relation to LINE-1 hypomethylation in colon cancer. Gut 59, 794–799 (2010).
Torlakovic, E. E. et al. Sessile serrated adenoma (SSA) vs. traditional serrated adenoma (TSA). Am. J. Surg. Pathol. 32, 21–29 (2008).
Silver, A. et al. A distinct DNA methylation profile associated with microsatellite and chromosomal stable sporadic colorectal cancers. Int. J. Cancer doi:10.1002/ijc.26104.
Shen, L. et al. Integrated genetic and epigenetic analysis identifies three different subclasses of colon cancer. Proc. Natl Acad. Sci. USA 104, 18654–18659 (2007).
Barault, L. et al. Hypermethylator phenotype in sporadic colon cancer: study on a population-based series of 582 cases. Cancer Res. 68, 8541–8546 (2008).
Ogino, S., Kawasaki, T., Kirkner, G. J., Loda, M. & Fuchs, C. S. CpG island methylator phenotype-low (CIMP-low) in colorectal cancer: possible associations with male sex and KRAS mutations. J. Mol. Diagn. 8, 582–588 (2006).
Kim, J. H., Shin, S. H., Kwon, H. J., Cho, N. Y. & Kang, G. H. Prognostic implications of CpG island hypermethylator phenotype in colorectal cancers. Virchows Arch. 455, 485–494 (2009).
Yagi, K. et al. Three DNA methylation epigenotypes in human colorectal cancer. Clin. Cancer Res. 16, 21–33 (2010).
Iacopetta, B., Kawakami, K. & Watanabe, T. Predicting clinical outcome of 5-fluorouracil-based chemotherapy for colon cancer patients: is the CpG island methylator phenotype the 5-fluorouracil-responsive subgroup? Int. J. Clin. Oncol. 13, 498–503 (2008).
Tanaka, N. et al. Novel application of structural equation modeling to correlation structure analysis of CpG island methylation in colorectal cancer. Am. J. Pathol. 177, 2731–2740 (2010).
Hinoue, T. et al. Analysis of the association between CIMP and BRAF in colorectal cancer by DNA methylation profiling. PLoS ONE 4, e8357 (2009).
Bastian, P. J. et al. GSTP1 hypermethylation as a molecular marker in the diagnosis of prostatic cancer: is there a correlation with clinical stage, Gleason grade, PSA value or age? Eur. J. Med. Res. 9, 523–527 (2004).
Smith, R. A., Cokkinides, V., Brooks, D., Saslow, D. & Brawley, O. W. Cancer screening in the United States, 2010: a review of current American Cancer Society guidelines and issues in cancer screening. CA Cancer J. Clin. 60, 99–119 (2010).
Ahlquist, D. A. et al. Stool DNA and occult blood testing for screen detection of colorectal neoplasia. Ann. Intern. Med. 149, 441–450, W481 (2008).
Itzkowitz, S. H. et al. Improved fecal DNA test for colorectal cancer screening. Clin. Gastroenterol. Hepatol. 5, 111–117 (2007).
Chen, W. D. et al. Detection in fecal DNA of colon cancer-specific methylation of the nonexpressed vimentin gene. J. Natl Cancer Inst. 97, 1124–1132 (2005).
Itzkowitz, S. et al. A simplified, noninvasive stool DNA test for colorectal cancer detection. Am. J. Gastroenterol. 103, 2862–2870 (2008).
Li, M. et al. Sensitive digital quantification of DNA methylation in clinical samples. Nat. Biotechnol. 27, 858–863 (2009).
Bouzourene, H., Hutter, P., Losi, L., Martin, P. & Benhattar, J. Selection of patients with germline MLH1 mutated Lynch syndrome by determination of MLH1 methylation and BRAF mutation. Fam. Cancer 9, 167–172 (2010).
Ide, T. et al. Expression of the hMLH1 gene is a possible predictor for the clinical response to 5-fluorouracil after a surgical resection in colorectal cancer. Oncol. Rep. 19, 1571–1576 (2008).
Jover, R. et al. 5-Fluorouracil adjuvant chemotherapy does not increase survival in patients with CpG island methylator phenotype colorectal cancer. Gastroenterology 140, 1174–1181 (2011).
Ogino, S. et al. CpG island methylation, response to combination chemotherapy, and patient survival in advanced microsatellite stable colorectal carcinoma. Virchows Arch. 450, 529–537 (2007).
Ogino, S. et al. A cohort study of tumoral LINE-1 hypomethylation and prognosis in colon cancer. J. Natl Cancer Inst. 100, 1734–1738 (2008).
Ahn, J. B. et al. DNA methylation predicts recurrence from resected stage III proximal colon cancer. Cancer 117, 1847–1854 (2011).
Acknowledgements
This work was supported in part by funds from the FHCRC CCSG (5P30CA015704), Burroughs Wellcome Fund, NCI/EDRN (UO1CA152756) and NCI (U54CA143862, PO1CA077852) (W. M. Grady) and ASCRS (GSRRIG 10) and NIH T32 (T32DK007742-14) (V. V. Lao).
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lao, V., Grady, W. Epigenetics and colorectal cancer. Nat Rev Gastroenterol Hepatol 8, 686–700 (2011). https://doi.org/10.1038/nrgastro.2011.173
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2011.173
This article is cited by
-
Lysine demethylase 5C inhibits transcription of prefoldin subunit 5 to activate c-Myc signal transduction and colorectal cancer progression
Molecular Medicine (2024)
-
DNA demethylation and tri-methylation of H3K4 at the TACSTD2 promoter are complementary players for TROP2 regulation in colorectal cancer cells
Scientific Reports (2024)
-
DNA hypomethylation patterns and their impact on the tumor microenvironment in colorectal cancer
Cellular Oncology (2024)
-
Integration of single-cell transcriptomics and epigenetic analysis reveals enhancer-controlled TIMP1 as a regulator of ferroptosis in colorectal cancer
Genes & Genomics (2024)
-
SPOCK1 and POSTN are valuable prognostic biomarkers and correlate with tumor immune infiltrates in colorectal cancer
BMC Gastroenterology (2023)