Key Points
-
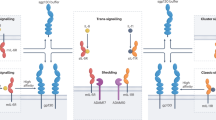
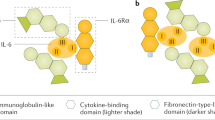
IL-6 can signal via the membrane-bound and the soluble IL-6 receptor (IL-6R); classic signalling via the membrane-bound receptor is regenerative and protects from bacterial infections, whereas trans-signalling via the soluble receptor is proinflammatory
-
The soluble gp130Fc fusion protein specifically blocks IL-6 trans-signalling without affecting classic signalling
-
Preclinical models strongly suggest the efficacy of IL-6-directed therapies for a variety of immunological conditions
-
The approval and use of tocilizumab, a first-in-class human monoclonal antibody directed at IL-6R, has demonstrated that this strategy is both highly effective and safe
-
New agents with unique bioengineering features targeting either IL-6 or the soluble IL-6R, with varying selectivity for classic signalling and trans-signalling pathways, are entering clinical trials and offer alternative strategies for IL-6-based therapies
-
Selective IL-6-based therapeutic targeting has several unique toxicity signatures, including paradoxical effects on surrogate markers of cardiovascular risk, and awaits clinical studies to determine net effects on morbidity and mortality
Abstract
IL-6 has been linked to numerous diseases associated with inflammation, including rheumatoid arthritis, inflammatory bowel disease, vasculitis and several types of cancer. Moreover, IL-6 is important in the induction of hepatic acute-phase proteins for the trafficking of acute and chronic inflammatory cells, the differentiation of adaptive T-cell responses, and tissue regeneration and homeostatic regulation. Studies have investigated IL-6 biology using cell-bound IL-6 receptors expressed predominantly on hepatocytes and certain haematopoietic cells versus activation mediated by IL-6 and soluble IL-6 receptors via a second protein, gp130, which is expressed throughout the body. Advances in this research elucidating the differential effects of IL-6 activation provide important insights into the role of IL-6 in health and disease, as well as its potential as a therapeutic target. Knowledge of the basic biology of IL-6 and its signalling pathways can better inform both the research agenda for IL-6-based targeted therapies as well as the clinical use of strategies affecting IL-6-mediated inflammation. This Review covers novel, emerging aspects of the biology of IL-6, which might lead to more specific blockade of IL-6 signalling without compromising the protective function of this cytokine in the body's defence against infections.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
19 September 2014
In the version of this article initially published online, findings from the Kuchroo et al. group were incorrectly reported in the 'Role in inflammation' section. This inaccuracy has been corrected for the HTML, PDF and print versions of the article.
References
Naka, T., Nishimoto, N. & Kishimoto, T. The paradigm of IL-6: from basic science to medicine. Arthritis Res. 4 (Suppl. 3), S233–S242 (2002).
Hirano, T. et al. Complementary DNA for a novel human interleukin (BSF-2) that induces B lymphocytes to produce immunoglobulin. Nature 324, 73–76 (1986).
Wolvekamp, M. C. & Marquet, R. L. Interleukin-6: historical background, genetics and biological significance. Immunol. Lett. 24, 1–9 (1990).
Nishimoto, N. Interleukin-6 as a therapeutic target in candidate inflammatory diseases. Clin. Pharmacol. Ther. 87, 483–487 (2006).
Cronstein, B. N. Interleukin-6—a key mediator of systemic and local symptoms in rheumatoid arthritis. Bull. NYU Hosp. Jt Dis. 65 (Suppl. 1), S11–S15 (2007).
Rose-John, S. IL-6 trans-signaling via the soluble IL-6 receptor: importance for the pro-inflammatory activities of IL-6. Int. J. Biol. Sci. 8, 1237–1247 (2012).
Jones, S. A., Scheller, J. & Rose-John, S. Therapeutic strategies for the clinical blockade of IL-6/gp130 signaling. J. Clin. Invest. 121, 3375–3383 (2011).
Scheller, J., Chalaris, A., Garbers, C. & Rose-John, S. ADAM17: a molecular switch to control inflammation and tissue regeneration. Trends Immunol. 32, 380–387 (2011).
