Abstract
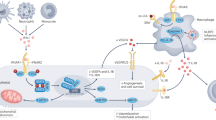
Rapid-onset cardiovascular disease (CVD) is a major concern for many patients with systemic lupus erythematosus (SLE). Cardiovascular events occur more frequently and with earlier onset in patients with SLE compared with healthy individuals. Traditional risk factors, such as altered lipid levels, aging and smoking, do not fully explain this increased risk of CVD, strongly suggesting that autoimmunity contributes to accelerated atherosclerosis. Altered immune system function is recognized as the primary contributor to both the initiation and progression of atherosclerosis. Multiple manifestations of autoimmunity, including changes in cytokine levels and innate immune responses, autoantibodies, adipokines, dysfunctional lipids, and oxidative stress, could heighten atherosclerotic risk. In addition, multiple SLE therapeutics seem to affect the development and progression of atherosclerosis both positively and negatively. SLE-specific cardiovascular risk factors are beginning to be discovered by several groups, and development of a comprehensive, clinically feasible biomarker panel could be invaluable for identification and treatment of patients at risk of developing accelerated atherosclerosis. Here, we discuss the epidemiology of CVD in SLE and the implications of immune system dysfunction on the development and progression, monitoring and treatment of atherosclerosis in individuals with this disease.
Key Points
-
Cardiovascular disease (CVD) is a substantial contributor to morbidity and mortality in patients with systemic lupus erythematosus (SLE)
-
SLE-specific risk factors for accelerated atherosclerosis exist, but are poorly understood
-
Endothelial cell dysfunction plays a major part in accelerated atherosclerosis in patients with SLE
-
Identification of SLE-specific mechanisms of, and biomarkers, for accelerated atherosclerosis should lead to the development of novel screening protocols for early detection of CVD and discovery of new therapeutic targets
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Libby, P. Inflammation in atherosclerosis. Nature 420, 868–874 (2002).
Urowitz, M. B. et al. The bimodal mortality pattern of systemic lupus erythematosus. Am. J. Med. 60, 221–225 (1976).
Nossent, J. et al. Current causes of death in systemic lupus erythematosus in Europe, 2000–2004: relation to disease activity and damage accrual. Lupus 16, 309–317 (2007).
Esdaile, J. M. et al. Traditional Framingham risk factors fail to fully account for accelerated atherosclerosis in systemic lupus erythematosus. Arthritis Rheum. 44, 2331–2337 (2001).
Manzi, S. et al. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am. J. Epidemiol. 145, 408–415 (1997).
Roman, M. J. et al. Prevalence and correlates of accelerated atherosclerosis in systemic lupus erythematosus. N. Engl. J. Med. 349, 2399–2406 (2003).
Roman, M. J. et al. Rate and determinants of progression of atherosclerosis in systemic lupus erythematosus. Arthritis Rheum. 56, 3412–3419 (2007).
Manzi, S. et al. Prevalence and risk factors of carotid plaque in women with systemic lupus erythematosus. Arthritis Rheum. 42, 51–60 (1999).
Asanuma, Y. et al. Premature coronary-artery atherosclerosis in systemic lupus erythematosus. N. Engl. J. Med. 349, 2407–2415 (2003).
Bruce, I. N., Burns, R. J., Gladman, D. D. & Urowitz, M. B. Single photon emission computed tomography dual isotope myocardial perfusion imaging in women with systemic lupus erythematosus. I. Prevalence and distribution of abnormalities. J. Rheumatol. 27, 2372–2377 (2000).
El-Magadmi, M. et al. Systemic lupus erythematosus: an independent risk factor for endothelial dysfunction in women. Circulation 110, 399–404 (2004).
Recio-Mayoral, A. et al. Chronic inflammation and coronary microvascular dysfunction in patients without risk factors for coronary artery disease. Eur. Heart J. 30, 1837–1843 (2009).
Folsom, A. R. et al. Coronary artery calcification compared with carotid intima-media thickness in the prediction of cardiovascular disease incidence: the Multi-Ethnic Study of Atherosclerosis (MESA). Arch. Intern. Med. 168, 1333–1339 (2008).
