Key Points
-
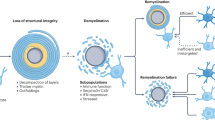
Multiple sclerosis (MS) involves profound destruction of oligodendrocytes and myelin
-
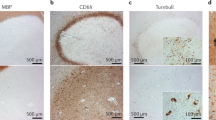
MRI and histological studies suggest that iron levels are dysregulated in MS: iron accumulates in grey matter and is depleted in normal-appearing white matter
-
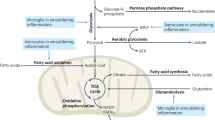
Iron accumulation promotes cytotoxicity through a variety of mechanisms, including chemical reactions leading to oxidative stress, increased proinflammatory cytokine levels, glutamate toxicity, and impaired DNA repair
-
Iron is a cofactor for a variety of enzymes involved in maintaining the health of oligodendrocytes and myelin, and may be a crucial component of remyelination
-
Treatments for iron excess in MS must address not only the increased iron levels within grey matter, but also the requirement for iron in remyelination and repair
-
The extracellular matrix, a key regulator of remyelination, may also modulate iron availability
Abstract
MRI and histological studies have shown global alterations in iron levels in the brains of patients with multiple sclerosis (MS), including increases in the iron stored by macrophages and microglia. Excessive free iron can be toxic, and accumulation of iron in MS has generally been thought to be detrimental. However, iron maintains the integrity of oligodendrocytes and myelin, and facilitates their regeneration following injury. The extracellular matrix, a key regulator of remyelination, might also modulate iron levels. This Review highlights key histological and MRI studies that have investigated changes in iron distribution associated with MS. Potential sources of iron, as well as iron regulatory proteins and the detrimental roles of excessive iron within the CNS, are also discussed, with emphasis on the importance of iron within cells for oxidative metabolism, proliferation and differentiation of oligodendrocytes, and myelination. In light of the beneficial and detrimental properties of iron within the CNS, we present considerations for treatments that target iron in MS. Such treatments must balance trophic and toxic properties of iron, by providing sufficient iron levels for remyelination and repair while avoiding excesses that might overwhelm homeostatic mechanisms and contribute to damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ropele, S. et al. MRI assessment of iron deposition in multiple sclerosis. J. Magn. Reson. Imaging 34, 13–21 (2011).
Chavhan, G. B., Babyn, P. S., Thomas, B., Shroff, M. M. & Haacke, E. M. Principles, techniques, and applications of T2*-based MR imaging and its special applications. Radiographics 29, 1433–1449 (2009).
Haider, L. et al. Multiple sclerosis deep grey matter: the relation between demyelination, neurodegeneration, inflammation and iron. J. Neurol. Neurosurg. Psychiatry http://dx.doi.com/10.1136/jnnp-2014-307712.
Walsh, A. et al. Multiple sclerosis: validation of MR imaging for quantification and detection of iron. Radiology 267, 531–542 (2013).
Zheng, W., Nichol, H., Liu, S., Cheng, Y.-C. & Haacke, E. M. Measuring iron in the brain using quantitative susceptibility mapping and X-ray fluorescence imaging. Neuroimage 78, 68–74 (2013).
Yao, B. et al. Chronic multiple sclerosis lesions: characterization with high-field-strength MR imaging. Radiology 262, 206–215 (2012).
Bagnato, F. et al. Tracking iron in multiple sclerosis: a combined imaging and histopathological study at 7 Tesla. Brain 134, 3602–3615 (2011).
Reichard, P. & Ehrenberg, A. Ribonucleotide reductase—a radical enzyme. Science 221, 514–519 (1983).
Outten, F. W. & Theil, E. C. Iron-based redox switches in biology. Antioxid. Redox Signal. 11, 1029–1046 (2009).
Núñez, M. T. et al. Iron toxicity in neurodegeneration. Biometals 25, 761–776 (2012).
Koskenkorva-Frank, T., Weiss, G., Koppenol, W. & Burckhardt, S. The complex interplay of iron metabolism, reactive oxygen species, and reactive nitrogen species: insights into the potential of various iron therapies to induce oxidative and nitrosative stress. Free Radic. Biol. Med. 65, 1174–1194 (2013).
Fridovich, I. Fundamental aspects of reactive oxygen species, or what's the matter with oxygen? Ann. N. Y. Acad. Sci. 893, 13–18 (1999).
Valko, M., Rhodes, C. J., Moncol, J., Izakovic, M. & Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 160, 1–40 (2006).
