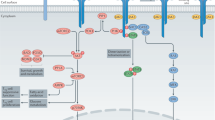
Key Points
-
Colony-stimulating factors (CSFs) — granulocyte/macrophage CSF (GM-CSF), macrophage CSF (M-CSF) and granulocyte CSF (G-CSF) — have wider functions than their original in vitro definition as haematopoietic-cell growth factors.
-
Recent extensive animal data indicate that CSF depletion provides benefit in many inflammatory and/or autoimmune conditions.
-
Early-phase clinical trials targeting GM-CSF and M-CSF have commenced.
-
The distinct biological properties of the CSFs offer specific targeting opportunities, but with some associated risks.
-
Information about the background biology of the CSFs is required to predict the probable specific outcomes of CSF blockade.
-
There are several outstanding questions regarding CSF biology that need to be addressed to aid the design of future clinical trials.
Abstract
Although they were originally defined as haematopoietic-cell growth factors, colony-stimulating factors (CSFs) have been shown to have additional functions by acting directly on mature myeloid cells. Recent data from animal models indicate that the depletion of CSFs has therapeutic benefit in many inflammatory and/or autoimmune conditions and as a result, early-phase clinical trials targeting granulocyte/macrophage colony-stimulating factor and macrophage colony-stimulating factor have now commenced. The distinct biological features of CSFs offer opportunities for specific targeting, but with some associated risks. Here, I describe these biological features, discuss the probable specific outcomes of targeting CSFs in vivo and highlight outstanding questions that need to be addressed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Burgess, A. W. & Metcalf, D. The nature and action of granulocyte–macrophage colony stimulating factors. Blood 56, 947–958 (1980). This is a seminal early review of CSFs and haematopoiesis.
Handman, E. & Burgess, A. W. Stimulation by granulocyte–macrophage colony-stimulating factor of Leishmania tropica killing by macrophages. J. Immunol. 122, 1134–1137 (1979).
Hamilton, J. A., Stanley, E. R., Burgess, A. W. & Shadduck, R. K. Stimulation of macrophage plasminogen activator activity by colony-stimulating factors. J. Cell. Physiol. 103, 435–445 (1980). This paper was the first to indicate that both GM-CSF and M-CSF could be considered as pro-inflammatory cytokines.
Whetton, A. D. & Dexter, T. M. Myeloid haemopoietic growth factors. Biochim. Biophys. Acta 989, 111–132 (1989).
Hamilton, J. A. Rheumatoid arthritis: opposing actions of haemopoietic growth factors and slow-acting anti-rheumatic drugs. Lancet 342, 536–539 (1993). This hypothesis paper is seminal to many concepts in the Review.
Verreck, F. A. et al. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc. Natl Acad. Sci. USA 101, 4560–4565 (2004).
Fleetwood, A. J., Lawrence, T., Hamilton, J. A. & Cook, A. D. GM-CSF and M-CSF (CSF-1)-dependent macrophage phenotypes display differences in cytokine profiles and transcription factor activities — implications for CSF blockade in inflammation. J. Immunol. 178, 5245–5252 (2007).
McKenzie, B. S., Kastelein, R. A. & Cua, D. J. Understanding the IL-23–IL-17 immune pathway. Trends Immunol. 27, 17–23 (2006).
Stark, M. A. et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 22, 285–294 (2005).
Paniagua, R. T. et al. Selective tyrosine kinase inhibition by imatinib mesylate for the treatment of autoimmune arthritis. J. Clin. Invest. 116, 2633–2642 (2006).
Stanley, E. R. et al. Biology and action of colony-stimulating factor-1. Mol. Reprod. Dev. 46, 4–10 (1997).
Griffin, J. D. et al. The biology of GM-CSF: regulation of production and interaction with its receptor. Int. J. Cell Cloning 8 (Suppl. 1), 35–44 (1990).
Metcalf, D. Hematopoietic regulators: redundancy or subtlety? Blood 82, 3515–3523 (1993).
Hamilton, J. A. GM-CSF in inflammation and autoimmunity. Trends Immunol. 23, 403–408 (2002).
Hamilton, J. A. & Anderson, G. P. GM-CSF biology. Growth Factors 22, 225–231 (2004).
Fleetwood, A. J., Cook, A. D. & Hamilton, J. A. Functions of granulocyte–macrophage colony-stimulating factor. Crit. Rev. Immunol. 25, 405–428 (2005).
Chitu, V. & Stanley, E. R. Colony-stimulating factor-1 in immunity and inflammation. Curr. Opin. Immunol. 18, 39–48 (2006).
