Abstract
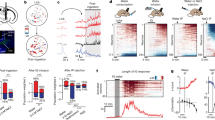
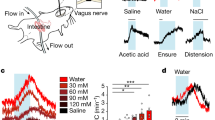
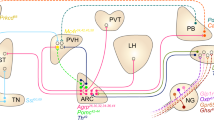
Thirst motivates animals to find and consume water. More than 40 years ago, a set of interconnected brain structures known as the lamina terminalis was shown to govern thirst. However, owing to the anatomical complexity of these brain regions, the structure and dynamics of their underlying neural circuitry have remained obscure. Recently, the emergence of new tools for neural recording and manipulation has reinvigorated the study of this circuit and prompted re-examination of longstanding questions about the neural origins of thirst. Here, we review these advances, discuss what they teach us about the control of drinking behaviour and outline the key questions that remain unanswered.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cannon, W. B. The physiological basis of thirst. Proc. R. Soc. B Biol. Sci. 90, 283–301 (1918).
Montgomery, M. F. The role of the salivary glands in the thirst mechanism. Am. J. Physiol. 96, 221–227 (1931).
Leschke, E. Ueber die durstempfindung [German]. Arch. Psychiatrie Nervenkrankheiten 59, 773–781 (1918).
Gilman, A. The relation between blood osmotic pressure, fluid distribution and voluntary water intake. Am. J. Physiol. 120, 323–328 (1937).
Wolf, A. V. Osmometric analysis of thirst in man and dog. Am. J. Physiol. 161, 75–86 (1950).
Fitzsimons, J. T. The effects of slow infusions of hypertonic solutions on drinking and drinking thresholds in rats. J. Physiol. 167, 344–354 (1963).
Fitzsimons, J. T. Drinking by rats depleted of body fluid without increase in osmotic pressure. J. Physiol. 159, 297–309 (1961).
Stricker, E. M. Extracellular fluid volume and thirst. Am. J. Physiol. 211, 232–238 (1966).
Lehr, D., Mallow, J. & Krukowski, M. Copious drinking and simultaneous inhibition of urine flow elicited by β-adrenergic stimulation and contrary effect of α-adrenergic stimulation. J. Pharmacol. Exp. Ther. 158, 150–163 (1967).
Fitzsimons, J. T. Angiotensin, thirst, and sodium appetite. Physiol. Rev. 78, 583–686 (1998).
Bourque, C. W. Central mechanisms of osmosensation and systemic osmoregulation. Nat. Rev. Neurosci. 9, 519–531 (2008).
Leib, D. E., Zimmerman, C. A. & Knight, Z. A. Thirst. Curr. Biol. 26, R1260–R1265 (2016).
Oka, Y., Ye, M. & Zuker, C. S. Thirst driving and suppressing signals encoded by distinct neural populations in the brain. Nature 520, 349–352 (2015).
Betley, J. N. et al. Neurons for hunger and thirst transmit a negative-valence teaching signal. Nature 521, 180–185 (2015).
Nation, H. L., Nicoleau, M., Kinsman, B. J., Browning, K. N. & Stocker, S. D. DREADD-induced activation of subfornical organ neurons stimulates thirst and salt appetite. J. Neurophysiol. 115, 3123–3129 (2016).
Zimmerman, C. A. et al. Thirst neurons anticipate the homeostatic consequences of eating and drinking. Nature 537, 680–684 (2016).
Abbott, S. B. G., Machado, N. L. S., Geerling, J. C. & Saper, C. B. Reciprocal control of drinking behavior by median preoptic neurons in mice. J. Neurosci. 36, 8228–8237 (2016).
Gizowski, C., Zaelzer, C. & Bourque, C. W. Clock-driven vasopressin neurotransmission mediates anticipatory thirst prior to sleep. Nature 537, 685–688 (2016).
Mandelblat-Cerf, Y. et al. Bidirectional anticipation of future osmotic challenges by vasopressin neurons. Neuron 93, 57–65 (2017).
Jarvie, B. C. & Palmiter, R. D. HSD2 neurons in the hindbrain drive sodium appetite. Nat. Neurosci. 20, 167–169 (2017).
Matsuda, T. et al. Distinct neural mechanisms for the control of thirst and salt appetite in the subfornical organ. Nat. Neurosci. 20, 230–241 (2017).
Andersson, B. Polydipsia caused by intrahypothalamic injections of hypertonic NaCl-solutions. Experientia 8, 157–158 (1952).
Andersson, B. The effect of injections of hypertonic NaCl-solutions into different parts of the hypothalamus of goats. Acta Physiol. Scand. 28, 188–201 (1953).