Esposito, B. P. et al. Labile plasma iron in iron overload: redox activity and susceptibility to chelation. Blood 102, 2670–2677 (2003).
Li, H., Swiercz, R. & Englander, E. W. Elevated metals compromise repair of oxidative DNA damage via the base excision repair pathway: implications of pathologic iron overload in the brain on integrity of neuronal DNA. J. Neurochem. 110, 1774–1783 (2009).
Yakes, F. & Van Houten, B. Mitochondrial DNA damage is more extensive and persists longer than nuclear DNA damage in human cells following oxidative stress. Proc. Natl Acad. Sci. USA 94, 514–519 (1997).
Shen, G. X. Oxidative stress and diabetic cardiovascular disorders: roles of mitochondria and NADPH oxidase. Can. J. Physiol. Pharmacol. 88, 241–248 (2010).
Mahad, D., Ziabreva, I., Lassmann, H. & Turnbull, D. Mitochondrial defects in acute multiple sclerosis lesions. Brain 131, 1722–1735 (2008).
Trapp, B. D. & Stys, P. K. Virtual hypoxia and chronic necrosis of demyelinated axons in multiple sclerosis. Lancet Neurol. 8, 280–291 (2009).
Eaton, J. W. & Qian, M. Molecular bases of cellular iron toxicity. Free Radic. Biol. Med. 32, 833–840 (2002).
Kress, G. J., Dineley, K. E. & Reynolds, I. J. The relationship between intracellular free iron and cell injury in cultured neurons, astrocytes, and oligodendrocytes. J. Neurosci. 22, 5848–5855 (2002).
Iwata-Ichikawa, E., Kondo, Y., Miyazaki, I., Asanuma, M. & Ogawa, N. Glial cells protect neurons against oxidative stress via transcriptional up-regulation of the glutathione synthesis. J. Neurochem. 72, 2334–2344 (1999).
Makar, T. et al. Vitamin E, ascorbate, glutathione, glutathione disulfide, and enzymes of glutathione metabolism in cultures of chick astrocytes and neurons: evidence that astrocytes play an important role in antioxidative processes in the brain. J. Neurochem. 62, 45–53 (1994).
Thorburne, S. K. & Juurlink, B. H. Low glutathione and high iron govern the susceptibility of oligodendroglial precursors to oxidative stress. J. Neurochem. 67, 1014–1022 (1996).
Benkovic, S. A. & Connor, J. R. Ferritin, transferrin, and iron in selected regions of the adult and aged rat brain. J. Comp. Neurol. 338, 97–113 (1993).
Connor, J. R. & Menzies, S. L. Relationship of iron to oligodendrocytes and myelination. Glia 17, 83–93 (1996).
Connor, J. R., Pavlick, G., Karli, D., Menzies, S. L. & Palmer, C. A histochemical study of iron-positive cells in the developing rat brain. J. Comp. Neurol. 355, 111–123 (1995).
Juurlink, B. H. Response of glial cells to ischemia: roles of reactive oxygen species and glutathione. Neurosci. Biobehav. Rev. 21, 151–166 (1997).
Matute, C., Sánchez-Gómez, M. V., Martínez-Millán, L. & Miledi, R. Glutamate receptor-mediated toxicity in optic nerve oligodendrocytes. Proc. Natl Acad. Sci. USA 94, 8830–8835 (1997).
Yu, J. et al. Iron is a potential key mediator of glutamate excitotoxicity in spinal cord motor neurons. Brain Res. 1257, 102–107 (2009).
McGahan, M. C. et al. Iron alters glutamate secretion by regulating cytosolic aconitase activity. Am. J. Physiol. Cell Physiol. 288, C1117–C1124 (2005).
Káradóttir, R., Cavelier, P., Bergersen, L. H. & Attwell, D. NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 438, 1162–1166 (2005).
Cui, Q.-L. et al. Oligodendrocyte progenitor cell susceptibility to injury in multiple sclerosis. Am. J. Pathol. 183, 516–525 (2013).
Hametner, S. et al. Iron and neurodegeneration in the multiple sclerosis brain. Ann. Neurol. 74, 848–861 (2013).
Durafourt, B. A. et al. Comparison of polarization properties of human adult microglia and blood-derived macrophages. Glia 60, 717–727 (2012).
Mills, C. D., Kincaid, K., Alt, J. M., Heilman, M. J. & Hill, A. M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J. Immunol. 164, 6166–6173 (2000).