Metcalf, D. & Nicola, N. A. The Hemopoietic Colony-Stimulating Factors: from Biology to Clinical Applications (University Press, Cambridge, 1995).
Dai, X. M., Zong, X. H., Sylvestre, V. & Stanley, E. R. Incomplete restoration of colony-stimulating factor 1 (CSF-1) function in CSF-1-deficient Csf1op/Csf1op mice by transgenic expression of cell surface CSF-1. Blood 103, 1114–1123 (2004).
Sherr, C. J. et al. The c-fms proto-oncogene product is related to the receptor for the mononuclear phagocyte growth factor, CSF-1. Cell 41, 665–676 (1985). This paper defined the M-CSF receptor as the proto-oncogene Fms.
Pixley, F. J. & Stanley, E. R. CSF-1 regulation of the wandering macrophage: complexity in action. Trends Cell Biol. 14, 628–638 (2004).
Stanley, E. R., Chen, D. M. & Lin, H. S. Induction of macrophage production and proliferation by a purified colony stimulating factor. Nature 274, 168–170 (1978).
Inaba, K. et al. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J. Exp. Med. 176, 1693–1702 (1992).
Hashimoto, S., Suzuki, T., Dong, H. Y., Yamazaki, N. & Matsushima, K. Serial analysis of gene expression in human monocytes and macrophages. Blood 94, 837–844 (1999).
Falk, L. A. & Vogel, S. N. Comparison of bone marrow progenitors responsive to granulocyte–macrophage colony stimulating factor and macrophage colony stimulating factor-1. J. Leukocyte Biol. 43, 148–157 (1988).
Hamilton, J. A. Colony stimulating factors, cytokines and monocyte–macrophages — some controversies. Immunol. Today 14, 18–24 (1993).
Mantovani, A., Sozzani, S., Locati, M., Allavena, P. & Sica, A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 23, 549–555 (2002). A review of macrophage polarization.
Hancock, G. E., Kaplan, G. & Cohn, Z. A. Keratinocyte growth regulation by the products of immune cells. J. Exp. Med. 168, 1395–1402 (1988).
Herembert, T., Gogusev, J., Zhu, D. L., Drueke, T. B. & Marche, P. Control of vascular smooth-muscle cell growth by macrophage-colony-stimulating factor. Biochem. J. 325 (Pt 1), 123–128 (1997).
Soldi, R. et al. Activation of JAK2 in human vascular endothelial cells by granulocyte–macrophage colony-stimulating factor. Blood 89, 863–872 (1997).
Ramsay, R. G. et al. Colony-stimulating factor-1 promotes clonogenic growth of normal murine colonic crypt epithelial cells in vitro. J. Interferon Cytokine Res. 24, 416–427 (2004).
Choi, J. K. et al. Signal transduction pathways of GM-CSF in neural cell lines. Neurosci. Lett. 420, 217–222 (2007).
Wang, Y., Berezovska, O. & Fedoroff, S. Expression of colony stimulating factor-1 receptor (CSF-1R) by CNS neurons in mice. J. Neurosci. Res. 57, 616–632 (1999).
Wiktor-Jedrzejczak, W. et al. Total absence of colony-stimulating factor 1 in the macrophage-deficient osteopetrotic (op/op) mouse. Proc. Natl Acad. Sci. USA 87, 4828–4832 (1990). This paper defined the defect in this osteopetrotic mouse as being due to loss of M-CSF function.
Cecchini, M. G. et al. Role of colony stimulating factor-1 in the establishment and regulation of tissue macrophages during postnatal development of the mouse. Development 120, 1357–1372 (1994).
Stanley, E. et al. Granulocyte/macrophage colony-stimulating factor-deficient mice show no major perturbation of hematopoiesis but develop a characteristic pulmonary pathology. Proc. Natl Acad. Sci. USA 91, 5592–5596 (1994). This report showed, surprisingly at the time, that GM-CSF-deficient mice have relatively normal haematopoiesis with the main phenotype being pulmonary alveolar proteinosis.
Bezbradica, J. S. et al. Granulocyte–macrophage colony-stimulating factor regulates effector differentiation of invariant natural killer T cells during thymic ontogeny. Immunity 25, 487–497 (2006).
Antman, K. S. et al. Effect of recombinant human granulocyte–macrophage colony-stimulating factor on chemotherapy-induced myelosuppression. N. Engl. J. Med. 319, 593–598 (1988).
Takahashi, T. et al. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nature Med. 5, 434–438 (1999).
Shiba, Y. et al. M-CSF accelerates neointimal formation in the early phase after vascular injury in mice: the critical role of the SDF-1–CXCR4 system. Arterioscler. Thromb. Vasc. Biol. 27, 283–289 (2007).
