Key Points
-
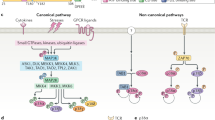
The mitogen-activated protein kinase (MAPK)-activated protein kinase (MAPKAP or MK) subfamily of protein kinases consists of the three structurally related enzymes MK2, MK3 and MK5. Besides the catalytic domain of the Ca2+/calmodulin-dependent protein kinase (CaMK) type, MKs contain a C-terminal region that includes nuclear localization signal (NLS)-, nuclear export signal (NES)- and MAPK-docking sites, and might also contain autoinhibitory and proline-rich elements.
-
MKs are activated by Thr phosphorylation at the activation loop and by at least one other Thr phosphorylation in a hinge region between the catalytic domain and the C terminus of MK2 and MK3. This latter phosphorylation leads to unmasking of the NES and activation-coupled nuclear export of MK2 and MK3. MK5 is activated by phosphorylation of a single site in the activation loop after cytoplasmic anchoring by protein kinases, such as exrtacellular signal-regulated kinase-3 (ERK3).
-
MKs are involved in actin remodelling, cell migration, development, cell-cycle regulation, chromatin remodelling and post-transcriptional control of mRNA stability and translation. More than 20 MK substrates have been identified.
-
MK2 and MK3 interact with components of the polycomb transcription-repressive complex and might modulate gene silencing through phosphorylation of the members of the complex. MK2 can function as an ultraviolet light (UV)-induced kinase for cell-cycle-checkpoint control through CDC25B and CDC25C, and can also downregulate p53 by phosphorylating a specific ubiquitin ligase.
-
The control of mRNA stability and translation by MK2 is dependent on AU-rich elements in the 3′ untranslated mRNA region, and on RNA-binding proteins. Deletion of MK2 leads to an impaired inflammatory response, which is mainly due to reduced TNF-mRNA stability or translation.
-
MK5 shows sustained activation in development, which is dependent on the cytoplasmic protein levels of its activator, ERK3, which is mainly regulated by protein-stability mechanisms. The ERK3–MK5 signalling module is of physiological relevance for embryonic development in mice.
-
A molecular mechanism of MK action is the generation of 14-3-3-protein-binding sites in some of its targets. For MK substrates — such as tristetraprolin, small heat-shock protein HSP27, CDC25B, CDC25C and tuberin — binding to 14-3-3 proteins after phosphorylation by MKs could be essential for the regulation of their function.
Abstract
Downstream of mitogen-activated protein kinases (MAPKs), three structurally related MAPK-activated protein kinases (MAPKAPKs or MKs) — MK2, MK3 and MK5 — signal to diverse cellular targets. Although there is no known common function for all three MKs, these kinases are involved in important processes: MKs regulate gene expression at the transcriptional and post-transcriptional level, control cytoskeletal architecture and cell-cycle progression, and are implicated in inflammation and cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Manning, G., Whyte, D. B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934 (2002).
Caenepeel, S., Charydczak, G., Sudarsanam, S., Hunter, T. & Manning, G. The mouse kinome: discovery and comparative genomics of all mouse protein kinases. Proc. Natl Acad. Sci. USA 101, 11707–11712 (2004).
Manning, G., Plowman, G. D., Hunter, T. & Sudarsanam, S. Evolution of protein kinase signaling from yeast to man. Trends Biochem. Sci. 27, 514–520 (2002).
Soloaga, A. et al. MSK2 and MSK1 mediate the mitogen- and stress-induced phosphorylation of histone H3 and HMG-14. EMBO J. 22, 2788–2797 (2003).
Waskiewicz, A. J., Flynn, A., Proud, C. G. & Cooper, J. A. Mitogen-activated protein kinases activate the serine/threonine kinases Mnk1 and Mnk2. EMBO J. 16, 1909–1920 (1997).
Fukunaga, R. & Hunter, T. MNK1, a new MAP kinase-activated protein kinase, isolated by a novel expression screening method for identifying protein kinase substrates. EMBO J. 16, 1921–1933 (1997).
Winzen, R. et al. The p38 MAP kinase pathway signals for cytokine-induced mRNA stabilization via MAP kinase-activated protein kinase 2 and an AU-rich region-targeted mechanism. EMBO J. 18, 4969–4980 (1999). First evidence that MK2 stabilizes specific cytokine mRNAs by an ARE-dependent mechanism.