Sindrilaru, A. et al. An unrestrained proinflammatory M1 macrophage population induced by iron impairs wound healing in humans and mice. J. Clin. Invest. 121, 985–997 (2011).
Mehta, V. et al. Iron is a sensitive biomarker for inflammation in multiple sclerosis lesions. PLoS ONE 8, e57573 (2013).
Friese, M. A., Schattling, B. & Fugger, L. Mechanisms of neurodegeneration and axonal dysfunction in multiple sclerosis. Nat. Rev. Neurol. 10, 225–238 (2014).
Bamm, V. V. & Harauz, G. Hemoglobin as a source of iron overload in multiple sclerosis: does multiple sclerosis share risk factors with vascular disorders? Cell. Mol. Life Sci. 71, 1789–1798 (2014).
Adams, C. W. Perivascular iron deposition and other vascular damage in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 51, 260–265 (1988).
Balla, J. et al. Heme, heme oxygenase and ferritin in vascular endothelial cell injury. Mol. Nutr. Food Res. 49, 1030–1043 (2005).
Nathoo, N. et al. Susceptibility-weighted imaging in the experimental autoimmune encephalomyelitis model of multiple sclerosis indicates elevated deoxyhemoglobin, iron deposition and demyelination. Mult. Scler. 19, 721–731 (2013).
Williams, R. et al. Iron deposition is independent of cellular inflammation in a cerebral model of multiple sclerosis. BMC Neurosci. 12, 59 (2011).
Cornelia, S. et al. Oxidative tissue injury in multiple sclerosis is only partly reflected in experimental disease models. Acta Neuropathol. http://dx.doi.org/10.1007/s00401-014-1263-5.
Cammer, W. in Oligodendroglia (ed. Norton, W. T.) 199–232 (Springer, 1984). [Series Ed. Agranoff, B. W. & Suzuki, K. Advances in Neurochemistry Vol. 5].
Chang, A. et al. Cortical remyelination: a new target for repair therapies in multiple sclerosis. Ann. Neurol. 72, 918–926 (2012).
Gille, G. & Reichmann, H. Iron-dependent functions of mitochondria—relation to neurodegeneration. J. Neural Transm. 118, 349–359 (2011).
Pleasure, D. & Kim, S. U. Enzyme markers for myelination of mouse cerebellum in vivo and in tissue culture. Brain Res. 104, 193–196 (1976).
Saher, G., et al. High cholesterol level is essential for myelin membrane growth. Nat. Neurosci. 8, 468–475 (2005).
Lange, S. J. & Que, L. Jr. Oxygen activating nonheme iron enzymes. Curr. Opin. Chem. Biol. 2, 159–172 (1998).
Todorich, B., Pasquini, J. M., Garcia, C. I., Paez, P. M. & Connor, J. R. Oligodendrocytes and myelination: the role of iron. Glia 57, 467–478 (2009).
Schonberg, D. L. & McTigue, D. M. Iron is essential for oligodendrocyte genesis following intraspinal macrophage activation. Exp. Neurol. 218, 64–74 (2009).
Schulz, K., Kroner, A. & David, S. Iron efflux from astrocytes plays a role in remyelination. J. Neurosci. 32, 4841–4847 (2012).
Morath, D. J. & Mayer-Proschel, M. Iron modulates the differentiation of a distinct population of glial precursor cells into oligodendrocytes. Dev. Biol. 237, 232–243 (2001).
Beard, J. L., Wiesinger, J. A. & Connor, J. R. Pre- and postweaning iron deficiency alters myelination in Sprague–Dawley rats. Dev. Neurosci. 25, 308–315 (2003).
Lieblein-Boff, J. C. et al. Neonatal E. coli infection causes neuro-behavioral deficits associated with hypomyelination and neuronal sequestration of iron. J. Neurosci. 33, 16334–16345 (2013).
Badaracco, M. E., Siri, M. V. & Pasquini, J. M. Oligodendrogenesis: the role of iron. Biofactors 36, 98–102 (2010).
Arnett, H. A. et al. TNFα promotes proliferation of oligodendrocyte progenitors and remyelination. Nat. Neurosci. 4, 1116–1122 (2001).
Mason, J. L., Suzuki, K., Chaplin, D. D. & Matsushima, G. K. Interleukin-1β promotes repair of the CNS. J. Neurosci. 21, 7046–7052 (2001).
