Key Points
-
This review summarizes the 3′–5′ exonucleases and their possible roles in DNA-synthetic events.
-
DNA polymerases are required to maintain the integrity of the genome during processes such as DNA replication, DNA repair, translesion DNA synthesis, DNA recombination, cell-cycle control and DNA-damage-checkpoint function.
-
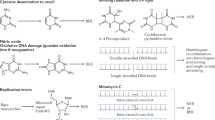
An associated 3′–5′ exonuclease activity allows a polymerase to remove misincorporated nucleotides, and ensures the high-fidelity DNA synthesis that is required for faithful replication.
-
Proofreading 3′–5′ exonucleases can be divided into intrinsic, polymerase-associated enzymes, or independent autonomous enzymes. Polymerase-associated enzymes include Escherichia coli pol I, bacteriophage polymerases, and eukaryotic pols δ, ɛ and γ. Autonomous exonucleases include the mammalian TREX1 and TREX2 enzymes.
-
As well as the proofreading 3′–5′ exonucleases, several new exonucleases have recently been discovered. These include the Werner syndrome (WRN) protein; p53; MRE11 (which is involved in double-strand break repair); two checkpoint proteins, RAD1 and RAD9; APE1 (involved in base-excision repair); and the VDJP protein, which acts in V(D)J recombination.
Abstract
Over the past few years, several new 3′–5′ exonucleases have been identified. In vitro studies of these enzymes have uncovered much about their potential functions in vivo, and certain organisms with a defect in 3′–5′ exonucleases have an increased susceptibility to cancer, especially under conditions of stress. Here, we look at not only the newly discovered enzymes, but also at the roles of other 3′–5′ exonucleases in the quality control of DNA synthesis, where they act as proofreading exonucleases for DNA polymerases during DNA replication, repair and recombination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nakamura, J. et al. Highly sensitive apurinic/apyrimidinic site assay can detect spontaneous and chemical induced depurination under physiological conditions. Cancer Res. 58, 222–225 (1998).
Lindahl, T. Instability and decay of the primary structure of DNA. Nature 362, 709–715 (1993).
Hübscher, U., Maga, G. & Spadari, S. Eukaryotic DNA polymerases. Annu. Rev. Biochem. 71, 133–163 (2002).
Jackson, A. L. & Loeb, L. A. The mutation rate and cancer. Genetics 148, 1483–1490 (1998).Here, and in reference 40 , the authors provide a mutator phenotype of a cancer cell.
Fijalkowska, I. J. & Schaaper, R. M. Mutants in the Exo I motif of Escherichia coli dnaQ: defective proofreading and inviability due to error catastrophe. Proc. Natl Acad. Sci. USA 93, 2856–2861 (1996).The first demonstration that a defective proofreading function of an autonomous 3′–5′ exonuclease leads to cell inviability in bacteria.
Morrison, A., Johnston, A. L., Johnston, L. H. & Sugino, A. Pathway correction DNA replication errors in Saccharomyces cerevisiae. EMBO J. 12, 1467–1473 (1993).This (and references 38, 39 and 101 ) describes the strong mutator phenotype and inviability owing to defective proofreading and/or mismatch repair in yeast.
Goldsby, R. E. et al. Defective DNA polymerase-δ proofreading causes cancer susceptibility in mice. Nature Med. 7, 638–639 (2001).The first demonstration that the loss of pol δ proofreading function alone can lead to cancer in mice.
Morley, A. A. The somatic mutation theory of ageing. Mutat. Res. 338, 19–23 (1995).
Derbyshire, V., Grindley, N. D. F. & Joyce, C. M. The 3′–5′ exonuclease of DNA polymerase I of Escherichia coli: contribution of each amino acid at the active site to the reaction. EMBO J. 10, 17–24 (1991).
Reha-Krantz, L. J. & Nonay, R. L. Genetic and biochemical studies of bacteriophage T4 DNA polymerase 3′–5′ exonuclease activity. Genomics 36, 449–458 (1993).
Esteban, J. A., Soengas, M. S., Salas, M. & Blanco, L. 3′–5′ exonuclease active site of ϕ29 DNA polymerase. J. Biol. Chem. 269, 31946–31954 (1994).