Kotlyarov, A. et al. MAPKAP kinase 2 is essential for LPS-induced TNF-α biosynthesis. Nature Cell Biol. 1, 94–97 (1999). The mouse knockout of MK2 clearly demonstrated that MK2 is the main target of p38 that is involved in post-translational regulation of cytokine expression.
McCormick, C. & Ganem, D. The kaposin B protein of KSHV activates the p38/MK2 pathway and stabilizes cytokine mRNAs. Science 307, 739–741 (2005).
Chrestensen, C. A. et al. MAPKAP kinase 2 phosphorylates tristetraprolin on in vivo sites including Ser178, a site required for 14-3-3 binding. J. Biol. Chem. 279, 10176–10184 (2004).
Stoecklin, G. et al. MK2-induced tristetraprolin:14-3-3 complexes prevent stress granule association and ARE-mRNA decay. EMBO J. 23, 1313–1324 (2004).
Abraham, R. T. MAPKAP kinase-2: three's company at the G(2) checkpoint. Mol. Cell. 17, 163–164 (2005).
Manke, I. A. et al. MAPKAP kinase-2 is a cell cycle checkpoint kinase that regulates the G2/M transition and S phase progression in response to UV irradiation. Mol. Cell. 17, 37–48 (2005). Identification of MK2 as a cell-cycle checkpoint kinase. This observation shows how UV-induced p38 activation inhibits mitotic entry in eukaryotic cells.
Weber, H. O. et al. HDM2 phosphorylation by MAPKAP kinase 2. Oncogene 24, 1965–1972 (2005).
Bettencourt-Dias, M. et al. Genome-wide survey of protein kinases required for cell cycle progression. Nature 432, 980–987 (2004).
Voncken, J. W. et al. MAPKAP kinase 3pK phosphorylates and regulates chromatin association of the polycomb group protein Bmi1. J. Biol. Chem. 280, 5178–5187 (2005). Establishes a link between MKs, transcriptional repression and chromatin remodelling.
Schumacher, S. et al. Scaffolding by ERK3 regulates MK5 in development. EMBO J. 23, 4770–4779 (2004). References 17 and 18 show the identification of MK5 as a target of ERK3 and the role of the ERK3–MK5 signalling module in development.
Seternes, O. M. et al. Activation of MK5/PRAK by the atypical MAP kinase ERK3 defines a novel signal transduction pathway. EMBO J. 23, 4780–4791 (2004).
Stokoe, D. et al. MAPKAP kinase-2: a novel protein kinase activated by mitogen-activated protein kinase. EMBO J. 11, 3985–3994 (1992). Purification and biochemical characterization of the first MK as a protein kinase that can be activated by a MAPK in vitro . Identification of the main regulatory phosphorylation site in MK2.
Stokoe, D., Caudwell, B., Cohen, P. T. & Cohen, P. The substrate specificity and structure of mitogen-activated protein (MAP) kinase-activated protein kinase-2. Biochem. J. 296, 843–849 (1993).
Engel, K., Plath, K. & Gaestel, M. The MAP kinase-activated protein kinase 2 contains a proline-rich SH3-binding domain. FEBS Lett. 336, 143–147 (1993).
Freshney, N. W. et al. Interleukin-1 activates a novel protein kinase cascade that results in the phosphorylation of Hsp27. Cell 78, 1039–1049 (1994). Describes the purification and characterization of the kinase that activates MK2 as a new MAPK (later designated p38). It also shows that this kinase cascade is activated by the cytokine IL-1.
Rouse, J. et al. A novel kinase cascade triggered by stress and heat shock that stimulates MAPKAP kinase-2 and phosphorylation of the small heat shock proteins. Cell 78, 1027–1037 (1994). Description and characterization of the kinase cascade that activates MK2 as a new stress-activated signal transduction pathway in vertebrates, which is distinct from the classic MAPK cascade.
McLaughlin, M. M. et al. Identification of mitogen-activated protein (MAP) kinase-activated protein kinase-3, a novel substrate of CSBP p38 MAP kinase. J. Biol. Chem. 271, 8488–8492 (1996).
Sithanandam, G. et al. 3pK, a new mitogen-activated protein kinase-activated protein kinase located in the small cell lung cancer tumor suppressor gene region. Mol. Cell. Biol. 16, 868–876 (1996).