Andersson, B. & McCann, S. M. A further study of polydipsia evoked by hypothalamic stimulation of the goat. Acta Physiol. Scand. 33, 333–346 (1955).
Epstein, A. N., Fitzsimons, J. T. & Rolls, B. J. Drinking induced by injection of angiotensin into the brain of the rat. J. Physiol. 210, 457–474 (1970).
Fitzsimons, J. T. The effect on drinking of peptide precursors and of shorter chain peptide fragments of angiotensin II injected into the rat's diencephalon. J. Physiol. 214, 295–303 (1971).
Andersson, B. & McCann, S. M. The effect of hypothalamic lesions on the water intake of the dog. Acta Physiol. Scand. 35, 312–320 (1955).
Simpson, J. B. & Routtenberg, A. Subfornical organ: site of drinking elicitation by angiotensin II. Science 181, 1172–1175 (1973).
Andersson, B., Leksell, L. G. & Lishajko, F. Perturbations in fluid balance induced by medially placed forebrain lesions. Brain Res. 99, 261–275 (1975).
Buggy, J. & Jonhson, A. K. Preoptic-hypothalamic periventricular lesions: thirst deficits and hypernatremia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 233, R44–R52 (1977).
McKinley, M. J., Denton, D. A. & Weisinger, R. S. Sensors for antidiuresis and thirst: osmoreceptors or CSF sodium detectors? Brain Res. 141, 89–103 (1978).
McKinley, M. J. et al. Osmoregulatory thirst in sheep is disrupted by ablation of the anterior wall of the optic recess. Brain Res. 236, 210–215 (1982).
Thrasher, T. N., Keil, L. C. & Ramsay, D. J. Lesions of the organum vasculosum of the lamina terminalis (OVLT) attenuate osmotically-induced drinking and vasopressin secretion in the dog. Endocrinology 110, 1837–1839 (1982).
McKinley, M. J. et al. The sensory circumventricular organs of the mammalian brain. Adv. Anat. Embryol. Cell Biol. 172, 1–127 (2003).
Sayer, R. J., Hubbard, J. I. & Sirett, N. E. Rat organum vasculosum laminae terminalis in vitro: responses to transmitters. Am. J. Physiol. Regul. Integr. Comp. Physiol. 247, R374–R379 (1984).
Sibbald, J. R., Hubbard, J. I. & Sirett, N. E. Responses from osmosensitive neurons of the rat subfornical organ in vitro. Brain Res. 461, 205–214 (1988).
Vivas, L., Chiaraviglio, E. & Carrer, H. F. Rat organum vasculosum laminae terminalis in vitro: responses to changes in sodium concentration. Brain Res. 519, 294–300 (1990).
Anderson, J. W., Washburn, D. L. S. & Ferguson, A. V. Intrinsic osmosensitivity of subfornical organ neurons. Neuroscience 100, 539–547 (2000).
Felix, D. & Akert, K. The effect of angiotensin II on neurones of the cat subfornical organ. Brain Res. 76, 350–353 (1974).
Knowles, W. D. & Phillips, M. I. Angiotensin II responsive cells in the organum vasculosum lamina terminalis (OVLT) recorded in hypothalamic brain slices. Brain Res. 197, 256–259 (1980).
Gutman, M. B., Ciriello, J. & Mogenson, G. J. Effects of plasma angiotensin II and hypernatremia on subfornical organ neurons. Am. J. Physiol. Regul. Integr. Comp. Physiol. 254, R746–R754 (1988).
McKinley, M. J., Badoer, E. & Oldfield, B. J. Intravenous angiotensin II induces Fos-immunoreactivity in circumventricular organs of the lamina terminalis. Brain Res. 594, 295–300 (1992).
Gebke, E., Müller, A. R., Jurzak, M. & Gerstberger, R. Angiotensin II-induced calcium signalling in neurons and astrocytes of rat circumventricular organs. Neuroscience 85, 509–520 (1998).
Babic, T., Roder, S. & Ciriello, J. Direct projections from caudal ventrolateral medullary depressor sites to the subfornical organ. Brain Res. 1003, 113–121 (2004).
Ciriello, J. Caudal ventrolateral medulla mediates baroreceptor afferent inputs to subfornical organ angiotensin II responsive neurons. Brain Res. 1491, 127–135 (2013).
McKinley, M. J. et al. The median preoptic nucleus: front and centre for the regulation of body fluid, sodium, temperature, sleep and cardiovascular homeostasis. Acta Physiol. 214, 8–32 (2015).
Swanson, L. W. An autoradiographic study of the efferent connections of the preoptic region in the rat. J. Comp. Neurol. 167, 227–256 (1976).
Miselis, R. R., Shapiro, R. E. & Hand, P. J. Subfornical organ efferents to neural systems for control of body water. Science 205, 1022–1025 (1979).