Salel, A. F. et al. Accuracy of numerical coronary profile. Correlation of risk factors with arteriographically documented severity of atherosclerosis. N. Engl. J. Med. 296, 1447–1450 (1977).
Moore, K. J. & Tabas, I. Macrophages in the pathogenesis of atherosclerosis. Cell 145, 341–355 (2011).
Rho, Y. H. et al. Macrophage activation and coronary atherosclerosis in systemic lupus erythematosus and rheumatoid arthritis. Arthritis Care Res. (Hoboken) 63, 535–541 (2011).
Shah, P. K. et al. High-dose recombinant apolipoprotein A-I(milano) mobilizes tissue cholesterol and rapidly reduces plaque lipid and macrophage content in apolipoprotein E-deficient mice. Potential implications for acute plaque stabilization. Circulation 103, 3047–3050 (2001).
Wright, S. A. et al. Microcirculatory hemodynamics and endothelial dysfunction in systemic lupus erythematosus. Arterioscler. Thromb. Vasc. Biol. 26, 2281–2287 (2006).
Kahlenberg, J. M. & Kaplan, M. J. The interplay of inflammation and cardiovascular disease in systemic lupus erythematosus. Arthritis Res. Ther. 13, 203 (2011).
Rajagopalan, S. et al. Endothelial cell apoptosis in systemic lupus erythematosus: a common pathway for abnormal vascular function and thrombosis propensity. Blood 103, 3677–3683 (2004).
Denny, M. F. et al. Interferon-α promotes abnormal vasculogenesis in lupus: a potential pathway for premature atherosclerosis. Blood 110, 2907–2915 (2007).
Denny, M. F. et al. A distinct subset of proinflammatory neutrophils isolated from patients with systemic lupus erythematosus induces vascular damage and synthesizes type I IFNs. J. Immunol. 184, 3284–3297 (2010).
Villanueva, E. et al. Netting neutrophils induce endothelial damage, infiltrate tissues, and expose immunostimulatory molecules in systemic lupus erythematosus. J. Immunol. 187, 538–552 (2011).
Gualtierotti, R., Biggioggero, M. & Meroni, P. L. Cutting-edge issues in coronary disease and the primary antiphospholipid syndrome. Clin. Rev. Allergy Immunol. http://dx.doi.org/10.1007/s12016-011-8268–9.
Ames, P. R., Margarita, A., Sokoll, K. B., Weston, M. & Brancaccio, V. Premature atherosclerosis in primary antiphospholipid syndrome: preliminary data. Ann. Rheum. Dis. 64, 315–317 (2005).
Wu, R. et al. Antibodies against cardiolipin and oxidatively modified LDL in 50-year-old men predict myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 17, 3159–3163 (1997).
Vaarala, O. et al. Anti-cardiolipin antibodies and risk of myocardial infarction in a prospective cohort of middle-aged men. Circulation 91, 23–27 (1995).
Petri, M. in Dubois' Lupus Erythematosus, 7th edn (eds Wallace, D. J. & Hahn, B. H.) 1262–1297 (Lippincott Williams & Wilkins, Philadelphia, 2007).
Toloza, S. M. et al. Systemic lupus erythematosus in a multiethnic US cohort (LUMINA). XXIII. Baseline predictors of vascular events. Arthritis Rheum. 50, 3947–57 (2004).
McMahon, M. et al. Dysfunctional proinflammatory high-density lipoproteins confer increased risk of atherosclerosis in women with systemic lupus erythematosus. Arthritis Rheum. 60, 2428–2437 (2009).
Pengo, V., Bison, E., Ruffatti, A. & Iliceto, S. Antibodies to oxidized LDL/β2-glycoprotein I in antiphospholipid syndrome patients with venous and arterial thromboembolism. Thromb. Res. 122, 556–559 (2008).
Meroni, P. L., Raschi, E., Testoni, C. & Borghi, M. O. Endothelial cell activation by antiphospholipid antibodies. Clin. Immunol. 112, 169–174 (2004).