Armitage, J. O. Emerging applications of recombinant human granulocyte–macrophage colony-stimulating factor. Blood 92, 4491–4508 (1998).
Shortman, K. & Naik, S. H. Steady-state and inflammatory dendritic-cell development. Nature Rev. Immunol. 7, 19–30 (2007).
Menetrier-Caux, C. et al. Inhibition of the differentiation of dendritic cells from CD34+ progenitors by tumor cells: role of interleukin-6 and macrophage colony-stimulating factor. Blood 92, 4778–4791 (1998).
Willman, C. L., Stewart, C. C., Miller, V., Yi, T. L. & Tomasi, T. B. Regulation of MHC class II gene expression in macrophages by hematopoietic colony-stimulating factors (CSF). Induction by granulocyte/macrophage CSF and inhibition by CSF-1. J. Exp. Med. 170, 1559–1567 (1989).
Phillips, W. A. & Hamilton, J. A. Colony stimulating factor-1 is a negative regulator of the macrophage respiratory burst. J. Cell. Physiol. 144, 190–196 (1990).
Lari, R. et al. Macrophage lineage phenotypes and osteoclastogenesis — complexity in the control by GM-CSF and TGF-β. Bone 40, 323–336 (2007).
De, A. K., Laudanski, K. & Miller-Graziano, C. L. Failure of monocytes of trauma patients to convert to immature dendritic cells is related to preferential macrophage-colony-stimulating factor-driven macrophage differentiation. J. Immunol. 170, 6355–6362 (2003).
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Guidez, F., Li, A. C., Horvai, A., Welch, J. S. & Glass, C. K. Differential utilization of Ras signaling pathways by macrophage colony-stimulating factor (CSF) and granulocyte–macrophage CSF receptors during macrophage differentiation. Mol. Cell Biol. 18, 3851–3861 (1998).
Firestein, G. S. & Zvaifler, N. J. How important are T cells in chronic rheumatoid synovitis? Arthritis Rheum. 33, 768–773 (1990).
Hamilton, J. A., Campbell, I. K., Wojta, J. & Cheung, D. Plasminogen activators and their inhibitors in arthritic disease. Ann. NY Acad. Sci. 667, 87–100 (1992).
Bresnihan, B. et al. Synovial macrophages as a biomarker of response to therapeutic intervention in rheumatoid arthritis: standardization and consistency across centers. J. Rheumatol. 34, 620–622 (2007).
Leizer, T., Cebon, J., Layton, J. E. & Hamilton, J. A. Cytokine regulation of colony-stimulating factor production in cultured human synovial fibroblasts: I. Induction of GM-CSF and G-CSF production by interleukin-1 and tumor necrosis factor. Blood 76, 1989–1996 (1990).
Hamilton, J. A., Filonzi, E. L. & Ianches, G. Regulation of macrophage colony-stimulating factor (M-CSF) production in cultured human synovial fibroblasts. Growth Factors 9, 157–165 (1993).
Campbell, I. K., Novak, U., Cebon, J., Layton, J. E. & Hamilton, J. A. Human articular cartilage and chondrocytes produce hemopoietic colony-stimulating factors in culture in response to IL-1. J. Immunol. 147, 1238–1246 (1991).
Campbell, I. K., Ianches, G. & Hamilton, J. A. Production of macrophage colony-stimulating factor (M-CSF) by human articular cartilage and chondrocytes. Modulation by interleukin-1 and tumor necrosis factor-α. Biochim. Biophys. Acta 1182, 57–63 (1993).
Nakano, K. et al. Rheumatoid synovial endothelial cells produce macrophage colony-stimulating factor leading to osteoclastogenesis in rheumatoid arthritis. Rheumatology (Oxford) 46, 597–603 (2007).
Niki, Y. et al. Administration of cyclooxygenase-2 inhibitor reduces joint inflammation but exacerbates osteopenia in IL-1α transgenic mice due to GM-CSF overproduction. J. Immunol. 179, 639–646 (2007).
Campbell, I. K., Bendele, A., Smith, D. A. & Hamilton, J. A. Granulocyte–macrophage colony stimulating factor exacerbates collagen induced arthritis in mice. Ann. Rheum. Dis. 56, 364–368 (1997).
Bischof, R. J., Zafiropoulos, D., Hamilton, J. A. & Campbell, I. K. Exacerbation of acute inflammatory arthritis by the colony-stimulating factors CSF-1 and granulocyte macrophage (GM)-CSF: evidence of macrophage infiltration and local proliferation. Clin. Exp. Immunol. 119, 361–367 (2000).