Camacho, A. & Phillips, M. I. Horseradish peroxidase study in rat of the neural connections of the organum vasculosum of the lamina terminalis. Neurosci. Lett. 25, 201–204 (1981).
Lind, R. W., van Hoesen, G. W. & Johnson, A. K. An HRP study of the connections of the subfornical organ of the rat. J. Comp. Neurol. 210, 265–277 (1982).
Saper, C. B. & Levisohn, D. Afferent connections of the median preoptic nucleus in the rat: anatomical evidence for a cardiovascular integrative mechanism in the anteroventral third ventricular (AV3V) region. Brain Res. 288, 21–31 (1983).
Buggy, J. & Fisher, A. E. Evidence for a dual central role for angiotensin in water and sodium intake. Nature 250, 733–735 (1974).
Robertson, A., Kucharczyk, J. & Mogenson, G. J. Drinking behavior following electrical stimulation of the subfornical organ in the rat. Brain Res. 274, 197–200 (1983).
Fluharty, S. J. & Epstein, A. N. Sodium appetite elicited by intracerebroventricular infusion of angiotensin II in the rat: II. Synergistic interaction with systemic mineralocorticoids. Behav. Neurosci. 97, 746–758 (1983).
Mangiapane, M. L. & Simpson, J. B. Subfornical organ: forebrain site of pressor and dipsogenic action of angiotensin II. Am. J. Physiol. Regul. Integr. Comp. Physiol. 239, R382–R389 (1980).
Ishibashi, S. & Nicolaïdis, S. Hypertension induced by electrical stimulation of the subfornical organ (SFO). Brain Res. Bull. 6, 135–139 (1981).
Ferguson, A. V. & Kasting, N. W. Electrical stimulation in subfornical organ increases plasma vasopressin concentrations in the conscious rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 251, R425–R428 (1986).
Ferguson, A. V. & Kasting, N. W. Activation of subfornical organ efferents stimulates oxytocin secretion in the rat. Regul. Pept. 18, 93–100 (1987).
Verbalis, J. G., Mangione, M. P. & Stricker, E. M. Oxytocin produces natriuresis in rats at physiological plasma concentrations. Endocrinology 128, 1317–1322 (1991).
Huang, W., Lee, S. L. & Sjöquist, M. Natriuretic role of endogenous oxytocin in male rats infused with hypertonic NaCl. Am. J. Physiol. Regul. Integr. Comp. Physiol. 268, R634–R640 (1995).
Rasmussen, M. S., Simonsen, J. A., Sandgaard, N. C. F., Høilund-Carlsen, P. F. & Bie, P. Mechanisms of acute natriuresis in normal humans on low sodium diet. J. Physiol. 546, 591–603 (2003).
Rasmussen, M. S., Simonsen, J. A., Sandgaard, N. C. F., Høilund-Carlsen, P. F. & Bie, P. Effects of oxytocin in normal man during low and high sodium diets. Acta Physiol. Scand. 181, 247–257 (2004).
Verney, E. B. The antidiuretic hormone and the factors which determine its release. Proc. R. Soc. B Biol. Sci. 135, 25–106 (1947).
Antunes-Rodrigues, J., de Castro, M., Elias, L. L. K., Valença, M. M. & McCann, S. M. Neuroendocrine control of body fluid metabolism. Physiol. Rev. 84, 169–208 (2004).
Abdelaal, A. E., Assaf, S. Y., Kucharczyk, J. & Mogenson, G. J. Effect of ablation of the subfornical organ on water intake elicited by systemically administered angiotensin-II. Can. J. Physiol. Pharmacol. 52, 1217–1220 (1974).
Simpson, J. B. & Routtenberg, A. Subfornical organ lesions reduce intravenous angiotensin-induced drinking. Brain Res. 88, 154–161 (1975).
Buggy, J. & Fisher, A. E. Anteroventral third ventricle site of action for angiotensin induced thirst. Pharmacol. Biochem. Behav. 4, 651–660 (1976).
Simpson, J. B., Epstein, A. N. & Camardo, J. S. Localization of receptors for the dipsogenic action of angiotensin II in the subfornical organ of rat. J. Comp. Physiol. Psychol. 92, 581–601 (1978).
Lind, R. W., Thunhorst, R. L. & Johnson, A. K. The subfornical organ and the integration of multiple factors in thirst. Physiol. Behav. 32, 69–74 (1984).
Mangiapane, M. L., Thrasher, T. N., Keil, L. C., Simpson, J. B. & Ganong, W. F. Role for the subfornical organ in vasopressin release. Brain Res. Bull. 13, 43–47 (1984).