Chow, D., He, X., Snow, A. L., Rose-John, S. & Garcia, K. C. Structure of an extracellular gp130 cytokine receptor signaling complex. Science 291, 2150–2155 (2001).
Galun, E. & Rose-John, S. The regenerative activity of interleukin-6. Methods Mol. Biol. 982, 59–77 (2013).
Redlich, K. & Smolen, J. S. Inflammatory bone loss: pathogenesis and therapeutic intervention. Nat. Rev. Drug Discov. 11, 234–250 (2012).
Murakami, M. & Nishimoto, N. The value of blocking IL-6 outside of rheumatoid arthritis: current perspective. Curr. Opin. Rheumatol. 23, 273–277 (2011).
Rose-John, S. et al. Studies on the structure and regulation of the human hepatic interleukin-6 receptor. Eur. J. Biochem. 190, 79–83 (1990).
Scheller, J., Garbers, C. & Rose-John, S. Interleukin-6: from basic biology to selective blockade of pro-inflammatory activities. Semin. Immunol. 26, 2–12 (2014).
Scheller, J. & Rose-John, S. Interleukin-6 and its receptor: from bench to bedside. Med. Microbiol. Immunol. 195, 173–183 (2006).
Scheller, J., Grötzinger, J. & Rose-John, S. Updating interleukin-6 classic- and trans-signaling. Signal Transduction 6, 240–259 (2006).
O'Shea, J. J., Gadina, M. & Schreiber, R. D. Cytokine signaling in 2002: new surprises in the Jak/Stat pathway. Cell 109 (Suppl.), S121–S131 (2002).
Kishimoto, T. Interleukin-6: from basic science to medicine—40 years in immunology. Annu. Rev. Immunol. 23, 1–21 (2005).
Mullberg, J. et al. The soluble interleukin-6 receptor is generated by shedding. Eur. J. Immunol. 23, 473–480 (1993).
Mullberg, J., Schooltink, H., Stoyan, T., Heinrich, P. C. & Rose-John, S. Protein kinase C activity is rate limiting for shedding of the interleukin-6 receptor. Biochem. Biophys. Res. Commun. 189, 794–800 (1992).
Lust, J. A. et al. Isolation of an mRNA encoding a soluble form of the human interleukin-6 receptor. Cytokine 4, 96–100 (1992).
Horiuchi, S. et al. Soluble interleukin-6 receptors released from T cell or granulocyte/macrophage cell lines and human peripheral blood mononuclear cells are generated through an alternative splicing mechanism. Eur. J. Immunol. 24, 1945–1948 (1994).
Rose-John, S., & Heinrich, P. C. Soluble receptors for cytokines and growth factors: generation and biological function. Biochem. J. 300, 281–290 (1994).
Fischer, M. et al. A bioactive designer cytokine for human hematopoietic progenitor cell expansion. Nat. Biotechnol. 15, 142–145 (1997).
Galun, E. et al. Human interleukin-6 facilitates hepatitis B virus infection in vitro and in vivo. Virology 270, 299–309 (2000).
Peters, M. et al. Combined interleukin 6 and soluble interleukin 6 receptor accelerates murine liver regeneration. Gastroenterology 119, 1663–1671 (2000).
Hecht, N. et al. Hyper-IL-6 gene therapy reverses fulminant hepatic failure. Mol. Ther. 3, 683–687 (2001).
Audet, J., Miller, C. L., Rose-John, S., Piret, J. M. & Eaves, C. J. Distinct role of gp130 activation in promoting self-renewal divisions by mitogenically stimulated murine hematopoietic stem cells. Proc. Natl Acad. Sci. USA 98, 1757–1762 (2001).
März, P. et al. Sympathetic neurons can produce and respond to interleukin 6. Proc. Natl Acad. Sci. USA 95, 3251–3256 (1998).
März, P., Otten, U. & Rose-John, S. Neural activities of IL-6-type cytokines often depend on soluble cytokine receptors. Eur. J. Neurosci. 11, 2995–3004 (1999).