New, L. et al. PRAK, a novel protein kinase regulated by the p38 MAP kinase. EMBO J. 17, 3372–3384 (1998).
Ni, H., Wang, X. S., Diener, K. & Yao, Z. MAPKAPK5, a novel mitogen-activated protein kinase (MAPK)-activated protein kinase, is a substrate of the extracellular-regulated kinase (ERK) and p38 kinase. Biochem. Biophys. Res. Commun. 243, 492–496 (1998).
Komatsu, S. et al. Identification of MAPKAPK homolog (MAPKAPK-4) as a myosin II regulatory light-chain kinase in sea urchin egg extracts. Arch. Biochem. Biophys. 343, 55–62 (1997).
Dahlkvist, A., Kanter-Smoler, G. & Sunnerhagen, P. The RCK1 and RCK2 protein kinase genes from Saccharomyces cerevisiae suppress cell cycle checkpoint mutations in Schizosaccharomyces pombe. Mol. Gen. Genet. 246, 316–326 (1995).
Asp, E. & Sunnerhagen, P. Mkp1 and Mkp2, two MAPKAP-kinase homologues in Schizosaccharomyces pombe, interact with the MAP kinase Sty1. Mol. Genet. Genom. 268, 585–597 (2003).
Plath, K., Engel, K., Schwedersky, G. & Gaestel, M. Characterization of the proline-rich region of mouse MAPKAP kinase 2: influence on catalytic properties and binding to the c-abl-SH3 domain in vitro. Biochem. Biophys. Res. Commun. 203, 1188–1194 (1994).
Zu, Y. L., Ai, Y. & Huang, C. K. Characterization of an autoinhibitory domain in human mitogen-activated protein kinase-activated protein kinase 2. J. Biol. Chem. 270, 202–206 (1995).
Engel, K. et al. Constitutive activation of mitogen-activated protein kinase-activated protein kinase 2 by mutation of phosphorylation sites and an A-helix motif. J. Biol. Chem. 270, 27213–27221 (1995).
Tanoue, T., Adachi, M., Moriguchi, T. & Nishida, E. A conserved docking motif in MAP kinases common to substrates, activators and regulators. Nature Cell Biol. 2, 110–116 (2000).
Lukas, S. M. et al. Catalysis and function of the p38α·MK2a signaling complex. Biochemistry 43, 9950–9960 (2004).
Cano, E., Hazzalin, C. A. & Mahadevan, L. C. Anisomycin-activated protein kinases p45 and p55 but not mitogen-activated protein kinases ERK-1 and -2 are implicated in the induction of c-fos and c-jun. Mol. Cell. Biol. 14, 7352–7362 (1994).
Chevalier, D. & Allen, B. G. Two distinct forms of MAPKAP kinase-2 in adult cardiac ventricular myocytes. Biochemistry 39, 6145–6156 (2000).
Zu, Y. L. et al. The primary structure of a human MAP kinase activated protein kinase 2. Biochem. Biophys. Res. Commun. 200, 1118–1124 (1994).
Ludwig, S. et al. 3pK, a novel mitogen-activated protein (MAP) kinase-activated protein kinase, is targeted by three MAP kinase pathways. Mol. Cell. Biol. 16, 6687–6697 (1996).
Ben-Levy, R. et al. Identification of novel phosphorylation sites required for activation of MAPKAP kinase-2. EMBO J. 14, 5920–5930 (1995).
Lee, J. C. et al. A protein kinase involved in the regulation of inflammatory cytokine biosynthesis. Nature 372, 739–746 (1994). Identification of the p38 MAPKs as targets of the anti-inflammatory small-molecule-inhibitor class that is represented by SB203580, and explanation of the mechanism of action of these compounds. Evidence for post-transcriptional regulation of cytokine biosynthesis by p38.
Clifton, A. D., Young, P. R. & Cohen, P. A comparison of the substrate specificity of MAPKAP kinase-2 and MAPKAP kinase-3 and their activation by cytokines and cellular stress. FEBS Lett. 392, 209–214 (1996).
Allen, M. et al. Deficiency of the stress kinase p38α results in embryonic lethality: characterization of the kinase dependence of stress responses of enzyme-deficient embryonic stem cells. J. Exp. Med. 191, 859–870 (2000).