Weisinger, R. S. et al. Subfornical organ lesion decreases sodium appetite in the sodium-depleted rat. Brain Res. 526, 23–30 (1990).
McKinley, M. J., Mathai, M. L., Pennington, G., Rundgren, M. & Vivas, L. Effect of individual or combined ablation of the nuclear groups of the lamina terminalis on water drinking in sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 276, R673–R683 (1999).
Thunhorst, R. L., Beltz, T. G. & Johnson, A. K. Effects of subfornical organ lesions on acutely induced thirst and salt appetite. Am. J. Physiol. Regul. Integr. Comp. Physiol. 277, R56–R65 (1999).
Guyenet, P. G. The sympathetic control of blood pressure. Nat. Rev. Neurosci. 7, 335–346 (2006).
Adolph, E. F. Measurements of water drinking in dogs. Am. J. Physiol. 125, 75–86 (1938).
Bellows, R. T. Time factors in water drinking in dogs. Am. J. Physiol. 125, 87–97 (1938).
Adolph, E. F., Barker, J. P. & Hoy, P. A. Multiple factors in thirst. Am. J. Physiol. 178, 538–562 (1954).
Rolls, B. J. et al. Thirst following water deprivation in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 239, R476–R482 (1980).
Figaro, M. K. & Mack, G. W. Regulation of fluid intake in dehydrated humans: role of oropharyngeal stimulation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 272, R1740–R1746 (1997).
Hoffmann, M. L., DenBleyker, M., Smith, J. C. & Stricker, E. M. Inhibition of thirst when dehydrated rats drink water or saline. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R1199–R1207 (2006).
Denton, D. et al. Correlation of regional cerebral blood flow and change of plasma sodium concentration during genesis and satiation of thirst. Proc. Natl Acad. Sci. USA 96, 2532–2537 (1999).
Denton, D. et al. Neuroimaging of genesis and satiation of thirst and an interoceptor-driven theory of origins of primary consciousness. Proc. Natl Acad. Sci. USA 96, 5304–5309 (1999).
Egan, G. et al. Neural correlates of the emergence of consciousness of thirst. Proc. Natl Acad. Sci. USA 100, 15241–15246 (2003).
Farrell, M. J. et al. Cortical activation and lamina terminalis functional connectivity during thirst and drinking in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 301, R623–R631 (2011).
Saker, P. et al. Regional brain responses associated with drinking water during thirst and after its satiation. Proc. Natl Acad. Sci. USA 111, 5379–5384 (2014).
Saker, P., Farrell, M. J., Egan, G. F., McKinley, M. J. & Denton, D. A. Overdrinking, swallowing inhibition, and regional brain responses prior to swallowing. Proc. Natl Acad. Sci. USA 113, 12274–12279 (2016).
Mendelson, J. & Chillag, D. Tongue cooling: a new reward for thirsty rodents. Science 170, 1418–1421 (1970).
Kapatos, G. & Gold, R. M. Tongue cooling during drinking: a regulator of water intake in rats. Science 176, 685–686 (1972).
Deaux, E. Thirst satiation and the temperature of ingested water. Science 181, 1166–1167 (1973).
Boulze, D., Montastruc, P. & Cabanac, M. Water intake, pleasure and water temperature in humans. Physiol. Behav. 30, 97–102 (1983).
Salata, R. A., Verbalis, J. G. & Robinson, A. G. Cold water stimulation of oropharyngeal receptors in man inhibits release of vasopressin. J. Clin. Endocrinol. Metab. 65, 561–567 (1987).
Peyrot des Gachons, C. et al. Oral cooling and carbonation increase the perception of drinking and thirst quenching in thirsty adults. PLoS ONE 11, e0162261 (2016).
Conchon, M. F. & Fonseca, L. F. Efficacy of an ice popsicle on thirst management in the immediate postoperative period: a randomized clinical trial. J. PeriAnesthesia Nurs. http://dx.doi.org/10.1016/j.jopan.2016.03.009 (2017).
McKemy, D. D., Neuhausser, W. M. & Julius, D. Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature 416, 52–58 (2002).
Dhaka, A., Earley, T. J., Watson, J. & Patapoutian, A. Visualizing cold spots: TRPM8-expressing sensory neurons and their projections. J. Neurosci. 28, 566–575 (2008).
Yarmolinsky, D. A. et al. Coding and plasticity in the mammalian thermosensory system. Neuron 92, 1079–1092 (2016).
Bott, E., Denton, D. A. & Weller, S. Water drinking in sheep with oesophageal fistulae. J. Physiol. 176, 323–336 (1965).
Maddison, S., Wod, R. J., Rolls, E. T., Rolls, B. J. & Gibbs, J. Drinking in the rhesus monkeys: peripheral factors. J. Comp. Physiol. Psychol. 94, 365–374 (1980).