Bernard, A., Blanco, L., Lazaro, J. M., Martin, G. & Salas, M. A conserved 3′–5′ exonuclease active site in prokaryotic and eukaryotic DNA polymerases. Cell 59, 219–228 (1989).
Derbyshire, V., Pinsonneault, J. K. & Joyce, C. M. Structure–function analysis of 3′→5′ exonuclease of DNA polymerases. Methods Enzymol. 262, 363–385 (1995).
Shafritz, K.-M., Sandigursky, M. & Franklin, W. A. Exonuclease IX of Escherichia coli. Nucleic Acids Res. 26, 2593–2597 (1998).
Viswanathan, M. & Lovett, S. T. Exonuclease X of Escherichia coli. J. Biol. Chem. 274, 30094–30100 (1999).
Mazur, D. J. & Perrino, F. W. Identification and expression of the TREX1 and TREX2 cDNA sequences encoding mammalian 3′–5′ exonucleases. J. Biol. Chem. 274, 19655–19660 (1999).This, and reference 51 , describes the identification of the main autonomous 3′–5′ exonuclease activities in mammalian cells.
Viswanathan, M., Dower, K. W. & Lovett, S. Identification of protein DNase activity associated with RNase T of Escherichia coli. J. Biol. Chem. 273, 35126–35131 (1998).
Huang, S. et al. The premature ageing syndrome protein, WRN, is a 3′→5′ exonuclease. Nature Genet. 20, 114–116 (1998).This paper identifies a 3′–5′ exonuclease activity in the Werner syndrome (WRN) protein.
Kunkel, T. A. Exonucleolytic proofreading. Cell 53, 837–840 (1988).
Shevelev, I. V., Kravetskaya, T. P., Legina, O. K. & Krutyakov, V. M. External proofreading of DNA replication errors and mammalian autonomous 3′–5′ exonucleases. Mutat. Res. 352, 51–55 (1996).
Reddy, M. K., Weitzel, S. E. & von Hippel, P. H. Processive proofreading is intrinsic to T4 DNA polymerase. J. Biol. Chem. 267, 14157–14166 (1992).
Wang, J., Sattar, A. K. M. A., Wong, C. C., Karam, J. D. & Steitz, T. A. Crystal structure of a pol α family replication DNA polymerase from bacteriophage RB69. Cell 89, 1087–1099 (1997).
Donlin, M. J., Patel, S. S. & Johnson, K. A. Kinetic partitioning between the exonuclease and polymerase sites in DNA error correction. Biochemistry 30, 538–546 (1991).
deVega, M., Blanco, L. & Salas, M. Processive proofreading and the spatial relationship between polymerase and exonuclease active sites of bacteriophage ϕ29 DNA polymerase. J. Mol. Biol. 292, 39–51 (1999).
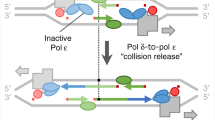
Joyce, C. M. How DNA travels between the separate polymerase and 3′–5′ exonuclease sites of DNA polymerase I (Klenow fragment). J. Biol. Chem. 264, 10858–10866 (1989).This work provides evidence for the mechanism of intermolecular proofreading.
Olson, M. W. & Kaguni, L. S. 3′–5′ exonuclease in Drosophila mitochondrial DNA polymerase. J. Biol. Chem. 267, 23136–23142 (1992).
Perrino, F. W. & Loeb, L. A. Hydrolysis of 3′-terminal mispairs in vitro by the 3′–5′ exonuclease of DNA polymerase δ permits subsequent extension by DNA polymerase α. Biochemistry 29, 5226–5231 (1990).
Perrino, F. W. & Loeb, L. A. Proofreading by the ɛ subunit of Escherichia coli DNA polymerase III increases the fidelity of calf thymus DNA polymerase α. Proc. Natl Acad. Sci. USA 86, 3085–3088 (1989).This work provides evidence that external proofreading is possible.
Shevelev, I. V., Belyakova, N. V., Kravetskaya, T. P. & Krutyakov, V. M. Autonomous 3′–5′ exonucleases can proofread for DNA polymerase β from rat liver. Mutat. Res. 459, 237–242 (2000).
Fijalkowska, I. J., Jonczyc, P., Tkaczhyk, M. M., Bialoskorska, M. & Schaaper, R. M. Unequal fidelity of leading strand and lagging strand DNA replication on the Escherichia coli chromosome. Proc. Natl Acad. Sci. USA 95, 10020–10025 (1998).