Ben-Levy, R., Hooper, S., Wilson, R., Paterson, H. F. & Marshall, C. J. Nuclear export of the stress-activated protein kinase p38 mediated by its substrate MAPKAP kinase-2. Curr. Biol. 8, 1049–1057 (1998). Detection of activation-dependent nuclear export of the MK2–p38 MAPK complex.
Kotlyarov, A. et al. Distinct cellular functions of MK2. Mol. Cell. Biol. 22, 4827–4835 (2002).
Sudo, T., Kawai, K., Matsuzaki, H. & Osada, H. p38 mitogen-activated protein kinase plays a key role in regulating MAPKAPK2 expression. Biochem. Biophys. Res. Commun. 337, 415–421 (2005).
Neufeld, B. et al. Serine/threonine kinases 3pK and MAPK-activated protein kinase 2 interact with the basic helix–loop–helix transcription factor E47 and repress its transcriptional activity. J. Biol. Chem. 275, 20239–20242 (2000).
Engel, K., Kotlyarov, A. & Gaestel, M. Leptomycin B-sensitive nuclear export of MAPKAP kinase 2 is regulated by phosphorylation. EMBO J. 17, 3363–3371 (1998). Demonstration of coupled phosphorylation-dependent activation and cytoplasmic translocation of MK2.
New, L., Jiang, Y. & Han, J. Regulation of PRAK subcellular location by p38 MAP kinases. Mol. Biol. Cell 14, 2603–2616 (2003).
Tanoue, T., Maeda, R., Adachi, M. & Nishida, E. Identification of a docking groove on ERK and p38 MAP kinases that regulates the specificity of docking interactions. EMBO J. 20, 466–479 (2001).
Seternes, O. M. et al. Both binding and activation of p38 mitogen-activated protein kinase (MAPK) play essential roles in regulation of the nucleocytoplasmic distribution of MAPK-activated protein kinase 5 by cellular stress. Mol. Cell. Biol. 22, 6931–6945 (2002).
Meng, W. et al. Structure of mitogen-activated protein kinase-activated protein (MAPKAP) kinase 2 suggests a bifunctional switch that couples kinase activation with nuclear export. J. Biol. Chem. 277, 37401–37405 (2002). Crystal structure of MK2 in its inactive non-phosphorylated form. Structural evidence that the C-terminal NES is masked by interaction with the catalytic domain and that the C terminus interferes with the activation loop and the substrate binding site of the kinase.
Underwood, K. W. et al. Catalytically active MAP KAP kinase 2 structures in complex with staurosporine and ADP reveal differences with the autoinhibited enzyme. Structure 11, 627–636 (2003).
Neininger, A., Thielemann, H. & Gaestel, M. FRET-based detection of different conformations of MK2. EMBO Rep. 2, 703–708 (2001).
Shi, Y. et al. Elimination of protein kinase MK5/PRAK activity by targeted homologous recombination. Mol. Cell. Biol. 23, 7732–7741 (2003).
Coulombe, P., Rodier, G., Pelletier, S., Pellerin, J. & Meloche, S. Rapid turnover of extracellular signal-regulated kinase 3 by the ubiquitin–proteasome pathway defines a novel paradigm of mitogen-activated protein kinase regulation during cellular differentiation. Mol. Cell. Biol. 23, 4542–4558 (2003).
Julien, C. et al. Nuclear export of ERK3 by a CRM1-dependent mechanism regulates its inhibitory action on cell cycle progression. J. Biol. Chem. 278, 42615–42624 (2003).
Baas, A. F. et al. Activation of the tumour suppressor kinase LKB1 by the STE20-like pseudokinase STRAD. EMBO J. 22, 3062–3072 (2003).
Stokoe, D., Engel, K., Campbell, D. G., Cohen, P. & Gaestel, M. Identification of MAPKAP kinase 2 as a major enzyme responsible for the phosphorylation of the small mammalian heat shock proteins. FEBS Lett. 313, 307–313 (1992).
Thomas, G., Haavik, J. & Cohen, P. Participation of a stress-activated protein kinase cascade in the activation of tyrosine hydroxylase in chromaffin cells. Eur. J. Biochem. 247, 1180–1189 (1997).
Werz, O., Klemm, J., Samuelsson, B. & Radmark, O. 5-lipoxygenase is phosphorylated by p38 kinase-dependent MAPKAP kinases. Proc. Natl Acad. Sci. USA 97, 5261–5266 (2000).