Abd, A. H., Savage, N. W., Halliday, W. J. & Hume, D. A. The role of macrophages in experimental arthritis induced by Streptococcus agalactiae sonicate: actions of macrophage colony-stimulating factor (CSF-1) and other macrophage-modulating agents. Lymphokine Cytokine Res. 10, 43–50 (1991).
Campbell, I. K., Rich, M. J., Bischof, R. J. & Hamilton, J. A. The colony-stimulating factors and collagen-induced arthritis: exacerbation of disease by M-CSF and G-CSF and requirement for endogenous M-CSF. J. Leukocyte Biol. 68, 144–150 (2000). This was the first paper to show that neutralization of M-CSF suppressed an inflammatory and autoimmune disease.
Hazenberg, B. P., Van Leeuwen, M. A., Van Rijswijk, M. H., Stern, A. C. & Vellenga, E. Correction of granulocytopenia in Felty's syndrome by granulocyte–macrophage colony-stimulating factor. Simultaneous induction of interleukin-6 release and flare-up of the arthritis. Blood 74, 2769–2770 (1989).
Campbell, I. K. et al. Protection from collagen-induced arthritis in granulocyte–macrophage colony-stimulating factor-deficient mice. J. Immunol. 161, 3639–3644 (1998). This paper showed for the first time that collagen-induced arthritis could be induced significantly in C57BL/6 mice, enabling the authors to show that GM-CSF-deficient mice had almost no disease.
Cook, A. D., Braine, E. L., Campbell, I. K., Rich, M. J. & Hamilton, J. A. Blockade of collagen-induced arthritis post-onset by antibody to granulocyte–macrophage colony-stimulating factor (GM-CSF): requirement for GM-CSF in the effector phase of disease. Arthritis Res. 3, 293–298 (2001).
Ohno, H. et al. The orally-active and selective c-Fms tyrosine kinase inhibitor Ki20227 inhibits disease progression in a collagen-induced arthritis mouse model. Eur. J. Immunol. 38, 283–291 (2008).
Ando, W. et al. Imatinib mesylate inhibits osteoclastogenesis and joint destruction in rats with collagen-induced arthritis (CIA). J. Bone Miner. Metab. 24, 274–282 (2006).
Kitaura, H. et al. M-CSF mediates TNF-induced inflammatory osteolysis. J. Clin. Invest. 115, 3418–3427 (2005).
Yang, Y. H. & Hamilton, J. A. Dependence of interleukin-1-induced arthritis on granulocyte–macrophage colony-stimulating factor. Arthritis Rheum. 44, 111–119 (2001).
Plater-Zyberk, C. et al. GM-CSF neutralisation suppresses inflammation and protects cartilage in acute streptococcal cell wall arthritis of mice. Ann. Rheum. Dis. 66, 452–457 (2007).
Lawlor, K. E., Wong, P. K., Campbell, I. K., van Rooijen, N. & Wicks, I. P. Acute CD4+ T lymphocyte-dependent interleukin-1-driven arthritis selectively requires interleukin-2 and interleukin-4, joint macrophages, granulocyte–macrophage colony-stimulating factor, interleukin-6, and leukemia inhibitory factor. Arthritis Rheum. 52, 3749–3754 (2005).
Yao, Z. et al. Tumor necrosis factor-α increases circulating osteoclast precursor numbers by promoting their proliferation and differentiation in the bone marrow through up-regulation of c-Fms expression. J. Biol. Chem. 281, 11846–11855 (2006).
McQualter, J. L. et al. Granulocyte macrophage colony-stimulating factor: a new putative therapeutic target in multiple sclerosis. J. Exp. Med. 194, 873–882 (2001).
Fischer, H. G., Bielinsky, A. K., Nitzgen, B., Daubener, W. & Hadding, U. Functional dichotomy of mouse microglia developed in vitro: differential effects of macrophage and granulocyte/macrophage colony-stimulating factor on cytokine secretion and antitoxoplasmic activity. J. Neuroimmunol. 45, 193–201 (1993).
Marusic, S. et al. Local delivery of granulocyte macrophage colony-stimulating factor by retrovirally transduced antigen-specific T cells leads to severe, chronic experimental autoimmune encephalomyelitis in mice. Neurosci. Lett. 332, 185–189 (2002).
Ponomarev, E. D. et al. GM-CSF production by autoreactive T cells is required for the activation of microglial cells and the onset of experimental autoimmune encephalomyelitis. J. Immunol. 178, 39–48 (2007).