Kraly, F. S., Kim, Y.-M., Dunham, L. M. & Tribuzio, R. A. Drinking after intragastric NaCl without increase in systemic plasma osmolality in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 269, R1085–R1092 (1995).
Stricker, E. M., Callahan, J. B., Huang, W. & Sved, A. F. Early osmoregulatory stimulation of neurohypophyseal hormone secretion and thirst after gastric NaCl loads. Am. J. Physiol. Regul. Integr. Comp. Physiol. 282, R1710–R1717 (2002).
Hyde, T. M. & Miselis, R. R. Area postrema and adjacent nucleus of the solitary tract in water and sodium balance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 247, R173–R182 (1984).
Curtis, K. S., Verbalis, J. G. & Stricker, E. M. Area postrema lesions in rats appear to disrupt rapid feedback inhibition of fluid intake. Brain Res. 726, 31–38 (1996).
Krashes, M. J. Forecast for water balance. Nature 537, 626–627 (2016).
Thrasher, T. N., Nistal-Herrera, J. F., Keil, L. C. & Ramsay, D. J. Satiety and inhibition of vasopressin secretion after drinking in dehydrated dogs. Am. J. Physiol. Endocrinol. Metab. 240, E394–E401 (1981).
Arnauld, E. & du Pont, J. Vasopressin release and firing of supraoptic neurosecretory neurones during drinking in the dehydrated monkey. Pflügers Arch. 394, 195–201 (1982).
Geelen, G. et al. Inhibition of plasma vasopressin after drinking in dehydrated humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 247, R968–R971 (1984).
Huang, W., Sved, A. F. & Stricker, E. M. Water ingestion provides an early signal inhibiting osmotically stimulated vasopressin secretion in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279, R756–R760 (2000).
Vincent, J. D., Arnauld, E. & Bioulac, B. Activity of osmosensitive single cells in the hypothalamus of the behaving monkey during drinking. Brain Res. 44, 371–384 (1972).
Bellisle, F. & Le Magnen, J. The structure of meals in humans: eating and drinking patterns in lean and obese subjects. Physiol. Behav. 27, 649–658 (1981).
Phillips, P. A., Rolls, B. J., Ledingham, J. G. G. & Morton, J. J. Body fluid changes, thirst and drinking in man during free access to water. Physiol. Behav. 33, 357–363 (1984).
Cizek, L. J. & Nocenti, M. R. Relationship between water and food ingestion in the rat. Am. J. Physiol. 208, 615–620 (1965).
Fitzsimons, J. T. & Le Magnen, J. Eating as a regulatory control of drinking in the rat. J. Comp. Physiol. Psychol. 67, 273–283 (1969).
Kissileff, H. R. Food-associated drinking in the rat. J. Comp. Physiol. Psychol. 67, 284–300 (1969).
Epstein, A. N., Spector, D., Samman, A. & Goldblum, C. Exaggerated prandial drinking in the rat without salivary glands. Nature 201, 1342–1343 (1964).
Kissileff, H. R. Oropharyngeal control of prandial drinking. J. Comp. Physiol. Psychol. 67, 309–319 (1969).
Lepkovsky, S., Lyman, R., Fleming, D., Nagumo, M. & Dimick, M. M. Gastrointestinal regulation of water and its effect on food intake and rate of digestion. Am. J. Physiol. 188, 237–231 (1957).
Gutman, Y. & Krausz, M. Regulation of food and water intake in rats as related to plasma osmolarity and volume. Physiol. Behav. 4, 311–313 (1969).
Pernice, B. & Scagliosi, G. Ueber die wirkung der wasserentziehung auf thiere [German]. Arch. Pathol. Anatomie Physiol. Klinische Med. 139, 155–184 (1895).
Bing, F. C. & Mendel, L. B. The relationship between food and water intakes in mice. Am. J. Physiol. 98, 169–179 (1931).
Adolph, E. F. Urges to eat and drink in rats. Am. J. Physiol. 151, 110–125 (1947).
Ørskov, C., Poulsen, S. S., Møller, M. & Holst, J. J. Glucagon-like peptide-1 receptors in the subfornical organ and the area postrema are accessible to circulating glucagon-like peptide-1. Diabetes 45, 832–835 (1996).
Samson, W. K., White, M. M., Price, C. & Ferguson, A. V. Obestatin acts in brain to inhibit thirst. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292, R637–R643 (2007).
Fry, M., Hoyda, T. D. & Ferguson, A. V. Making sense of it: roles of the sensory circumventricular organs in feeding and regulation of energy homeostasis. Exp. Biol. Med. 232, 14–26 (2007).