Roberts, J. D., Thomas, D. C. & Kunkel, T. A. Exonucleolytic proofreading of leading and lagging strand DNA replication errors. Proc. Natl Acad. Sci. USA 88, 3465–3469 (1991).
Mozzherin, D. J. et al. Proliferating cell nuclear antigen promotes misincorporation catalyzed by calf thymus DNA polymerase δ. J. Biol. Chem. 271, 31711–31717 (1996).
Perrino, F. W. & Loeb, L. A. Differential extension of 3′ mispairs is a major contribution to the high fidelity of calf thymus DNA polymerase α. J. Biol. Chem. 264, 2898–2905 (1989).
Kelman, Z. & O' Donnell, M. DNA polymerase III holoenzyme: structure and function of a chromosomal replicating machine. Annu. Rev. Biochem. 64, 171–200 (1995).
Ciesla, Z., Jonczyk, P. & Fijalkowska, I. J. Effect of enhanced synthesis of the ɛ subunit of DNA polymerase III on spontaneous and UV-induced mutagenesis of the Escherichia coli glyU gene. Mol. Gen. Genet. 221, 251–255 (1990).The first evidence that increasing the cellular level of an autonomous 3′–5′ exonuclease has an antimutator effect.
Hübscher, U., Nasheuer, H. P. & Syväoja, J. Eukaryotic DNA polymerases, a growing family. Trends Biochem. Sci. 25, 143–147 (2000).
Karthikeyan, R. et al. Evidence from mutational specificity studies yeast DNA polymerases δ and ɛ replicate different DNA strands at an intracellular replication fork. J. Mol. Biol. 299, 405–419 (2000).
Tran, H. T., Gordenin, D. A. & Resnick, M. A. The 3′→5′ exonucleases of DNA polymerases δ and ɛ and the 5′–3′ exonuclease Exo1 have major roles in postreplication mutation avoidance in Saccharomyces cerevisiae. Mol. Cell. Biol. 19, 2000–2007 (1999).
Vanderstraeten, S., Van den Brule, S., Hu, J. & Foury, F. The role of 3′–5′ exonucleolytic proofreading and mismatch repair in yeast mitochondrial DNA error avoidance. J. Biol. Chem. 273, 23690–23697 (1998).
Loeb, L. A. Mutator phenotype may be required for multistage carcinogenesis. Cancer Res. 51, 3075–3079 (1991).
Chen, X., Zao, S., Kelman, Z., Hurwitz, J. & Goodman, M. F. Fidelity of eukaryotic DNA polymerase δ holoenzyme from Schizosaccharomyces pombe. J. Biol. Chem. 275, 17677–17685 (2000).
Perrino, F. W., Mazur, D. J., Ward, H. & Harvey, S. Exonucleases and the incorporation of aranucleotides into DNA. Cell Biochem. Biophys. 30, 331–352 (1999).
Perrino, F. W., Miller, H. & Ealey, K. A. Identification of a 3′–5′ exonuclease that removes cytosine arabinoside monophosphate from 3′ termini of DNA. J. Biol. Chem. 269, 16357–16363 (1994).
Shevelev, I. V., Belyakova, N. V., Kravetskaya, T. P., Smirnova, E. A. & Krutyakov, V. M. High accuracy of DNA synthesis catalyzed by the complex of DNA polymerase α family in the presence of autonomous 3′–5′ exonucleases. Docl. Biochem. Biophys. (Russian) 378, 156–159 (2001).
Shevelev, I. V., Ramadan, K. & Hubscher, U. The TREX2 3′–5′ exonuclease physically interacts with DNA polymerase δ and increases its accuracy. The Scientific World Journal 2, 275–281 (2002).
Hübscher, U. & Seo, Y. S. Replication of the lagging strand: a concert of at least 23 polypeptides. Mol. Cells 12, 149–157 (2001).
Thomas, D. C. et al. Fidelity of mammalian DNA replication and replicative DNA polymerases. Biochemistry 30, 11751–11759 (1991).
Wood, R. D., Mitchell, M., Sgouros, J. & Lindahl, T. Human DNA repair genes. Science 291, 1284–1289 (2001).The authors propose a classification for human DNA repair genes according to the function of their proteins.