Schneider, U. C. et al. Granulocyte–macrophage colony-stimulating factor-induced vessel growth restores cerebral blood supply after bilateral carotid artery occlusion. Stroke 38, 1320–1328 (2007).
Hao, A. J., Dheen, S. T. & Ling, E. A. Expression of macrophage colony-stimulating factor and its receptor in microglia activation is linked to teratogen-induced neuronal damage. Neuroscience 112, 889–900 (2002).
Vincent, V. A., Robinson, C. C., Simsek, D. & Murphy, G. M. Macrophage colony stimulating factor prevents NMDA-induced neuronal death in hippocampal organotypic cultures. J. Neurochem. 82, 1388–1397 (2002).
Kitching, A. R. et al. The requirement for granulocyte–macrophage colony-stimulating factor and granulocyte colony-stimulating factor in leukocyte-mediated immune glomerular injury. J. Am. Soc. Nephrol. 13, 350–358 (2002).
Timoshanko, J. R., Kitching, A. R., Semple, T. J., Holdsworth, S. R. & Tipping, P. G. Granulocyte macrophage colony-stimulating factor expression by both renal parenchymal and immune cells mediates murine crescentic glomerulonephritis. J. Am. Soc. Nephrol. 16, 2646–2656 (2005).
Lenda, D. M., Kikawada, E., Stanley, E. R. & Kelley, V. R. Reduced macrophage recruitment, proliferation, and activation in colony-stimulating factor-1-deficient mice results in decreased tubular apoptosis during renal inflammation. J. Immunol. 170, 3254–3262 (2003).
Le Meur, Y. et al. Macrophage accumulation at a site of renal inflammation is dependent on the M-CSF/c-Fms pathway. J. Leukocyte Biol. 72, 530–537 (2002).
Bozinovski, S., Jones, J. E., Vlahos, R., Hamilton, J. A. & Anderson, G. P. Granulocyte/macrophage-colony-stimulating factor (GM-CSF) regulates lung innate immunity to lipopolysaccharide through Akt/Erk activation of NFκB and AP-1 in vivo. J. Biol. Chem. 277, 42808–42814 (2002).
Bozinovski, S. et al. Innate immune responses to LPS in mouse lung are suppressed and reversed by neutralization of GM-CSF via repression of TLR-4. Am. J. Physiol. Lung Cell. Mol. Physiol. 286, L877–L885 (2004).
Cates, E. C. et al. Intranasal exposure of mice to house dust mite elicits allergic airway inflammation via a GM-CSF-mediated mechanism. J. Immunol. 173, 6384–6392 (2004).
Vlahos, R., Bozinovski, S., Hamilton, J. A. & Anderson, G. P. Therapeutic potential of treating chronic obstructive pulmonary disease (COPD) by neutralising granulocyte macrophage colony-stimulating factor (GM-CSF). Pharmacol. Ther. 112, 106–115 (2006).
Yamashita, N. et al. Attenuation of airway hyperresponsiveness in a murine asthma model by neutralization of granulocyte–macrophage colony-stimulating factor (GM-CSF). Cell. Immunol. 219, 92–97 (2002).
Su, Y. C., Rolph, M. S., Hansbro, N. G., Mackay, C. R. & Sewell, W. A. Granulocyte–macrophage colony-stimulating factor is required for bronchial eosinophilia in a murine model of allergic airway inflammation. J. Immunol. 180, 2600–2607 (2008).
Ishikawa, T., Aoshiba, K., Yokohori, N. & Nagai, A. Macrophage colony-stimulating factor aggravates rather than regenerates emphysematous lungs in mice. Respiration 73, 538–545 (2006).
Baran, C. P. et al. Important roles for macrophage colony-stimulating factor, CC chemokine ligand 2, and mononuclear phagocytes in the pathogenesis of pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 176, 78–89 (2007).
Haghighat, A., Weiss, D., Whalin, M. K., Cowan, D. P. & Taylor, W. R. Granulocyte colony-stimulating factor and granulocyte macrophage colony-stimulating factor exacerbate atherosclerosis in apolipoprotein E-deficient mice. Circulation 115, 2049–2054 (2007).
Ditiatkovski, M., Toh, B. H. & Bobik, A. GM-CSF deficiency reduces macrophage PPAR-γ expression and aggravates atherosclerosis in ApoE-deficient mice. Arterioscler. Thromb. Vasc. Biol. 26, 2337–2344 (2006).
Shindo, J. et al. Granulocyte–macrophage colony-stimulating factor prevents the progression of atherosclerosis via changes in the cellular and extracellular composition of atherosclerotic lesions in watanabe heritable hyperlipidemic rabbits. Circulation 99, 2150–2156 (1999).