Mason, J. L., Ye, P., Suzuki, K., D'Ercole, A. J. & Matsushima, G. K. Insulin-like growth factor-1 inhibits mature oligodendrocyte apoptosis during primary demyelination. J. Neurosci. 20, 5703–5708 (2000).
Zeger, M. et al. Insulin-like growth factor type 1 receptor signaling in the cells of oligodendrocyte lineage is required for normal in vivo oligodendrocyte development and myelination. Glia 55, 400–411 (2007).
Zhang, X., Surguladze, N., Slagle-Webb, B., Cozzi, A. & Connor, J. R. Cellular iron status influences the functional relationship between microglia and oligodendrocytes. Glia 54, 795–804 (2006).
Schonberg, D. L. et al. Ferritin stimulates oligodendrocyte genesis in the adult spinal cord and can be transferred from macrophages to NG2 cells in vivo. J. Neurosci. 32, 5374–5384 (2012).
Cairo, G., Recalcati, S., Mantovani, A. & Locati, M. Iron trafficking and metabolism in macrophages: contribution to the polarized phenotype. Trends Immunol. 32, 241–247 (2011).
Corna, G. et al. Polarization dictates iron handling by inflammatory and alternatively activated macrophages. Haematologica 95, 1814–1822 (2010).
Orino, K., et al. Ferritin and the response to oxidative stress. Biochem. J. 357, 241–247 (2001).
Connor, J. R. & Benkovic, S. A. Iron regulation in the brain: histochemical, biochemical, and molecular considerations. Ann. Neurol. 32 (Suppl.), S51–S61 (1992).
Benkovic, S. A. & Connor, J. R. Ferritin, transferrin, and iron in selected regions of the adult and aged rat brain. J. Comp. Neurol. 338, 97–113 (1993).
Brissot, P., Ropert, M., Le Lan, C. & Loréal, O. Non-transferrin bound iron: a key role in iron overload and iron toxicity. Biochimic. Biophys. Acta 1820, 403–410 (2012).
Petrat, F., de Groot, H. & Rauen, U. Subcellular distribution of chelatable iron: a laser scanning microscopic study in isolated hepatocytes and liver endothelial cells. Biochem. J. 356, 61–69 (2001).
Moos, T. Brain iron homeostasis. Dan. Med. Bull. 49, 279–301 (2002).
Rouault, T. Iron metabolism in the CNS: implications for neurodegenerative diseases. Nat. Rev. Neurosci. 14, 551–564 (2013).
Schulz, K., Vulpe, C. D., Harris, L. Z. & David, S. Iron efflux from oligodendrocytes is differentially regulated in gray and white matter. J. Neurosci. 31, 13301–13311 (2011).
Rouault, T. A. The role of iron regulatory proteins in mammalian iron homeostasis and disease. Nat. Chem. Biol. 2, 406–414 (2006).
Peyssonnaux, C., Nizet, V. & Johnson, R. S. Role of the hypoxia inducible factors HIF in iron metabolism. Cell Cycle 7, 28–32 (2008).
Rouault, T. A. & Cooperman, S. Brain iron metabolism. Semin. Pediatr. Neurol. 13, 142–148 (2006).
Anderson, C. P., Shen, M., Eisenstein, R. S. & Leibold, E. A. Mammalian iron metabolism and its control by iron regulatory proteins. Biochim. Biophys. Acta 1823, 1468–1483 (2012).
Lau, L. W., Cua, R., Keough, M. B., Haylock-Jacobs, S. & Yong, V. W. Pathophysiology of the brain extracellular matrix: a new target for remyelination. Nat. Rev. Neurosci. 14, 722–729 (2013).
Morawski, M. et al. Involvement of perineuronal and perisynaptic extracellular matrix in Alzheimer's disease neuropathology. Brain Pathol. 22, 547–561 (2012).
Sayed, R., Mubarak, W., Ohtsuka, A., Taguchi, T. & Murakami, T. Histochemical study of perineuronal nets in the retrosplenial cortex of adult rats. Ann. Anat. 184, 333–339 (2002).
Morawski, M., Brückner, M., Riederer, P., Brückner, G. & Arendt, T. Perineuronal nets potentially protect against oxidative stress. Exp. Neurol. 188, 309–315 (2004).
Bonneh-Barkay, D. & Wiley, C. A. Brain extracellular matrix in neurodegeneration. Brain Pathol. 19, 573–585 (2009).