Kato, K. et al. Phosphorylation of αB-crystallin in mitotic cells and identification of enzymatic activities responsible for phosphorylation. J. Biol. Chem. 273, 28346–28354 (1998).
Cheng, T. J. & Lai, Y. K. Identification of mitogen-activated protein kinase-activated protein kinase-2 as a vimentin kinase activated by okadaic acid in 9L rat brain tumor cells. J. Cell Biochem. 71, 169–181 (1998).
Rousseau, S. et al. Inhibition of SAPK2a/p38 prevents hnRNP A0 phosphorylation by MAPKAP-K2 and its interaction with cytokine mRNAs. EMBO J. 21, 6505–6514 (2002).
Bollig, F. et al. Affinity purification of ARE-binding proteins identifies polyA-binding protein 1 as a potential substrate in MK2-induced mRNA stabilization. Biochem. Biophys. Res. Commun. 301, 665–670 (2003).
Heidenreich, O. et al. MAPKAP kinase 2 phosphorylates serum response factor in vitro and in vivo. J. Biol. Chem. 274, 14434–14443 (1999).
Janknecht, R. Cell type-specific inhibition of the ETS transcription factor ER81 by mitogen-activated protein kinase-activated protein kinase 2. J. Biol. Chem. 276, 41856–41861 (2001).
Ueda, K., Kosako, H., Fukui, Y. & Hattori, S. Proteomic identification of Bcl2-associated athanogene 2 as a novel MAPK-activated protein kinase 2 substrate. J. Biol. Chem. 279, 41815–41821 (2004).
Rogalla, T. et al. Regulation of Hsp27 oligomerization, chaperone function, and protective activity against oxidative stress/tumor necrosis factor α by phosphorylation. J. Biol. Chem. 274, 18947–18956 (1999).
Guay, J. et al. Regulation of actin filament dynamics by p38 map kinase-mediated phosphorylation of heat shock protein 27. J. Cell Sci. 110, 357–368 (1997).
Jakob, U., Gaestel, M., Engel, K. & Buchner, J. Small heat shock proteins are molecular chaperones. J. Biol. Chem. 268, 1517–1520 (1993).
Ehrnsperger, M., Graber, S., Gaestel, M. & Buchner, J. Binding of non-native protein to Hsp25 during heat shock creates a reservoir of folding intermediates for reactivation. EMBO J. 16, 221–229 (1997).
Benndorf, R. et al. Phosphorylation and supramolecular organization of murine small heat shock protein HSP25 abolish its actin polymerization-inhibiting activity. J. Biol. Chem. 269, 20780–20784 (1994).
Huang, C. K., Zhan, L., Ai, Y. & Jongstra, J. LSP1 is the major substrate for mitogen-activated protein kinase-activated protein kinase 2 in human neutrophils. J. Biol. Chem. 272, 17–19 (1997).
Eyers, C. E. et al. The phosphorylation of CapZ-interacting protein (CapZIP) by stress-activated protein kinases triggers its dissociation from CapZ. Biochem. J. 389, 127–135 (2005).
Singh, S. et al. Identification of the p16-Arc subunit of the Arp 2/3 complex as a substrate of MAPK-activated protein kinase 2 by proteomic analysis. J. Biol. Chem. 278, 36410–36417 (2003).
Hedges, J. C. et al. A role for p38(MAPK)/HSP27 pathway in smooth muscle cell migration. J. Biol. Chem. 274, 24211–24219 (1999).
Hannigan, M. O. et al. Abnormal migration phenotype of mitogen-activated protein kinase-activated protein kinase 2−/− neutrophils in Zigmond chambers containing formyl-methionyl-leucyl-phenylalanine gradients. J. Immunol. 167, 3953–3961 (2001).
Turgeon, B., Saba-El-Leil, M. K. & Meloche, S. Cloning and characterization of mouse extracellular-signal-regulated protein kinase 3 as a unique gene product of 100 kDa. Biochem. J. 346, 169–175 (2000).
Bulavin, D. V. et al. Initiation of a G2/M checkpoint after ultraviolet radiation requires p38 kinase. Nature 411, 102–107 (2001).
Lopez-Aviles, S. et al. Inactivation of the Cdc25 phosphatase by the stress-activated Srk1 kinase in fission yeast. Mol. Cell 17, 49–59 (2005).