Shaposhnik, Z., Wang, X., Weinstein, M., Bennett, B. J. & Lusis, A. J. Granulocyte macrophage colony-stimulating factor regulates dendritic cell content of atherosclerotic lesions. Arterioscler. Thromb. Vasc. Biol. 27, 621–627 (2007).
Bergmann, C. E. et al. Arteriogenesis depends on circulating monocytes and macrophage accumulation and is severely depressed in op/op mice. J. Leukocyte Biol. 80, 59–65 (2006).
Pollard, J. W. Tumour-educated macrophages promote tumour progression and metastasis. Nature Rev. Cancer 4, 71–78 (2004). This review summarizes the role of M-CSF and tumour-associated macrophages in promoting tumour progression and metastasis.
Condeelis, J. & Pollard, J. W. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124, 263–266 (2006).
Lamagna, C., Aurrand-Lions, M. & Imhof, B. A. Dual role of macrophages in tumor growth and angiogenesis. J. Leukocyte Biol. 80, 705–713 (2006).
Young, M. R. et al. Mechanisms of immune suppression in patients with head and neck cancer: influence on the immune infiltrate of the cancer. Int. J. Cancer 67, 333–338 (1996).
Takeda, K., Hatakeyama, K., Tsuchiya, Y., Rikiishi, H. & Kumagai, K. A correlation between GM-CSF gene expression and metastases in murine tumors. Int. J. Cancer 47, 413–420 (1991).
Park, B. K. et al. NF-κB in breast cancer cells promotes osteolytic bone metastasis by inducing osteoclastogenesis via GM-CSF. Nature Med. 13, 62–69 (2007).
Paulus, P., Stanley, E. R., Schafer, R., Abraham, D. & Aharinejad, S. Colony-stimulating factor-1 antibody reverses chemoresistance in human MCF-7 breast cancer xenografts. Cancer Res. 66, 4349–4356 (2006).
Ohno, H. et al. A c-Fms tyrosine kinase inhibitor, Ki20227, suppresses osteoclast differentiation and osteolytic bone destruction in a bone metastasis model. Mol. Cancer Ther. 5, 2634–2643 (2006).
Reed, J. A. et al. GM-CSF action in the CNS decreases food intake and body weight. J. Clin. Invest. 115, 3035–3044 (2005).
Weisberg, S. P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Levine, J. A., Jensen, M. D., Eberhardt, N. L. & O'Brien, T. Adipocyte macrophage colony-stimulating factor is a mediator of adipose tissue growth. J. Clin. Invest. 101, 1557–1564 (1998).
Wei, S. et al. Modulation of CSF-1-regulated post-natal development with anti-CSF-1 antibody. Immunobiology 210, 109–119 (2005).
Gaudreau, S. et al. Granulocyte–macrophage colony-stimulating factor prevents diabetes development in NOD mice by inducing tolerogenic dendritic cells that sustain the suppressive function of CD4+CD25+ regulatory T cells. J. Immunol. 179, 3638–3647 (2007).
Watanabe, Y. et al. Effect of macrophage colony-stimulating factor on the development of diabetes mellitus in BB rats. Horm. Metab. Res. 25, 323–324 (1993).
Schon, M., Denzer, D., Kubitza, R. C., Ruzicka, T. & Schon, M. P. Critical role of neutrophils for the generation of psoriasiform skin lesions in flaky skin mice. J. Invest. Dermatol. 114, 976–983 (2000).
Keller, M. et al. T cell-regulated neutrophilic inflammation in autoinflammatory diseases. J. Immunol. 175, 7678–7686 (2005).
Kaplan, G. et al. Novel responses of human skin to intradermal recombinant granulocyte/macrophage-colony-stimulating factor: Langerhans cell recruitment, keratinocyte growth, and enhanced wound healing. J. Exp. Med. 175, 1717–1728 (1992).
Wu, L., Yu, Y. L., Galiano, R. D., Roth, S. I. & Mustoe, T. A. Macrophage colony-stimulating factor accelerates wound healing and upregulates TGF-β1 mRNA levels through tissue macrophages. J. Surg. Res. 72, 162–169 (1997).
Ginhoux, F. et al. Langerhans cells arise from monocytes in vivo. Nature Immunol. 7, 265–273 (2006).
Korzenik, J. R., Dieckgraefe, B. K., Valentine, J. F., Hausman, D. F. & Gilbert, M. J. Sargramostim for active Crohn's disease. N. Engl. J. Med. 352, 2193–2201 (2005).