Hansson, G. K. & Hermansson, A. The immune system in atherosclerosis. Nat. Immunol. 12, 204–212 (2011).
Fesmire, J., Wolfson-Reichlin, M. & Reichlin, M. Effects of autoimmune antibodies anti-lipoprotein lipase, anti-low density lipoprotein, and anti-oxidized low density lipoprotein on lipid metabolism and atherosclerosis in systemic lupus erythematosus. Rev. Bras. Reumatol. 50, 539–551 (2010).
Vuilleumier, N. et al. Presence of autoantibodies to apolipoprotein A-1 in patients with acute coronary syndrome further links autoimmunity to cardiovascular disease. J. Autoimmun. 23, 353–360 (2004).
Dinu, A. R. et al. Frequency of antibodies to the cholesterol transport protein apolipoprotein A1 in patients with SLE. Lupus 7, 355–360 (1998).
O'Neill, S. G. et al. Antibodies to apolipoprotein A-I, high-density lipoprotein, and C-reactive protein are associated with disease activity in patients with systemic lupus erythematosus. Arthritis Rheum. 62, 845–854 (2010).
Ait-Oufella, H., Taleb, S., Mallat, Z. & Tedgui, A. Recent advances on the role of cytokines in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 31, 969–979 (2011).
Hansson, G. K., Jonasson, L., Holm, J., Clowes, M. M. & Clowes, A. W. γ-interferon regulates vascular smooth muscle proliferation and Ia antigen expression in vivo and in vitro. Circ. Res. 63, 712–719 (1988).
McLaren, J. E. & Ramji, D. P. Interferon γ: a master regulator of atherosclerosis. Cytokine Growth Factor Rev. 20, 125–135 (2009).
Uyemura, K. et al. Cross-regulatory roles of interleukin (IL)-12 and IL-10 in atherosclerosis. J. Clin. Invest. 97, 2130–2138 (1996).
Hansson, G. K. Immune mechanisms in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 21, 1876–1890 (2001).
Svenungsson, E. et al. TNF-α: a link between hypertriglyceridaemia and inflammation in SLE patients with cardiovascular disease. Lupus 12, 454–461 (2003).
Rho, Y. H. et al. Novel cardiovascular risk factors in premature coronary atherosclerosis associated with systemic lupus erythematosus. J. Rheumatol. 35, 1789–1794 (2008).
Thacker, S. G. et al. The detrimental effects of IFN-α on vasculogenesis in lupus are mediated by repression of IL-1 pathways: potential role in atherogenesis and renal vascular rarefaction. J. Immunol. 185, 4457–4469 (2010).
Asanuma, Y. et al. Increased concentration of proatherogenic inflammatory cytokines in systemic lupus erythematosus: relationship to cardiovascular risk factors. J. Rheumatol. 33, 539–545 (2006).
Sabio, J. M. et al. Metabolic syndrome is associated with increased arterial stiffness and biomarkers of subclinical atherosclerosis in patients with systemic lupus erythematosus. J. Rheumatol. 36, 2204–2211 (2009).
Rua-Figueroa, I. et al. Factors involved in the progress of preclinical atherosclerosis associated with systemic lupus erythematosus: a 2-year longitudinal study. Ann. Rheum. Dis. 69, 1136–1139 (2010).
Shin, M. S., Lee, N. & Kang, I. Effector T-cell subsets in systemic lupus erythematosus: update focusing on TH17 cells. Curr. Opin. Rheumatol. 23, 444–448 (2011).
von Vietinghoff, S. et al. Mycophenolate mofetil decreases atherosclerotic lesion size by depression of aortic T-lymphocyte and interleukin-17-mediated macrophage accumulation. J. Am. Coll. Cardiol. 57, 2194–2204 (2011).
Marder, W. et al. Interleukin 17 as a novel predictor of vascular function in rheumatoid arthritis. Ann. Rheum. Dis. 70, 1550–1555 (2011).