Bulavin, D. V. et al. Phosphorylation of human p53 by p38 kinase coordinates N-terminal phosphorylation and apoptosis in response to UV radiation. EMBO J. 18, 6845–6854 (1999).
Yannoni, Y. M., Gaestel, M. & Lin, L. L. P66(ShcA) interacts with MAPKAP kinase 2 and regulates its activity. FEBS Lett. 564, 205–211 (2004).
Levine, S. S., King, I. F. & Kingston, R. E. Division of labor in polycomb group repression. Trends Biochem. Sci. 29, 478–485 (2004).
Zakowski, V., Keramas, G., Kilian, K., Rapp, U. R. & Ludwig, S. Mitogen-activated 3p kinase is active in the nucleus. Exp. Cell Res. 299, 101–109 (2004).
Lessard, J. & Sauvageau, G. Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 423, 255–260 (2003).
Lehner, M. D. et al. Mitogen-activated protein kinase-activated protein kinase 2-deficient mice show increased susceptibility to Listeria monocytogenes infection. J. Immunol. 168, 4667–4673 (2002).
Kontoyiannis, D. et al. Genetic dissection of the cellular pathways and signaling mechanisms in modeled tumor necrosis factor-induced Crohn's-like inflammatory bowel disease. J. Exp. Med. 196, 1563–1574 (2002).
Neininger, A. et al. MK2 targets AU-rich elements and regulates biosynthesis of tumor necrosis factor and interleukin-6 independently at different post-transcriptional levels. J. Biol. Chem. 277, 3065–3068 (2002).
Carballo, E., Lai, W. S. & Blackshear, P. J. Feedback inhibition of macrophage tumor necrosis factor-α production by tristetraprolin. Science 281, 1001–1005 (1998).
Han, Q. et al. Rac1–MKK3–p38–MAPKAPK2 pathway promotes urokinase plasminogen activator mRNA stability in invasive breast cancer cells. J. Biol. Chem. 277, 48379–48385 (2002).
Lasa, M. et al. Regulation of cyclooxygenase 2 mRNA stability by the mitogen-activated protein kinase p38 signaling cascade. Mol. Cell. Biol. 20, 4265–4274 (2000).
Tran, H., Maurer, F. & Nagamine, Y. Stabilization of urokinase and urokinase receptor mRNAs by HuR is linked to its cytoplasmic accumulation induced by activated mitogen-activated protein kinase-activated protein kinase 2. Mol. Cell. Biol. 23, 7177–7188 (2003).
Graziani, E. I., Ritacco, F. V., Bernan, V. S. & Telliez, J. B. Phaeochromycins A–E, anti-inflammatory polyketides isolated from the soil actinomycete Streptomyces phaeochromogenes LL-P018. J. Nat. Prod. 68, 1262–1265 (2005).
Davidson, W. et al. Discovery and characterization of a substrate selective p38α inhibitor. Biochemistry 43, 11658–11671 (2004).
Anderson, D. R. et al. Aminocyanopyridine inhibitors of mitogen activated protein kinase-activated protein kinase 2 (MK-2). Bioorg. Med. Chem. Lett. 15, 1587–1590 (2005).
Almholt, D. L. et al. Nuclear export inhibitors and kinase inhibitors identified using a MAPK-activated protein kinase 2 redistribution screen. Assay Drug Dev. Technol. 2, 7–20 (2004).
Vernier, W. F. et al. Mitogen activated protein kinase-activated protein kinase 2 inhibiting compounds. US Patent 2004/0209897A1 (2004). Description of the first small-molecule inhibitors for MK2 that show a significant effect in a rat arthritis model.
Esclatine, A., Taddeo, B. & Roizman, B. Herpes simplex virus 1 induces cytoplasmic accumulation of TIA-1/TIAR and both synthesis and cytoplasmic accumulation of tristetraprolin, two cellular proteins that bind and destabilize AU-rich RNAs. J. Virol. 78, 8582–8592 (2004).
Dreiza, C. M. et al. Transducible heat shock protein 20 (HSP20) phosphopeptide alters cytoskeletal dynamics. FASEB J. 19, 261–263 (2005).
Gringhuis, S. I., Garcia-Vallejo, J. J., van Het Hof, B. & van Dijk, W. Convergent actions of IκaB kinase β and protein kinase Cδ modulate mRNA stability through phosphorylation of 14-3-3β complexed with tristetraprolin. Mol. Cell. Biol. 25, 6454–6463 (2005).