Fiedler, A. et al. Intracellular iron concentration of neurons with and without perineuronal nets. Nucl. Instrum. Methods Phys. Res. B 260, 153–158 (2007).
Suttkus, A., Rohn, S., Jäger, C., Arendt, T. & Morawski, M. Neuroprotection against iron-induced cell death by perineuronal nets—an in vivo analysis of oxidative stress. Am. J. Neurodegener. Dis. 1, 122–129 (2012).
Suttkus, A. et al. Aggrecan, link protein and tenascin-R are essential components of the perineuronal net to protect neurons against iron-induced oxidative stress. Cell Death Dis. 5, e1119 (2014).
Sobel, R. A. & Ahmed, A. S. White matter extracellular matrix chondroitin sulfate/dermatan sulfate proteoglycans in multiple sclerosis. J. Neuropathol. Exp. Neurol. 60, 1198–1207 (2001).
Bekku, Y. et al. Bral1: its role in diffusion barrier formation and conduction velocity in the CNS. J. Neurosci. 30, 3113–3123 (2010).
Lau, L. W. et al. Chondroitin sulfate proteoglycans in demyelinated lesions impair remyelination. Ann. Neurol. 72, 419–432 (2012).
Chun, S. J., Rasband, M. N., Sidman, R. L., Habib, A. A. & Vartanian, T. Integrin-linked kinase is required for laminin-2-induced oligodendrocyte cell spreading and CNS myelination. J. Cell Biol. 163, 397–408 (2003).
Colognato, H., ffrench-Constant, C. & Feltri, M. L. Human diseases reveal novel roles for neural laminins. Trends Neurosci. 28, 480–486 (2005).
Nakanishi, K., Ito, M., Sato, Y. & Oohira, A. A highly-sulfated chondroitin sulfate, CS-E, adsorbs specifically to neurons with nuclear condensation. Neurosci. Res. 74, 223–229 (2012).
Grant, S. M., Wiesinger, J. A., Beard, J. L. & Cantorna, M. T. Iron-deficient mice fail to develop autoimmune encephalomyelitis. J. Nutr. 133, 2635–2638 (2003).
Brock, J. H. & Stevenson, J. Replacement of transferrin in serum-free cultures of mitogen-stimulated mouse lymphocytes by a lipophilic iron chelator. Immunol. Lett. 15, 23–25 (1987).
Kuvibidila, S., Dardenne, M., Savino, W. & Lepault, F. Influence of iron-deficiency anemia on selected thymus functions in mice: thymulin biological activity, T-cell subsets, and thymocyte proliferation. Am. J. Clin. Nutr. 51, 228–232 (1990).
Semenza, G. L. HIF-1: mediator of physiological and pathophysiological responses to hypoxia. J. Appl. Physiol. 88, 1474–1480 (2000).
Peyssonnaux, C. et al. Regulation of iron homeostasis by the hypoxia-inducible transcription factors (HIFs). J. Clin. Invest. 117, 1926–1932 (2007).
Lynch, S. G., Peters, K. & LeVine, S. M. Desferrioxamine in chronic progressive multiple sclerosis: a pilot study. Mult. Scler. 2, 157–160 (1996).
Lynch, S. G., Fonseca, T. & LeVine, S. M. A multiple course trial of desferrioxamine in chronic progressive multiple sclerosis. Cell. Mol. Biol. (Noisy-le-grand) 46, 865–869 (2000).
Norstrand, I. F. & Craelius, W. A trial of deferoxamine (Desferal) in the treatment of multiple sclerosis. A pilot study. Clin. Trials J. 26, 365–369 (1989).
Weigel, K. J., Lynch, S. G. & LeVine, S. M. Iron chelation and multiple sclerosis. ASN Neuro. 6, e00136 (2014).
Créange, A., Lefaucheur, J. P., Balleyguier, M. O. & Galactéros, F. Iron depletion induced by bloodletting and followed by rhEPO administration as a therapeutic strategy in progressive multiple sclerosis: a pilot, open-label study with neurophysiological measurements. Neurophysiol. Clin. 43, 303–312 (2013).
Lassmann, H., van Horssen, J. & Mahad, D. Progressive multiple sclerosis: pathology and pathogenesis. Nat. Rev. Neurol. 8, 647–656 (2012).
Mirshafiey, A. & Mohsenzadegan, M. Antioxidant therapy in multiple sclerosis. Immunopharmacol. Immunotoxicol. 31, 13–29 (2009).