Lieber, M. R. The FEN-1 family of structure-specific nucleases in eukaryotic DNA replication, recombination and repair. Bioessays 19, 233–240 (1997).
Genschel, J., Bazemore, L. R. & Modrich, P. Human exonuclease I is required for 5′ and 3′ mismatch repair. J. Biol. Chem. 277, 13302–13311 (2002).
Höss, M. et al. A human DNA editing enzyme homologous to the Escherichia coli DnaQ/MutD protein. EMBO J. 18, 3868–3875 (1999).
Mazur, D. J. & Perrino, F. W. Excision of 3′ termini by the TREX1 and TREX2 3′–5′' exonucleases. J. Biol. Chem. 276, 17022–17029 (2001).
Lebel, M. Werner syndrome: genetic and molecular basis of a premature ageing disorder. Cell Mol. Life Sci. 58, 857–867 (2001).
Huang, S. et al. Characterization of the human and mouse WRN 3′–5′ exonuclease. Nucleic Acids Res. 28, 2396–2405 (2000).
Li, B. & Comai, L. Requirements for the nucleolytic processing of DNA ends by the Werner syndrome protein–Ku70/80 complex. J. Biol. Chem. 276, 9896–9902 (2001).
Vogel, H., Lim, D. S., Karsenty, G., Finegold, M. & Hasty, P. Deletion of Ku86 causes early onset of senescence in mice. Proc. Natl Acad. Sci. USA 96, 10770–10775 (1999).
Opresco, P. L., Laine, J.-P., Brosh, R. M., Seidman, M. M. & Bohr, V. A. Coordinate action of the helicase and 3′–5′ exonuclease of Werner syndrome protein. J. Biol. Chem. 276, 44677–44687 (2001).
Kamath-Loeb, A. S., Loeb, L. A., Johansson, E., Burgers, P. M. & Fry, M. Interactions between the Werner syndrome helicase and DNA polymerase δ specifically facilitate copying of tetraplex and hairpin structures of the d(CGG)n trinucleotide repeat sequence. J. Biol. Chem. 276, 16439–16446 (2001).
Brosh, R. M. J. et al. p53 modulates the exonuclease activity of Werner syndrome protein. J. Biol. Chem. 276, 35093–35102 (2001).
Lombard, D. B. et al. Mutations in the WRN gene in mice accelerate mortality in a p53-null background. Mol. Cell. Biol. 20, 3286–3291 (2000).
Janus, F. et al. The dual role for p53 in maintaining genomic integrity. Cell. Mol. Life Sci. 55, 12–27 (1999).
Harvey, M. et al. Spontaneous and carcinogen-induced tumorigenesis in p53-deficient mice. Nature Genet. 5, 225–229 (1993).
Huang, P. Excision of mismatched nucleotides from DNA: a potential mechanism for enhancing DNA replication fidelity by the wild-type p53 protein. Oncogene 17, 261–270 (1998).
Janus, F. et al. Different regulation of the p53 core domain activities 3′-to-5′ exonuclease and sequence-specific DNA binding. Mol. Cell. Biol. 19, 2155–2168 (1999).
Okorokov, A. L., Ponehel, F. & Milner, J. Induced N- and C-terminal cleavage of p53: a core fragment of p53, generated by interaction with damaged DNA, promotes cleavage of the N-terminus of full-length p53, whereas ssDNA induces C-terminal cleavage of p53. EMBO J. 16, 6008–6017 (1997).
Zhou, J., Ahn, J., Wilson, S. H. & Prives, C. A role for p53 in base excision repair. EMBO J. 20, 914–923 (2001).
Bakhanashvili, M. Exonucleolytic proofreading by p53 protein. Eur. J. Biochem. 268, 2047–2054 (2001).
Wang, C., Bogue, M. A., Roth, D. B. & Meek, K. Normal junctional diversification of immune receptors in p53-deficient mice. J. Immunol. 159, 757–762 (1997).
Cranston, A. et al. Female embryonic lethality in mice nullizygous for both Msh2 and p53. Nature Genet. 17, 114–118 (1997).
Chen, C. & Kolodner, R. D. Cross chromosomal rearrangements in Saccharomyces cerevisiae replication and recombination defective mutants. Nature Genet. 23, 81–85 (1999).