Li, Y., Inoki, K., Vacratsis, P. & Guan, K. L. The p38 and MK2 kinase cascade phosphorylates tuberin, the tuberous sclerosis 2 gene product, and enhances its interaction with 14-3-3. J. Biol. Chem. 278, 13663–13671 (2003).
Powell, D. W. et al. Proteomic identification of 14-3-3ζ as a mitogen-activated protein kinase-activated protein kinase 2 substrate: role in dimer formation and ligand binding. Mol. Cell. Biol. 23, 5376–5387 (2003).
Buxade, M. et al. The Mnks are novel components in the control of TNFα biosynthesis and phosphorylate and regulate hnRNP A1. Immunity 23, 177–189 (2005).
Beals, C. R., Clipstone, N. A., Ho, S. N. & Crabtree, G. R. Nuclear localization of NF-ATc by a calcineurin-dependent, cyclosporin-sensitive intramolecular interaction. Genes Dev. 11, 824–834 (1997).
Briggs, L. J. et al. The cAMP-dependent protein kinase site (Ser312) enhances dorsal nuclear import through facilitating nuclear localization sequence/importin interaction. J. Biol. Chem. 273, 22745–22752 (1998).
Zink, B. & Paro, R. In vivo binding pattern of a trans-regulator of homoeotic genes in Drosophila melanogaster. Nature 337, 468–471 (1989).
van der Lugt, N. M. et al. Posterior transformation, neurological abnormalities, and severe hematopoietic defects in mice with a targeted deletion of the bmi-1 proto-oncogene. Genes Dev. 8, 757–769 (1994).
Voncken, J. W. et al. Chromatin-association of the Polycomb group protein BMI1 is cell cycle-regulated and correlates with its phosphorylation status. J. Cell Sci. 112, 4627–4639 (1999).
Valk-Lingbeek, M. E., Bruggeman, S. W. & van Lohuizen, M. Stem cells and cancer: the polycomb connection. Cell 118, 409–418 (2004).
Shaw, G. & Kamen, R. A conserved AU sequence from the 3′ untranslated region of GM-CSF mRNA mediates selective mRNA degradation. Cell 46, 659–667 (1986).
Chen, C. Y. & Shyu, A. B. AU-rich elements: characterization and importance in mRNA degradation. Trends Biochem. Sci. 20, 465–470 (1995).
Khabar, K. S. The AU-rich transcriptome: more than interferons and cytokines, and its role in disease. J. Interferon Cytokine Res. 25, 1–10 (2005).
Frevel, M. A. et al. p38 Mitogen-activated protein kinase-dependent and -independent signaling of mRNA stability of AU-rich element-containing transcripts. Mol. Cell. Biol. 23, 425–436 (2003). Use of gene array for the identification of the p38-regulated subset of ARE-containing mRNAs.
Hitti, E. et al. MAPK-activated protein kinase 2 (MK2) regulates TNF mRNA stability and translation mainly by altering tristetraprolin (TTP) expression, stability and binding to adenine/uridine-rich element. Mol. Cell. Biol. (in the press).
Brook, M. et al. Post-translational regulation of tristetraprolin subcellular localization and protein stability by p38 MAPK and ERK pathways. Mol. Cell. Biol. (in the press).
Acknowledgements
I wish to thank H. Holtmann, M. Kracht and A. Kotlyarov for critical reading and discussion of the manuscript. I apologize to all colleagues for the work I could not cite owing to space constrains. The work of my laboratory was, and is, continuously funded by the Deutsche Forschungsgemeinschaft and is also supported by the European Community.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Supplementary information
Related links
Related links
DATABASES
Saccharomyces genome database
OMIM
FURTHER INFORMATION
iHOP (information hyperlinked over proteins)
Phosphosite
The AfCS signalling gateway
Glossary
- SAPKs
-
Stress-activated protein kinases that consist of the JNK and p38 subgroups.
- Kinome
-
The protein kinase complement of the genome, which contains 518 genes and 106 pseudogenes in humans, and 540 genes and 97 pseudogenes in mice.
- Importins
-
Proteins that facilitate the nuclear import of cargo proteins. In many cases, importin-α binds to the NLS of the cargo protein, and then importin-β binds to and translocates the complex through the nuclear pore. In the nucleus, Ran·GTP displaces importin-β and the cargo protein is released.