Ahmed, A.-S. F., Dai, L., Ho, W., Ferguson, A. V. & Sharkey, K. A. The subfornical organ: a novel site of action of cholecystokinin. Am. J. Physiol. Regul. Integr. Comp. Physiol. 306, R363–R373 (2014).
Riediger, T., Rauch, M. & Schmid, H. A. Actions of amylin on subfornical organ neurons and on drinking behavior in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 276, R514–R521 (1999).
Riediger, T., Schmid, H. A., Young, A. A. & Simon, E. Pharmacological characterisation of amylin-related peptides activating subfornical organ neurones. Brain Res. 837, 161–168 (1999).
Barth, S. W., Riediger, T., Lutz, T. A. & Rechkemmer, G. Peripheral amylin activates circumventricular organs expressing calcitonin receptor a/b subtypes and receptor-activity modifying proteins in the rat. Brain Res. 997, 97–102 (2004).
Pulman, K. J., Fry, W. M., Cottrell, G. T. & Ferguson, A. V. The subfornical organ: a central target for circulating feeding signals. J. Neurosci. 26, 2022–2030 (2006).
Lakhi, S., Snow, W. & Fry, M. Insulin modulates the electrical activity of subfornical organ neurons. Neuroreport 24, 329–334 (2013).
Smith, P. M. et al. The subfornical organ: a central nervous system site for actions of circulating leptin. Am. J. Physiol. Regul. Integr. Comp. Physiol. 296, R512–R520 (2009).
Alim, I., Fry, W. M., Walsh, M. H. & Ferguson, A. V. Actions of adiponectin on the excitability of subfornical organ neurons are altered by food deprivation. Brain Res. 1330, 72–82 (2010).
Dai, L., Smith, P. M., Kuksis, M. & Ferguson, A. V. Apelin acts in the subfornical organ to influence neuronal excitability and cardiovascular function. J. Physiol. 591, 3421–3432 (2013).
Zucker, I. Light-dark rhythms in rat eating and drinking behavior. Physiol. Behav. 6, 115–126 (1971).
Reppert, S. M. & Weaver, D. R. Coordination of circadian timing in mammals. Nature 418, 935–941 (2002).
Dibner, C., Schibler, U. & Albrecht, U. The mammalian circadian timing system: organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 72, 517–549 (2010).
Stephan, F. K. & Zucker, I. Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proc. Natl Acad. Sci. USA 69, 1583–1586 (1972).
Trudel, E. & Bourque, C. W. Central clock excites vasopressin neurons by waking osmosensory afferents during late sleep. Nat. Neurosci. 13, 467–474 (2010).
Geerling, J. C. & Loewy, A. D. Central regulation of sodium appetite. Exp. Physiol. 93, 177–209 (2008).
Crews, E. C. & Rowland, N. E. Role of angiotensin in body fluid homeostasis of mice: effect of losartan on water and NaCl intakes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 288, R638–R644 (2005).
Richter, C. P. Increased salt appetite in adrenalectomized rats. Am. J. Physiol. 115, 155–161 (1936).
Rice, K. K. & Richter, C. P. Increased sodium chloride and water intake of normal rats treated with desoxycorticosterone acetate. Endocrinology 33, 106–115 (1943).
Wolf, G. Sodium appetite elicited by aldosterone. Psychonom. Sci. 1, 211–212 (1964).
Sakai, R. R., Nicolaïdis, S. & Epstein, A. N. Salt appetite is suppressed by interference with angiotensin II and aldosterone. Am. J. Physiol. Regul. Integr. Comp. Physiol. 251, R762–R768 (1986).
Simpson, S. A. et al. Isolierung eines neuen kristallisierten hormons aus nebennieren mit besonders hoher wirksamkeit auf den mineralstoffwechsel [German]. Experientia 9, 333–335 (1953).
Vander, A. J. et al. Effects of adrenalectomy and aldosterone on proximal and distal tubular sodium reabsorption. Proc. Soc. Exp. Biol. Med. 99, 323–325 (1958).
Funder, J. W., Pearce, P. T., Smith, R. & Smith, A. I. Mineralocorticoid action: target tissue specificity is enzyme, not receptor, mediated. Science 242, 583–585 (1988).
Geerling, J. C., Chimenti, P. C. & Loewy, A. D. Phox2b expression in the aldosterone-sensitive HSD2 neurons of the NTS. Brain Res. 1226, 82–88 (2008).
Geerling, J. C., Kawata, M. & Loewy, A. D. Aldosterone-sensitive neurons in the rat central nervous system. J. Comp. Neurol. 494, 515–527 (2006).