Paull, T. T. & Gellert, M. A mechanistic basis for Mre11-directed DNA joining at microhomologies. Proc. Natl Acad. Sci. USA 97, 6409–6414 (2000).
Volkmer, E. & Karnitz, I. M. Human homologs of Schizosaccharomyces pombe Rad1, Hus1 and Rad9 form a DNA damage-responsive protein complex. J. Biol. Chem. 274, 567–570 (1999).
Hawn, M. T. et al. Evidence for a connection between the mismatch repair system and G2 cell cycle checkpoint. Cancer Res. 55, 3721–3725 (1995).
Parker, A. E. et al. A human homologue of the Schizosaccharomyces pombe rad1+ checkpoint gene encodes an exonuclease. J. Biol. Chem. 273, 18332–18339 (1998).
Onel, K., Thelen, M. P., Ferguson, D. O., Bennet, R. L. & Holloman, W. K. Mutation avoidance and DNA repair proficiency in Ustilago maydis are differentially lost with progressive truncation of the REC1 gene product. Mol. Cell. Biol. 15, 5329–5338 (1995).
Bessho, T. & Sancar, A. Human DNA damage checkpoint protein hRad9 is a 3′ to 5′ exonuclease. J. Biol. Chem. 275, 7451–7454 (2000).
Venclovas, C. & Thelen, M. P. Structure-based predictions of Rad1, Rad9, Hus1 and Rad17 participation in sliding clamp and clamp-loading complexes. Nucleic Acids Res. 28, 2481–2493 (2000).
Udell, C. M., Lee, S. K. & Davey, S. HRAD1 and MRAD1 encode mammalian homolog of the fusion yeast rad1+ cell cycle checkpoint control gene. Nucleic Acids Res. 26, 3971–3976 (1998).
Robson, C. N. & Hickson, I. D. Isolation of cDNA clones encoding a human apurinic/apyrimidinic endonuclease that corrects DNA repair and mutagenesis defects in Escherichia coli xth (exonuclease III) mutants. Nucleic Acids Res. 19, 1087–1092 (1991).
Ludwig, D. L. et al. A murine AP-endonuclease gene-targeted deficiency with post-implantation embryonic progression and ionizing sensitivity. Mutat. Res. 409, 17–29 (1998).
Chou, K. M., Kukhanova, M. & Cheng, Y. C. A novel action of human apurinic/apyrimidinic endonuclease excision of L-configuration deoxyribonucleoside analogs from the 3′ termini of DNA. J. Biol. Chem. 275, 31009–31015 (2000).
Chou, K. M. & Cheng, Y. C. An exonucleolytic activity of human apurinic/apyrimidinic endonuclease on 3′ mispaired DNA. Nature 415, 655–659 (2002).This paper describes a new role for APE1 as the excision 3′–5′ exonuclease proofreader for DNA polymerase β, particularly for removing nucleotide analogues.
Gulliams, T.-G., Teng, M. & Halligan, B. D. Site directed DNA binding. Biochimie 79, 13–22 (1997).
Zhu, L. & Halligan, B. D. Characterization of a 3′–5′ exonuclease associated with VDJP. Biochem. Biophys. Res. Commun. 259, 262–270 (1999).
Halligan, B. D., Teng, M., Guilliams, T. G., Nauert, J. B. & Halligan, N. L. Cloning of the murine cDNA encoding VDJP, a protein homologous to the large subunit of replication factor C and bacterial DNA ligases. Gene 161, 217–222 (1995).
McAlear, M. A., Tuffo, K. M. & Holm, C. Large subunit of replication factor C (Rfc 1p/Cdc44p) is required for DNA replication and DNA repair in Saccharomyces cerevisiae. Genetics 142, 65–78 (1996).
Viswanathan, M. & Lovett, S. T. Single-strand DNA-specific exonucleases in Escherichia coli: roles in repair and mutation avoidance. Genetics 149, 7–16 (1998).
Setlow, P. & Kornberg, A. Deoxyribonucleic acid polymerase: two distinct enzymes in one polypeptide. II. A proteolytic fragment containing the 5′ leads to 3′ exonuclease function. Restoration of intact enzyme function from the two proteolytic fragments. J. Biol. Chem. 247, 232–240 (1972).
Mol, C. D., Kuo, C. F., Thayer, M.-M., Cunninghram, R. P. & Tainer, J. A. Structure and function of the multifunctional DNA-repair enzyme exonuclease III. Nature 374, 381–386 (1995).