Sainathan, S. K. et al. Granulocyte macrophage colony-stimulating factor ameliorates DSS-induced experimental colitis. Inflamm. Bowel Dis. 14, 88–99 (2008).
Marshall, D., Cameron, J., Lightwood, D. & Lawson, A. D. Blockade of colony stimulating factor-1 (CSF-I) leads to inhibition of DSS-induced colitis. Inflamm. Bowel Dis. 13, 219–224 (2007).
Ghia, J. E. et al. Role of M-CSF dependent macrophages in colitis is driven by the nature of the inflammatory stimulus. Am. J. Physiol. Gastrointest. Liver Physiol. 294, G770–G777 (2008).
Sweet, M. J. & Hume, D. A. CSF-1 as a regulator of macrophage activation and immune responses. Arch. Immunol. Ther. Exp. (Warsz) 51, 169–177 (2003).
McInnes, I. B. & Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nature Rev. Immunol. 7, 429–442 (2007).
Haworth, C. et al. Expression of granulocyte–macrophage colony-stimulating factor in rheumatoid arthritis: regulation by tumor necrosis factor-α. Eur. J. Immunol. 21, 2575–2579 (1991).
Niki, Y. et al. Macrophage- and neutrophil-dominant arthritis in human IL-1α transgenic mice. J. Clin. Invest. 107, 1127–1135 (2001).
Campbell, I. K., O'Donnell, K., Lawlor, K. E. & Wicks, I. P. Severe inflammatory arthritis and lymphadenopathy in the absence of TNF. J. Clin. Invest. 107, 1519–1527 (2001).
Cook, A. D., Braine, E. L. & Hamilton, J. A. Stimulus-dependent requirement for granulocyte–macrophage colony-stimulating factor in inflammation. J. Immunol. 173, 4643–4651 (2004).
Conway, J. G. et al. Inhibition of colony-stimulating-factor-1 signaling in vivo with the orally bioavailable cFMS kinase inhibitor GW2580. Proc. Natl Acad. Sci. USA 102, 16078–16083 (2005).
Seymour, J. F. Extra-pulmonary aspects of acquired pulmonary alveolar proteinosis as predicted by granulocyte–macrophage colony-stimulating factor-deficient mice. Respirology 11, S16–S22 (2006).
Robertson, S. A. GM-CSF regulation of embryo development and pregnancy. Cytokine Growth Factor Rev. 18, 287–298 (2007).
Roberts, A. W. G-CSF: a key regulator of neutrophil production, but that's not all! Growth Factors 23, 33–41 (2005).
Lieschke, G. J. et al. Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 84, 1737–1746 (1994).
Lawlor, K. E. et al. Critical role for granulocyte colony-stimulating factor in inflammatory arthritis. Proc. Natl Acad. Sci. USA 101, 11398–11403 (2004). This paper provides evidence of a role for G-CSF in models of arthritis.
Solaroglu, I., Jadhav, V. & Zhang, J. H. Neuroprotective effect of granulocyte-colony stimulating factor. Front. Biosci. 12, 712–724 (2007).
Geissmann, F., Jung, S. & Littman, D. R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 (2003).
Kawanaka, N. et al. CD14+, CD16+ blood monocytes and joint inflammation in rheumatoid arthritis. Arthritis Rheum. 46, 2578–2586 (2002).
Chapoval, A. I., Kamdar, S. J., Kremlev, S. G. & Evans, R. CSF-1 (M-CSF) differentially sensitizes mononuclear phagocyte subpopulations to endotoxin in vivo: a potential pathway that regulates the severity of Gram-negative infections. J. Leukocyte Biol. 63, 245–252 (1998).
Bruhns, P., Samuelsson, A., Pollard, J. W. & Ravetch, J. V. Colony-stimulating factor-1-dependent macrophages are responsible for IVIG protection in antibody-induced autoimmune disease. Immunity 18, 573–581 (2003).
Guan, Y. et al. Antigen presenting cells treated in vitro by macrophage colony-stimulating factor and autoantigen protect mice from autoimmunity. J. Neuroimmunol. 192, 68–78 (2007).
Hamilton, J. A. et al. Oxidized LDL can induce macrophage survival, DNA synthesis, and enhanced proliferative response to CSF-1 and GM-CSF. Arterioscler. Thromb. Vasc. Biol. 19, 98–105 (1999).
Lin, H. et al. Regulation of myeloid growth and differentiation by a novel cytokine, interleukin-34 (IL-34), via the CSF-1 receptor. Cytokine 39, 24 (2007).