Foks, A. C. et al. Differential effects of regulatory T cells on the initiation and regression of atherosclerosis. Atherosclerosis 218, 53–60 (2011).
Toma, I. & McCaffrey, T. A. Transforming growth factor-β and atherosclerosis: interwoven atherogenic and atheroprotective aspects. Cell Tissue Res. 347, 155–175 (2012).
Jackson, M., Ahmad, Y., Bruce, I. N., Coupes, B. & Brenchley, P. E. Activation of transforming growth factor-β1 and early atherosclerosis in systemic lupus erythematosus. Arthritis Res. Ther. 8, R81 (2006).
van Leuven, S. I. et al. Mycophenolate mofetil but not atorvastatin attenuates atherosclerosis in lupus-prone Ldlr−/− mice. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2011-200071.
Campbell, L. A. et al. The acute phase reactant response to respiratory infection with Chlamydia pneumoniae: implications for the pathogenesis of atherosclerosis. Microbes Infect. 12, 598–606 (2010).
Miller, Y. I. et al. Minimally modified LDL binds to CD14, induces macrophage spreading via TLR4/MD-2, and inhibits phagocytosis of apoptotic cells. J. Biol. Chem. 278, 1561–1568 (2003).
Huang, Q. & Pope, R. M. Toll-like receptor signaling: a potential link among rheumatoid arthritis, systemic lupus, and atherosclerosis. J. Leukoc. Biol. 88, 253–262 (2010).
Symmons, D. P. & Gabriel, S. E. Epidemiology of CVD in rheumatic disease, with a focus on RA and SLE. Nat. Rev. Rheumatol. 7, 399–408 (2011).
Von Feldt, J. M. et al. Homocysteine levels and disease duration independently correlate with coronary artery calcification in patients with systemic lupus erythematosus. Arthritis Rheum. 54, 2220–2227 (2006).
McMahon, M. et al. Proinflammatory high-density lipoprotein as a biomarker for atherosclerosis in patients with systemic lupus erythematosus and rheumatoid arthritis. Arthritis Rheum. 54, 2541–2549 (2006).
Charles-Schoeman, C. et al. Abnormal function of high-density lipoprotein is associated with poor disease control and an altered protein cargo in rheumatoid arthritis. Arthritis Rheum. 60, 2870–2879 (2009).
Charakida, M. et al. Vascular abnormalities, paraoxonase activity, and dysfunctional HDL in primary antiphospholipid syndrome. JAMA 302, 1210–1217 (2009).
Avalos, I. et al. Oxidative stress in systemic lupus erythematosus: relationship to disease activity and symptoms. Lupus 16, 195–200 (2007).
Malinow, M. R., Nieto, F. J., Szklo, M., Chambless, L. E. & Bond, G. Carotid artery intimal-medial wall thickening and plasma homocyst(e)ine in asymptomatic adults. The Atherosclerosis Risk in Communities Study. Circulation 87, 1107–1113 (1993).
Wall, R. T., Harlan, J. M., Harker, L. A. & Striker, G. E. Homocysteine-induced endothelial cell injury in vitro: a model for the study of vascular injury. Thromb. Res. 18, 113–121 (1980).
Hajjar, K. A. Homocysteine-induced modulation of tissue plasminogen activator binding to its endothelial cell membrane receptor. J. Clin. Invest. 91, 2873–2879 (1993).
Woo, K. S. et al. Hyperhomocyst(e)inemia is a risk factor for arterial endothelial dysfunction in humans. Circulation 96, 2542–2544 (1997).
Upchurch, G. R. Jr. et al. Homocyst(e)ine decreases bioavailable nitric oxide by a mechanism involving glutathione peroxidase. J. Biol. Chem. 272, 17012–17017 (1997).
McCully, K. S. Homocysteine and vascular disease. Nat. Med. 2, 386–389 (1996).
Petri, M. Detection of coronary artery disease and the role of traditional risk factors in the Hopkins Lupus Cohort. Lupus 9, 170–175 (2000).