Jorgensen, S. E. & Koerner, J. F. Separation and characterization of deoxyribonuclease of Escherichia coli B. Chromatographic separation and properties of two deoxyribo-oligonucleotidases. J. Biol. Chem. 241, 3090–3096 (1966).
Goldmark, P. J. & Linn, S. Purification and properties of the recBC DNase of Escherichia coli K-12. J. Biol. Chem. 247, 1849–1860 (1972).
Setlow, P., Brutlag, D. & Kornberg, A. Deoxyribonucleic acid polymerase: two distinct enzymes in one polypeptide. I. A proteolytic fragment containing the polymerase and 3′ leads to 5′ exonuclease functions. J. Biol. Chem. 247, 224–231 (1972).
Kushner, S. R., Nagaishi, H. & Clark, A. J. Isolation of exonuclease VIII: the enzyme associated with sbcA indirect suppressor. Proc. Natl Acad. Sci. USA 71, 3593–3597 (1974).
Qiu, Z. & Goodman, M. F. The Escherichia coli polB locus is identical to dinA, the structural gene for DNA polymerase II. Characterization of Pol II purified from polB mutant. J. Biol. Chem. 272, 8611–8617 (1997).
Lovett, S. T. & Clark, A. J. Genetic analysis of the recJ gene of Escherichia coli K-12. J. Bacteriol. 157, 190–196 (1984).
Connelly, J. C., deLeau, E. S., Okely, E. A. & Leach, D. R. F. Overexpression, purification, and characterization of the sbcCD protein from Escherichia coli. J. Biol. Chem. 272, 19819–19826 (1997).
Chung, D. W. et al. Primary structure of the catalytic subunit of human DNA polymerase δ and chromosomal location of the gene. Proc. Natl Acad. Sci. USA 88, 11197–11201 (1991).
Kesti, T., Frantti, H. & Syväoja, J. E. Molecular cloning of the cDNA for the catalytic subunit of human DNA polymerase ɛ. J. Biol. Chem. 268, 10238–10245 (1993).
Ropp, P. A. & Copeland, W. C. Cloning and characterization of the human mitochondrial DNA polymerase, DNA polymerase γ. Genomics 36, 449–458 (1996).
Mummenbrauer, T. et al. p53 protein exhibits 3′-to–5′ exonuclease activity. Cell 85, 1089–1099 (1996).
Morrison, A. & Sugino, A. The 3′ to 5′ exonucleases of both DNA polymerase δ and ɛ participate in correcting errors of DNA replication in Saccharomyces cerevisiae. Mol. Gen. Genet. 242, 289–296 (1994).
Stewart, G. S. et al. The DNA double-strand break repair gene hMre11 is mutated in individuals with an ataxia-telangiectasia-like disorder. Cell 99, 577–587 (1999).
Carney, J. P. et al. The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: linkage of double-strand break repair to the cellular DNA damage response. Cell 93, 477–486 (1998).
Franklin, M. C., Wang, J. & Steitz, T. A. Structure of the replicating complex of a pol α family DNA polymerase. Cell 105, 657–667 (2001).
Shamoo, Y. & Steitz, T. A. Building a replisome from interacting pieces: sliding clamp complexed to a peptide from DNA polymerase and a polymerase editing complex. Cell 99, 155–166 (1999).
Acknowledgements
We thank V. Kalinin, V. Krutyakov, G. Villani, C. Bieri and U. Sheveleva for their critical reading of the manuscript; Ursi Hübscher, R. Hobi, P. Schrafll and N. Pirogova for their assistance with graphics; T. Steitz for permission to reproduce Figure 1; and K. Ramadan for his assistance on the manuscript. The work carried out in the authors' laboratories was supported by the Swiss National Science Foundation, the European Union TMR grant, the Kanton of Zürich and the Russian Foundation for Basic Research. We apologize to those researchers whose work could not be cited directly because of the strict limit on the number of references.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Interpro
3′–5′ proofreading exonuclease domain
LocusLink
<i>Saccharomyces</i> Genome Database
Swiss-Prot
Glossary
- TRANSLESION DNA SYNTHESIS
-
DNA synthesis on a DNA molecule that contains altered or missing bases.