Geerling, J. C., Engeland, W. C., Kawata, M. & Loewy, A. D. Aldosterone target neurons in the nucleus tractus solitarius drive sodium appetite. J. Neurosci. 26, 411–417 (2006).
Geerling, J. C. & Loewy, A. D. Aldosterone-sensitive neurons in the nucleus of the solitary tract: efferent projections. J. Comp. Neurol. 497, 223–250 (2006).
Geerling, J. C. et al. FoxP2 expression defines dorsolateral pontine neurons activated by sodium deprivation. Brain Res. 1375, 19–27 (2011).
Davis, J. O., Carpenter, C. C. J., Ayers, C. R., Holman, J. E. & Bahn, R. C. Evidence for secretion of an aldosterone-stimulating hormone by the kidney. J. Clin. Invest. 40, 684–696 (1961).
Blair-West, J. R. et al. Humoral stimulation of adrenal cortical secretion. J. Clin. Invest. 41, 1606–1627 (1962).
Ames, R. P., Borkowski, A. J., Sicinski, A. M. & Laragh, J. H. Prolonged infusions of angiotensin II and norepinephrine and blood pressure, electrolyte balance, and aldosterone and cortisol secretion in normal man and in cirrhosis with ascites. J. Clin. Invest. 44, 1171–1186 (1965).
Thunhorst, R. L., Ehrlich, K. J. & Simpson, J. B. Subfornical organ participates in salt appetite. Behav. Neurosci. 104, 637–642 (1990).
Ruhf, A. A., Starbuck, E. M. & Fitts, D. A. Effects of SFO lesions on salt appetite during multiple sodium depletions. Physiol. Behav. 74, 629–636 (2001).
Wilson, W. L., Starbuck, E. M. & Fitts, D. A. Salt appetite of adrenalectomized rats after a lesion of the SFO. Behav. Brain Res. 136, 449–453 (2002).
Galaverna, O. G. et al. Lesions of the central nucleus of the amygdala I: effects on taste reactivity, taste aversion learning and sodium appetite. Behav. Brain Res. 59, 11–17 (1993).
Zardetto-Smith, A. M., Beltz, T. G. & Johnson, A. K. Role of the central nucleus of the amygdala and bed nucleus of the stria terminalis in experimentally-induced salt appetite. Brain Res. 645, 123–134 (1994).
Johnson, A. K., de Olmos, J., Pastuskovas, C. V., Zardetto-Smith, A. M. & Vivas, L. The extended amygdala and salt appetite. Ann. NY Acad. Sci. 877, 258–280 (1999).
Cameron, P., Hiroi, M., Ngai, J. & Scott, K. The molecular basis for water taste in Drosophila. Nature 465, 91–95 (2010).
Lindemann, B. Taste reception. Physiol. Rev. 76, 719–766 (1996).
Ciura, S. & Bourque, C. W. Transient receptor potential vanilloid 1 is required for intrinsic osmoreception in organum vasculosum lamina terminalis neurons and for normal thirst responses to systemic hyperosmolality. J. Neurosci. 26, 9069–9075 (2006).
Sharif-Naeini, R., Witty, M.-F., Séguéla, P. & Bourque, C. W. An N-terminal variant of Trpv1 channel is required for osmosensory transduction. Nat. Neurosci. 9, 93–98 (2006).
Liedtke, W. et al. Vanilloid receptor-related osmotically activated channel (VR-OAC), a candidate vertebrate osmoreceptor. Cell 103, 525–535 (2000).
Liedtke, W. & Friedman, J. M. Abnormal osmotic regulation in trpv4−/− mice. Proc. Natl Acad. Sci. USA 100, 13698–13703 (2003).
Lechner, S. G. et al. The molecular and cellular identity of peripheral osmoreceptors. Neuron 69, 332–344 (2011).
Watanabe, E. et al. Nav2/NaG channel is involved in control of salt-intake behavior in the CNS. J. Neurosci. 20, 7743–7751 (2000).
Hiyama, T. Y. et al. Nax channel involved in CNS sodium-level sensing. Nat. Neurosci. 5, 511–512 (2002).
Drummond, H. A., Price, M. P., Welsh, M. J. & Abboud, F. M. A molecular component of the arterial baroreceptor mechanotransducer. Neuron 21, 1435–1441 (1998).
Lu, Y. et al. The ion channel ASIC2 is required for baroreceptor and autonomic control of the circulation. Neuron 64, 885–897 (2009).
Taylor, A. C., McCarthy, J. J. & Stocker, S. D. Mice lacking the transient receptor vanilloid potential 1 channel display normal thirst responses and central Fos activation to hypernatremia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 294, 1285–1293 (2008).