Acknowledgements
I would like to thank C. Manthey, A. Nash, E. R. Stanley and A. Lopez for valuable discussions, and R. Sallay and J. Jackson for typing of the manuscript and figure preparation, respectively. I am supported by a Senior Principal Research Fellowship from the National Health and Medical Research Council of Australia. Owing to size limitations, this Review can cover only some of the CSF literature that has led to the clinical trials, and the reader will need to refer to other articles for additional information on CSF biology. My apologies are extended to the many authors whose work could not be cited.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
John Hamilton is employed by the University of Melbourne, Australia, which has licensed technology in this area to MorphoSys AG.
Related links
Glossary
- Priming
-
Optimal macrophage activation often requires exposure to at least two signals, such as priming by a cytokine (for example, IFNγ) and further stimulation or amplification by a microbial product (for example, LPS).
- Macrophage polarization
-
Macrophages respond to diverse microenvironmental signals and as a result have been classified as being polarized into a spectrum of M1 to M2 phenotypes. They are classically activated towards the M1 phenotype by microbial products or IFNγ and can thereby eradicate invading organisms and promote type I immune responses; alternative activation by stimulation with IL-4, IL-13 or IL-10 drives macrophages towards the M2 phenotype, which is characterized by hyporesponsiveness to pro-inflammatory stimuli and involvement in debris scavenging, angiogenesis, tissue remodelling, wound healing and the promotion of type II immunity. There is evidence that tumour-associated macrophages are of the M2 type.
- Osteoclasts
-
Multinucleated cells that mediate bone resorption or osteolysis.
- Pulmonary alveolar proteinosis
-
A rare lung disease in which abnormal accumulation of surfactant occurs, which interferes with gas exchange.
- Adjuvant
-
An agent mixed with an antigen that increases the immune response to that antigen after immunization.
- Respiratory burst
-
A large increase in oxygen consumption and the generation of reactive oxygen species that accompanies the exposure of neutrophils, for example, to microorganisms and/or inflammatory mediators.
- Insulin resistance
-
A condition in which normal amounts of insulin are inadequate to produce a normal insulin response in fat, muscle and liver cells. Various disease states make tissues more resistant to the effects of insulin, such as infection (mediated by TNF).
- Felty's syndrome
-
A condition with the features of rheumatoid arthritis, splenomegaly and granulocytopaenia.
- Collagen-induced arthritis
-
An experimental model of rheumatoid arthritis. The arthritis is induced by immunization of susceptible animals with type II collagen, which is the collagen present in cartilage.
- Osteoclastogenesis
-
A process whereby haematopoietic precursor cells differentiate into osteoclasts.
- Microglia
-
Macrophage-lineage cells that are present in the central nervous system.
- Experimental autoimmune encephalomyelitis
-
(EAE). An animal model of multiple sclerosis. EAE can be induced in several mammalian species by immunization with myelin-derived antigens together with adjuvant.
- Glomerulonephritis
-
An inflammation of the kidney glomeruli that can result in destruction of the glomeruli and renal failure.
- MRL–lpr mouse
-
A mouse strain that spontaneously develops glomerulonephritis and other symptoms of systemic lupus erythematosus (SLE). The lpr mutation causes a defect in CD95 (also known as FAS), which prevents the apoptosis of activated lymphocytes; the MRL strain contributes disease-associated mutations that have yet to be completely identified.
- Chronic obstructive pulmonary disease
-
(COPD). A group of lung diseases in which airflow is limited and there is airway inflammation and destruction of lung tissue.
- Arteriogenesis
-
The formation of medium-sized blood vessels.
- Trophoblast
-
A cell of the outer layer of the mammalian blastocyst that gives rise to the embryonic portion of the placenta.
Rights and permissions
About this article
Cite this article
Hamilton, J. Colony-stimulating factors in inflammation and autoimmunity. Nat Rev Immunol 8, 533–544 (2008). https://doi.org/10.1038/nri2356
Issue Date:
DOI: https://doi.org/10.1038/nri2356
This article is cited by
-
M2 macrophages-derived exosomes regulate osteoclast differentiation by the CSF2/TNF-α axis
BMC Oral Health (2024)
-
Granulocyte macrophage colony stimulating factor exerts dominant effects over macrophage colony stimulating factor during macrophage differentiation in vitro to induce an inflammatory phenotype
Inflammation Research (2024)
-
Macrophage re-programming by JAK inhibitors relies on MAFB
Cellular and Molecular Life Sciences (2024)
-
JAK-STAT signaling in inflammation and stress-related diseases: implications for therapeutic interventions
Molecular Biomedicine (2023)
-
Serum cytokine pattern in children with hemophagocytic lymphohistiocytosis
Annals of Hematology (2023)