- MISMATCH REPAIR
-
This repair process recognizes incorrectly paired bases after DNA replication has occurred. The mismatch repair complex can distinguish between the old and newly synthesized DNA strands.
- PRIMASE
-
A special type of RNA polymerase that can synthesize an RNA primer of ten nucleotides, which is subsequently elongated by a DNA polymerase (such as DNA polymerase α in eukaryotes).
- OKAZAKI FRAGMENTS
-
Short pieces of DNA that are synthesized on the lagging strand at the replication fork.
- PROCESSIVE DNA SYNTHESIS
-
A processive DNA polymerase can synthesize from a few to several thousand nucleotides per binding event to DNA.
- DISTRIBUTIVE DNA SYNTHESIS
-
A distributive DNA polymerase can synthesize only one nucleotide per binding event to DNA.
- DNA POLYMERASE (POL) HOLOENZYME
-
A replicative DNA polymerase complex that consists of a DNA polymerase and several auxiliary proteins (for example, pol δ, proliferating cell nuclear antigen and replication factor-C).
- ERROR CATASTROPHE
-
When mutations accumulate during the lifespan of a cell or organism, there is a point at which the mutations have accumulated to such an extent that the entire system breaks down.
- SPONTANEOUS MUTATION FREQUENCY
-
Mutations that occur in a genome without an obvious influence from the environment (for example, oxidative damage).
- ARANUCLEOTIDES
-
A class of chemical compounds that are analogous to dNTPs, but with an arabinose sugar instead of a deoxyribose (for example, cytosine arabinoside triphosphate). These compounds are widely used in anticancer and antiviral therapies.
- ERROR-PRONE CONDITIONS
-
Conditions in a cell that tend to lead to errors in DNA replication (for example, an imbalance of substrate dNTPs).
- ERROR-FREE CONDITIONS
-
Conditions in a cell that permit DNA replication to occur with a low frequency of errors.
- BASE-EXCISION REPAIR
-
An essential repair mechanism that removes damaged bases from the DNA. The remaining abasic site is recognized by an apurinic/apyrimidinic endonuclease. The gap is then filled by DNA polymerase and sealed by DNA ligase.
- KU70/80
-
An enzyme that is an essential part of the complex that is responsible for non-homologous end joining.
- V(D)J RECOMBINATION
-
The site-specific recombination of immunoglobulin coding regions from multiple copies in the germ line to just one variable (V), one diversity (D) and one joining (J) region in the process of forming a functional immunoglobulin gene in B cells.
- CAMPTOTHECIN
-
A specific inhibitor of DNA topoisomerase I, an enzyme that is responsible for the topology of DNA during replication. DNA topoisomerase I cuts DNA at one strand and subsequently reseals the nick. Camptothecin inhibits DNA topoisomerase I by freezing the enzyme at the nick, and this results in a replication defect.
- SECONDARY STRUCTURE
-
The double helix can form secondary structures when, for instance, palindromic sequences occur. Such structures include stem–loops or 'cloverleafs'.
- NON-HOMOLOGOUS END JOINING
-
A recombination event that allows DNA to be rejoined after double-strand breaks have occurred. This rejoining process does not require homologous DNA sequences.
- HETEROLOGOUS DNA
-
A DNA molecule that has no homology to an existing one.
Rights and permissions
About this article
Cite this article
Shevelev, I., Hübscher, U. The 3′–5′ exonucleases. Nat Rev Mol Cell Biol 3, 364–376 (2002). https://doi.org/10.1038/nrm804
Issue Date:
DOI: https://doi.org/10.1038/nrm804
This article is cited by
-
Recommendations for the classification of germline variants in the exonuclease domain of POLE and POLD1
Genome Medicine (2023)
-
An exonuclease V homologue is expressed predominantly during early megasporogenesis in apomictic Brachiaria brizantha
Planta (2023)
-
Coiled-coil heterodimer-based recruitment of an exonuclease to CRISPR/Cas for enhanced gene editing
Nature Communications (2022)
-
Self-assembled Nanosheets of Perylene Monoamide Derivative as Sensitive Fluorescent Biosensor for Exonuclease III Activity
Chemical Research in Chinese Universities (2022)
-
Chromosome-scale comparative sequence analysis unravels molecular mechanisms of genome dynamics between two wheat cultivars
Genome Biology (2018)