Kinsman, B. et al. Osmoregulatory thirst in mice lacking the transient receptor potential vanilloid type 1 (TRPV1) and/or type 4 (TRPV4) receptor. Am. J. Physiol. Regul. Integr. Comp. Physiol. 307, R1092–R1100 (2014).
Sakuta, H., Nishihara, E., Hiyama, T. Y., Lin, C.-H. & Noda, M. Nax signaling evoked by an increase in [Na+] in CSF induces water intake via EET-mediated TRPV4 activation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 311, R299–R306 (2016).
Tucker, A. B. & Stocker, S. D. Hypernatremia-induced vasopressin secretion is not altered in TRPV1−/− rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 311, R451–R456 (2016).
Roza, C. et al. Knockout of the ASIC2 channel in mice does not impair cutaneous mechanosensation, visceral mechanonociception and hearing. J. Physiol. 558, 659–669 (2004).
Zhao, Q. et al. Ion permeation and mechanotransduction mechanisms of mechanosensitive piezo channels. Neuron 89, 1248–1263 (2016).
Berridge, K. C. Motivation concepts in behavioral neuroscience. Physiol. Behav. 81, 179–209 (2004).
Travers, J. B., Dinardo, L. A. & Karimnamazi, H. Motor and premotor mechanisms of licking. Neurosci. Biobehav. Rev. 21, 631–647 (1997).
Rossi, M. A. et al. A GABAergic nigrotectal pathway for coordination of drinking behavior. Nat. Neurosci. 19, 742–748 (2016).
Abdelaal, A. E., Mercer, P. F. & Mogenson, G. J. Plasma angiotensin II levels and water intake following β-adrenergic stimulation, hypovolemia, cellular dehydration and water deprivation. Pharmacol. Biochem. Behav. 4, 317–321 (1976).
Thrasher, T. N., Keil, L. C. & Ramsay, D. J. Drinking, oropharyngeal signals, and inhibition of vasopressin secretion in dogs. Am. J. Physiol. Regul. Integr. Comp. Physiol. 253, R509–R515 (1987).
Appelgren, B. H., Thrasher, T. N., Keil, L. C. & Ramsay, D. J. Mechanism of drinking-induced inhibition of vasopressin secretion in dehydrated dogs. Am. J. Physiol. Regul. Integr. Comp. Physiol. 261, R1226–R1233 (1991).
Watts, A. G. & Boyle, C. N. The functional architecture of dehydration-anorexia. Physiol. Behav. 100, 472–477 (2010).
Zocchi, D., Wennemuth G. & Oka Y. The cellular mechanism for water detection in the mammalian taste system. Nat. Neurosci. http://dx.doi.org/10.1038/nn.4575 (2017).
Acknowledgements
The authors thank members of the Knight laboratory for comments and discussion. C.A.Z. acknowledges support from the Genentech Foundation Predoctoral Fellowship, the University of California San Francisco (UCSF) Discovery Fellowship and the US National Science Foundation Graduate Research Fellowship (1144247). D.E.L. acknowledges support from the Achievement Rewards for College Scientists (ARCS) Foundation Scholarship, the UCSF Discovery Fellowship and the US National Institutes of Health (NIH) (F31-HL131463). Z.A.K. is a New York Stem Cell Foundation (NYSCF)–Robertson Investigator. Z.A.K. acknowledges support from the NYSCF, the American Diabetes Association Pathway Program, the Rita Allen Foundation, the McKnight Foundation, the Alfred P. Sloan Foundation, the Brain & Behavior Research Foundation, the Esther A. & Joseph Klingenstein Foundation, the Program for Breakthrough Biological Research and the UCSF Diabetes and Endocrinology Research Center (P30-DK06372) and Nutrition and Obesity Research Center (P30-DK098722). This work was supported by an NIH Director's New Innovator Award (DP2-DK109533), R01-NS094781 and R01-DK106399.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Zimmerman, C., Leib, D. & Knight, Z. Neural circuits underlying thirst and fluid homeostasis. Nat Rev Neurosci 18, 459–469 (2017). https://doi.org/10.1038/nrn.2017.71
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn.2017.71
This article is cited by
-
TMEM63B channel is the osmosensor required for thirst drive of interoceptive neurons
Cell Discovery (2024)
-
A century of exercise physiology: concepts that ignited the study of human thermoregulation. Part 3: Heat and cold tolerance during exercise
European Journal of Applied Physiology (2024)
-
Comprehensive mapping of Epithelial Na+ channel α expression in the mouse brain
Brain Structure and Function (2024)
-
A melanopsin ganglion cell subtype forms a dorsal retinal mosaic projecting to the supraoptic nucleus
Nature Communications (2023)
-
Melanin-concentrating hormone and orexin shape social affective behavior via action in the insular cortex of rat
Psychopharmacology (2023)