Svenungsson, E. et al. Risk factors for cardiovascular disease in systemic lupus erythematosus. Circulation 104, 1887–1893 (2001).
Refai, T. M., Al-Salem, I. H., Nkansa-Dwamena, D. & Al-Salem, M. H. Hyperhomocysteinaemia and risk of thrombosis in systemic lupus erythematosus patients. Clin. Rheumatol. 21, 457–461 (2002).
Bruce, I. N., Urowitz, M. B., Gladman, D. D., Ibanˇez, D. & Steiner, G. Risk factors for coronary heart disease in women with systemic lupus erythematosus: the Toronto Risk Factor Study. Arthritis Rheum. 48, 3159–3167 (2003).
Manger, K. et al. Factors associated with coronary artery calcification in young female patients with SLE. Ann. Rheum. Dis. 62, 846–850 (2003).
Potter, K., Hankey, G. J., Green, D. J., Eikelboom, J. W. & Arnolda, L. F. Homocysteine or renal impairment: which is the real cardiovascular risk factor? Arterioscler. Thromb. Vasc. Biol. 28, 1158–1164 (2008).
Taleb, S. et al. Defective leptin/leptin receptor signaling improves regulatory T cell immune response and protects mice from atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 27, 2691–2698 (2007).
Garcia-Gonzalez, A. et al. Serum leptin levels in women with systemic lupus erythematosus. Rheumatol. Int. 22, 138–141 (2002).
Al, M. et al. Adipokines as novel biomarkers in paediatric systemic lupus erythematosus. Rheumatology (Oxford) 48, 497–501 (2009).
McMahon, M. et al. Plasma leptin levels are associated with carotid artery plaque and intima-media thickness (IMT) in women with SLE and a matched population of healthy women [abstract 2091]. Arthritis Rheum. 56 (Suppl.), S796 (2007).
Reynolds, H. R. et al. Association of plasma soluble E-selectin and adiponectin with carotid plaque in patients with systemic lupus erythematosus. Atherosclerosis 210, 569–574 (2010).
Chung, C. et al. Adipocytokines in systemic lupus erythematosus: relationship to inflammation, insulin resistance and coronary atherosclerosis. Lupus 18, 799–806 (2009).
Yazdany, J. et al. Provision of preventive health care in systemic lupus erythematosus: data from a large observational cohort study. Arthritis Res. Ther. 12, R84 (2010).
Mosca, M. et al. Development of quality indicators to evaluate the monitoring of SLE patients in routine clinical practice. Autoimmun. Rev. 10, 383–388 (2011).
Stone, N. J., Bilek, S. & Rosenbaum, S. Recent National Cholesterol Education Program Adult Treatment Panel III update: adjustments and options. Am. J. Cardiol. 96, 53–59 (2005).
Chobanian, A. V. et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA 289, 2560–2572 (2003).
Graham, I. et al. European guidelines on cardiovascular disease prevention in clinical practice: executive summary. Atherosclerosis 194, 1–45 (2007).
Costenbader, K. H. et al. Barriers to a trial of atherosclerosis prevention in systemic lupus erythematosus. Arthritis Rheum. 53, 718–723 (2005).
Duran-Barragan, S., McGwin, G. Jr, Vilá, L. M., Reveille, J. D. & Alarcón, G. S. Angiotensin-converting enzyme inhibitors delay the occurrence of renal involvement and are associated with a decreased risk of disease activity in patients with systemic lupus erythematosus—results from LUMINA (LIX): a multiethnic US cohort. Rheumatology (Oxford) 47, 1093–1096 (2008).
Peters, M. J. et al. EULAR evidence-based recommendations for cardiovascular risk management in patients with rheumatoid arthritis and other forms of inflammatory arthritis. Ann. Rheum. Dis. 69, 325–331 (2010).
Coffman, J. D. Raynaud's phenomenon. An update. Hypertension 17, 593–602 (1991).
Forrester, J. S. & Libby, P. The inflammation hypothesis and its potential relevance to statin therapy. Am. J. Cardiol. 99, 732–738 (2007).