Klouche, M., Bhakdi, S., Hemmes, M. & Rose-John, S. Novel path to activation of vascular smooth muscle cells: up-regulation of gp130 creates an autocrine activation loop by IL-6 and its soluble receptor. J. Immunol. 163, 4583–4589 (1999).
Jostock, T. et al. Soluble gp130 is the natural inhibitor of soluble interleukin-6 receptor transsignaling responses. Eur. J. Biochem. 268, 160–167 (2001).
Atreya, R. et al. Blockade of interleukin 6 trans signaling suppresses T-cell resistance against apoptosis in chronic intestinal inflammation: evidence in Crohn disease and experimental colitis in vivo. Nat. Med. 6, 583–588 (2000).
Mitsuyama, K. et al. STAT3 activation via interleukin 6 trans-signalling contributes to ileitis in SAMP1/Yit mice. Gut 55, 1263–1269 (2006).
Hurst, S. M. et al. IL-6 and its soluble receptor orchestrate a temporal switch in the pattern of leukocyte recruitment seen during acute inflammation. Immunity 14, 705–714 (2001).
Nowell, M. A. et al. Soluble IL-6 receptor governs IL-6 activity in experimental arthritis: blockade of arthritis severity by soluble glycoprotein 130. J. Immunol. 171, 3202–3209 (2003).
Nowell, M. A. et al. Therapeutic targeting of IL-6 trans signaling counteracts STAT3 control of experimental inflammatory arthritis. J. Immunol. 182, 613–622 (2009).
Doganci, A. et al. The IL-6R alpha chain controls lung CD4+CD25+ TREG development and function during allergic airway inflammation in vivo. J. Clin. Invest. 115, 313–325 (2005).
Zhang, H. et al. IL-6 trans-signaling promotes pancreatitis-associated lung injury and lethality. J. Clin. Invest. 123, 1019–1031 (2013).
Becker, C. et al. TGF-β suppresses tumor progression in colon cancer by inhibition of IL-6 trans-signaling. Immunity 21, 491–501 (2004).
Becker, C. et al. IL-6 signaling promotes tumor growth in colorectal cancer. Cell Cycle 4, 217–220 (2005).
Matsumoto, S. et al. Essential roles of IL-6 trans-signaling in colonic epithelial cells, induced by the IL-6/soluble-IL-6 receptor derived from lamina propria macrophages, on the development of colitis-associated premalignant cancer in a murine model. J. Immunol. 184, 1543–1551 (2010).
Grivennikov, S. et al. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell 15, 103–113 (2009).
Hoge, J. et al. IL-6 controls the innate immune response against Listeria monocytogenes via classical IL-6 signaling. J. Immunol. 190, 703–711 (2013).
Sodenkamp, J. et al. Therapeutic targeting of interleukin-6 trans-signaling does not affect the outcome of experimental tuberculosis. Immunobiology 217, 996–1004 (2012).
Scheller, J., Chalaris, A., Schmidt-Arras, D. & Rose-John, S. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim. Biophys. Acta 181, 878–888 (2011).
Matthews, V. et al. Cellular cholesterol depletion triggers shedding of the human interleukin-6 receptor by ADAM10 and ADAM17 (TACE). J. Biol. Chem. 278, 38829–38839 (2003).
Black, R. A. et al. A metalloproteinase disintegrin that releases tumour-necrosis factor-α from cells. Nature 385, 729–733 (1997).
Peschon, J. J. et al. An essential role for ectodomain shedding in mammalian development. Science 282, 1281–1284 (1998).
Chalaris, A. et al. Critical role of the disintegrin metalloprotease ADAM17 for intestinal inflammation and regeneration in mice. J. Exp. Med. 207, 1617–1624 (2010).
Spehlmann, M. E. et al. Trp53 deficiency protects against acute intestinal inflammation. J. Immunol. 191, 837–847 (2013).
Waetzig, G. H. & Rose-John, S. Hitting a complex target: an update on interleukin-6 trans-signalling. Expert Opin. Ther. Targets 16, 225–236 (2012).