- Exportins
-
Proteins that facilitate the nuclear export of proteins and mRNA. Exportins bind to their cargo in the nucleus together with Ran·GTP. After translocation through the nuclear pore, GTP is hydrolysed and the cargo is released.
- Src-homology-3 (SH3) domain
-
A protein domain of approximately 60 amino acids that has homology to an N-terminal region of the protein tyrosine kinase Src. There are more than 250 SH3 domains in the human proteome, which can bind to proline-rich protein regions that form a left-handed helix with the minimal consensus motif P-X-X-P (where X is any amino acid).
- Nuclear export signal
-
(NES). An amino-acid sequence that often contains several leucines or other hydrophobic residues and interacts with CRM/exportin-1, thereby functioning as a signal to facilitate export from the nucleus.
- Nuclear localization signal (NLS)
-
An amino-acid sequence that often consists of a bipartite arrangement of basic amino acids and interacts with importin-α, thereby functioning as a signal to facilitate import into the nucleus.
- Anisomycin
-
An antibiotic that inhibits protein synthesis and, at lower concentrations, stimulates stress signalling.
- Homeotic genes
-
Genes that specify how structures develop in different segments of the body during embryogenesis. Homeotic genes are often organized in clusters and encode DNA-binding proteins.
- Chaperone (molecular chaperone)
-
Proteins that assist the folding of nascent proteins, or the re-folding of partially misfolded proteins, without being part of the final folded protein structure.
- Barbed end
-
The rapidly growing end of the polar actin microfilaments at which globular actin polymerizes.
- 14-3-3 proteins
-
A family of ∼30-kDa proteins that form cup-like structures that bind to discrete phosphoserine-containing motifs. They are present in fungi, plants and mammals and, in mammals, nine structurally related family members have been identified.
- Boyden chamber
-
An experimental setting that was developed by Stephen Boyden in 1961 to test the chemotactic activity of motile cells in vitro. Two compartments are separated by a membrane of such pore size that only actively migrating cells can pass through it; a gradient of the chemotactic substance is applied, and the number of cells that pass through the membrane is determined.
- Polycomb repressive complex-1
-
(PRC1). One of the two subcomplexes (PRC1 and PRC2) that cooperate to form the nuclear polycomb repressive complex (PRC), which has a role in gene silencing during development.
- Kaposi's sarcoma
-
Cancer of the connective tissue that was named after the person who described it — the Hungarian dermatologist Moritz Kaposi (1837–1902) — and is caused by infection with the herpes virus, especially in patients with severe immunodeficiencies.
- 23-nucleotide GC-rich direct repeats
-
The kaposin locus of herpes virus contains a small coding region (open reading frame (orf) k12) preceded by two families of 23-nucleotide GC-rich direct repeats (termed DR1 and DR2). It is transcribed as a single mRNA and a complex translational programme generates various proteins from this mRNA. Kaposin B results from translation of the repeats alone and consists of a series of tandemly repeated copies of 23-amino-acid peptides that are derived from translation of the DR2 (HPRNPARRTPGTRRGAPQEPGAA) and DR1 (PGTWCPPPREPGALLPGNLVPSS) repeats.
- P-bodies
-
Cytoplasmic aggregates of translationally inactive ribonucleoproteins (mRNPs), which are sites of mRNA degradation.
Rights and permissions
About this article
Cite this article
Gaestel, M. MAPKAP kinases — MKs — two's company, three's a crowd. Nat Rev Mol Cell Biol 7, 120–130 (2006). https://doi.org/10.1038/nrm1834
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm1834
This article is cited by
-
Forward genetic screening identifies novel roles for N-terminal acetyltransferase C and histone deacetylase in C. elegans development
Scientific Reports (2022)
-
p38β - MAPK11 and its role in female cancers
Journal of Ovarian Research (2021)
-
Dexamethasone suppresses immune evasion by inducing GR/STAT3 mediated downregulation of PD-L1 and IDO1 pathways
Oncogene (2021)
-
Signaling of MK2 sustains robust AP1 activity for triple negative breast cancer tumorigenesis through direct phosphorylation of JAB1
npj Breast Cancer (2021)
-
Enhancing chemotherapy response through augmented synthetic lethality by co-targeting nucleotide excision repair and cell-cycle checkpoints
Nature Communications (2020)