Ferreira, G. A., Navarro, T. P., Telles, R. W., Andrade, L. E. & Sato, E. I. Atorvastatin therapy improves endothelial-dependent vasodilation in patients with systemic lupus erythematosus: an 8 weeks controlled trial. Rheumatology (Oxford) 46, 1560–1565 (2007).
Petri, M. A., Kiani, A. N., Post, W., Christopher-Stine, L. & Magder, L. S. Lupus Atherosclerosis Prevention Study (LAPS). Ann. Rheum. Dis. 70, 760–765 (2011).
Kang, S., Wu, Y. & Li, X. Effects of statin therapy on the progression of carotid atherosclerosis: a systematic review and meta-analysis. Atherosclerosis 177, 433–442 (2004).
Aprahamian, T. et al. Simvastatin treatment ameliorates autoimmune disease associated with accelerated atherosclerosis in a murine lupus model. J. Immunol. 177, 3028–3034 (2006).
Nord, J. E., Shah, P. K., Rinaldi, R. Z. & Weisman, M. H. Hydroxychloroquine cardiotoxicity in systemic lupus erythematosus: a report of 2 cases and review of the literature. Semin. Arthritis Rheum. 33, 336–351 (2004).
Selzer, F. et al. Vascular stiffness in women with systemic lupus erythematosus. Hypertension 37, 1075–1082 (2001).
Rahman, P. et al. The cholesterol lowering effect of antimalarial drugs is enhanced in patients with lupus taking corticosteroid drugs. J. Rheumatol. 26, 325–330 (1999).
Jung, H. et al. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis Rheum. 62, 863–868 (2010).
Sun, S., Rao, N. L., Venable, J., Thurmond, R. & Karlsson, L. TLR7/9 antagonists as therapeutics for immune-mediated inflammatory disorders. Inflamm. Allergy Drug Targets 6, 223–235 (2007).
Doria, A. et al. Risk factors for subclinical atherosclerosis in a prospective cohort of patients with systemic lupus erythematosus. Ann. Rheum. Dis. 62, 1071–1077 (2003).
Petri, M., Perez-Gutthann, S., Spence, D. & Hochberg, M. C. Risk factors for coronary artery disease in patients with systemic lupus erythematosus. Am. J. Med. 93, 513–519 (1992).
Bravo, Y., Quiroz, Y., Ferrebuz, A., Vaziri, N. D. & Rodríguez-Iturbe, B. Mycophenolate mofetil administration reduces renal inflammation, oxidative stress, and arterial pressure in rats with lead-induced hypertension. Am. J. Physiol. Renal Physiol. 293, F616–F623 (2007).
van Leuven, S. I. et al. Mycophenolate mofetil attenuates plaque inflammation in patients with symptomatic carotid artery stenosis. Atherosclerosis 211, 231–236 (2010).
David, K. M. et al. Mycophenolate mofetil vs. azathioprine is associated with decreased acute rejection, late acute rejection, and risk for cardiovascular death in renal transplant recipients with pre-transplant diabetes. Clin. Transplant. 19, 279–285 (2005).
Kiani, A. N., Magder, L. S. & Petri, M. Mycophenolate mofetil (MMF) does not slow the progression of subclinical atherosclerosis in SLE over 2 years. Rheumatol. Int. http://dx.doi.org/10.1007/s00296-011-2048-y.
Haque, S. et al. Risk factors for clinical coronary heart disease in systemic lupus erythematosus: the lupus and atherosclerosis evaluation of risk (LASER) study. J. Rheumatol. 37, 322–329 (2010).
Schanberg, L. E. et al. Premature atherosclerosis in pediatric systemic lupus erythematosus: risk factors for increased carotid intima-media thickness in the atherosclerosis prevention in pediatric lupus erythematosus cohort. Arthritis Rheum. 60, 1496–1507 (2009).
Caligiuri, G., Nicoletti, A., Poirier, B. & Hansson, G. K. Protective immunity against atherosclerosis carried by B cells of hypercholesterolemic mice. J. Clin. Invest. 109, 745–753 (2002).