Schwarz, S. & Leweling, H. Multiple sclerosis and nutrition. Mult. Scler. 11, 24–32 (2005).
Zhang, S. M. et al. Intakes of carotenoids, vitamin C, and vitamin E and MS risk among two large cohorts of women. Neurology 57, 75–80 (2001).
Halliwell, B. & Gutteridge, J. The importance of free radicals and catalytic metal ions in human diseases. Mol. Aspects Med. 8, 89–193 (1985).
Kell, D. B. Iron behaving badly: inappropriate iron chelation as a major contributor to the aetiology of vascular and other progressive inflammatory and degenerative diseases. BMC Med. Genomics 2, 2 (2009).
Youdim, M. B., Ben-Shachar, D. & Riederer, P. The possible role of iron in the etiopathology of Parkinson's disease. Mov. Disord. 8, 1–12 (1993).
Berg, D., Becker, G., Riederer, P. & Riess, O. Iron in neurodegenerative disorders. Neurotox. Res. 4, 637–653 (2002).
Balla, J. et al. Haem, haem oxygenase and ferritin in vascular endothelial cell injury. Nephrol. Dial. Transplant. 18 (Suppl. 5), v8–v12 (2003).
Hammond, K. E. et al. Quantitative in vivo magnetic resonance imaging of multiple sclerosis at 7 Tesla with sensitivity to iron. Ann. Neurol. 64, 707–713 (2008).
Walsh, A. J. et al. Longitudinal MR imaging of iron in multiple sclerosis: an imaging marker of disease. Radiology 270, 186–196 (2014).
Khalil, M. et al. Determinants of brain iron in multiple sclerosis: a quantitative 3 T MRI study. Neurology 77, 1691–1697 (2011).
Al-Radaideh, A. et al. Increased iron accumulation occurs in the earliest stages of demyelinating disease: an ultra-high field susceptibility mapping study in clinically isolated syndrome. Mult. Scler. 19, 896–903 (2013).
Lebel, R. M., Eissa, A., Seres, P., Blevins, G. & Wilman, A. H. Quantitative high-field imaging of sub-cortical gray matter in multiple sclerosis. Mult. Scler. 18, 433–441 (2012).
Khalil, M. et al. Quantitative assessment of brain iron by R2* relaxometry in patients with clinically isolated syndrome and relapsing–remitting multiple sclerosis. Mult. Scler. 15, 1048–1054 (2009).
Zivadinov, R. et al. Abnormal subcortical deep-gray matter susceptibility-weighted imaging filtered phase measurements in patients with multiple sclerosis: a case–control study. Neuroimage 59, 331–339 (2012).
Neema, M. et al. Deep gray matter involvement on brain MRI scans is associated with clinical progression in multiple sclerosis. J. Neuroimaging 19, 3–8 (2009).
Paling, D. et al. Reduced R2′ in multiple sclerosis normal appearing white matter and lesions may reflect decreased myelin and iron content. J. Neurol. Neurosurg. Psychiatry 83, 785–792 (2012).
Forge, J. K., Pedchenko, T. V. & LeVine, S. M. Iron deposits in the central nervous system of SJL mice with experimental allergic encephalomyelitis. Life Sci. 63, 2271–2284 (1998).
Acknowledgements
The authors' studies have been funded by the Canadian Institutes of Health Research, the Multiple Sclerosis Society of Canada, and the Alberta Innovates–Health Solutions CRIO Team programme.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, made substantial contributions to discussions of the content, and wrote and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Stephenson, E., Nathoo, N., Mahjoub, Y. et al. Iron in multiple sclerosis: roles in neurodegeneration and repair. Nat Rev Neurol 10, 459–468 (2014). https://doi.org/10.1038/nrneurol.2014.118
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2014.118
This article is cited by
-
Roles and regulation of microglia activity in multiple sclerosis: insights from animal models
Nature Reviews Neuroscience (2023)
-
Ferroptosis contributes to multiple sclerosis and its pharmacological targeting suppresses experimental disease progression
Cell Death & Differentiation (2023)
-
Potentially toxic elements in the brains of people with multiple sclerosis
Scientific Reports (2023)
-
Glial Cell Metabolic Profile Upon Iron Deficiency: Oligodendroglial and Astroglial Casualties of Bioenergetic Adjustments
Molecular Neurobiology (2023)
-
Iron Overload Causes Ferroptosis But Not Apoptosis in MO3.13 Oligodendrocytes
Neurochemical Research (2023)