Major, A. S., Fazio, S. & Linton, M. F. B-lymphocyte deficiency increases atherosclerosis in LDL receptor-null mice. Arterioscler. Thromb. Vasc. Biol. 22, 1892–1898 (2002).
Kyaw, T., Tipping, P., Toh, B. H. & Bobik, A. Current understanding of the role of B cell subsets and intimal and adventitial B cells in atherosclerosis. Curr. Opin. Lipidol. 22, 373–379 (2011).
Kyaw, T. et al. Conventional B2 B cell depletion ameliorates whereas its adoptive transfer aggravates atherosclerosis. J. Immunol. 185, 4410–4419 (2010).
Ait-Oufella, H. et al. B cell depletion reduces the development of atherosclerosis in mice. J. Exp. Med. 207, 1579–1587 (2010).
Pego-Reigosa, J. M. et al. Long-term improvement of lipid profile in patients with refractory systemic lupus erythematosus treated with B-cell depletion therapy: a retrospective observational study. Rheumatology (Oxford) 49, 691–696 (2010).
Van Lenten, B. J. et al. Apolipoprotein A-I mimetic peptides. Curr. Atheroscler. Rep. 11, 52–57 (2009).
Navab, M. et al. An oral apoJ peptide renders HDL antiinflammatory in mice and monkeys and dramatically reduces atherosclerosis in apolipoprotein E-null mice. Arterioscler. Thromb. Vasc. Biol. 25, 1932–1937 (2005).
Woo, J. M. et al. Treatment with apolipoprotein A-1 mimetic peptide reduces lupus-like manifestations in a murine lupus model of accelerated atherosclerosis. Arthritis Res. Ther. 12, R93 (2010).
Kaplan, M. J. & Salmon, J. E. How does interferon-α insult the vasculature? Let me count the ways. Arthritis Rheum. 63, 334–336 (2011).
Li, J. et al. Interferon- α priming promotes lipid uptake and macrophage-derived foam cell formation: a novel link between interferon-α and atherosclerosis in lupus. Arthritis Rheum. 63, 492–502 (2011).
Mosca, M. et al. European League Against Rheumatism recommendations for monitoring patients with systemic lupus erythematosus in clinical practice and in observational studies. Ann. Rheum. Dis. 69, 1269–1274 (2010).
Acknowledgements
We apologize to the many researchers whose relevant studies were not cited here owing to space constraints. Research pursuits of the authors were supported by funding from NIH/NIAMS (K01 AR-059,095-01 to B. J. Skaggs; K23 AR-053,864-01A1 to M. McMahon), the Arthritis National Research Foundation (B. J. Skaggs), the Arthritis Foundation, Pacific Region (B. J. Skaggs and M. McMahon), Rheuminations, Inc. (B. H. Hahn), Alliance for Lupus Research (B. H. Hahn and M. McMahon), and Lupus Research Institute (B. H. Hahn and M. McMahon). B. H. Hahn is also the recipient of a Kirkland Scholar award.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all stages of the preparation of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Skaggs, B., Hahn, B. & McMahon, M. Accelerated atherosclerosis in patients with SLE—mechanisms and management. Nat Rev Rheumatol 8, 214–223 (2012). https://doi.org/10.1038/nrrheum.2012.14
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2012.14
This article is cited by
-
Targeting proprotein convertase subtilisin/kexin type 9 (PCSK9): from bench to bedside
Signal Transduction and Targeted Therapy (2024)
-
Dysregulated endothelial cell markers in systemic lupus erythematosus: a systematic review and meta-analysis
Journal of Inflammation (2023)
-
Hyperlipidemia in immune thrombocytopenia: a retrospective study
Thrombosis Journal (2023)
-
A deeper look into the functions of right ventricle using three-dimensional echocardiography: the forgotten ventricle in children with systemic lupus erythematosus
European Journal of Pediatrics (2023)
-
Atherosclerosis in Systemic Lupus Erythematosus
Current Atherosclerosis Reports (2023)