Barkhausen, T. et al. Selective blockade of interleukin-6 trans-signaling improves survival in a murine polymicrobial sepsis model. Crit. Care Med. 39, 1407–1413 (2011).
Rafiq, S. et al. A common variant of the interleukin 6 receptor (IL-6r) gene increases IL-6r and IL-6 levels, without other inflammatory effects. Genes Immun. 8, 552–559 (2007).
Ferreira, R. C. et al. Functional IL6R 358Ala allele impairs classical IL-6 receptor signaling and influences risk of diverse inflammatory diseases. PLoS Genet. 9, e1003444 (2013).
Scheller, J. & Rose-John, S. The interleukin 6 pathway and atherosclerosis. Lancet 380, 338 (2012).
Chalaris, A. et al. Apoptosis is a natural stimulus of IL6R shedding and contributes to the proinflammatory trans-signaling function of neutrophils. Blood 110, 1748–1755 (2007).
Heinrich, P. C., Castell, J. V. & Andus, T. Interleukin-6 and the acute phase response. Biochem. J. 265, 621–636 (1990).
Febbraio, M. A., Rose-John, S. & Pedersen, B. K. Is interleukin-6 receptor blockade the Holy Grail for inflammatory diseases? Clin. Pharmacol. Ther. 87, 396–398 (2010).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Annunziato, F. & Romagnani, S. Heterogeneity of human effector CD4+ T cells. Arthritis Res. Ther. 11, 257 (2009).
Korn, T. et al. IL-6 controls TH17 immunity in vivo by inhibiting the conversion of conventional T cells into Foxp3+ regulatory T cells. Proc. Natl Acad. Sci. USA 105, 18460–18465 (2008).
Thiolat, A. et al. Interleukin-6 receptor blockade enhances CD39+ regulatory T cell development in rheumatoid arthritis and in experimental arthritis. Arthritis Rheum. 66, 273–283 (2014).
Briso, E. M., Dienz, O. & Rincon, M. Cutting edge: soluble IL-6R is produced by IL-6R ectodomain shedding in activated CD4 T cells. J. Immunol. 180, 7102–7106 (2008).
Dominitzki, S. et al. Cutting edge: trans-signaling via the soluble IL-6R abrogates the induction of FoxP3 in naive CD4+CD25 T cells. J. Immunol. 179, 2041–2045 (2007).
O'Shea, J. J. & Plenge, R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity 36, 542–550 (2012).
Tanaka, T., Narazaki, M. & Kishimoto, T. Therapeutic targeting of the interleukin-6 receptor. Annu. Rev. Pharmacol. Toxicol. 52, 199–219 (2012).
Gabay, C. et al. Tocilizumab monotherapy versus adalimumab monotherapy for treatment of rheumatoid arthritis (ADACTA): a randomised, double-blind, controlled phase 4 trial. Lancet 381, 1541–1550 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Singh, J. A. et al. Biologics for rheumatoid arthritis: an overview of Cochrane reviews. Cochrane Database of Systematic Reviews, Issue 4. Art. No.: CD007848 http://dx.doi.org/10.1002/14651858.CD007848.pub2.
Schoels, M. M. et al. Blocking the effects of interleukin-6 in rheumatoid arthritis and other inflammatory rheumatic diseases: systematic literature review and meta-analysis informing a consensus statement. Ann. Rheum. Dis. 72, 583–589 (2013).
Smolen, J. S. et al. Consensus statement on blocking the effects of interleukin-6 and in particular by interleukin-6 receptor inhibition in rheumatoid arthritis and other inflammatory conditions. Ann. Rheum. Dis. 72, 482–492 (2013).
de Boysson, H., Fevrier, J., Nicolle, A., Auzary, C. & Geffray, L. Tocilizumab in the treatment of the adult-onset Still's disease: current clinical evidence. Clin. Rheumatol. 32, 141–147 (2013).
Shima, Y. et al. The skin of patients with systemic sclerosis softened during the treatment with anti-IL-6 receptor antibody tocilizumab. Rheumatology (Oxford) 49, 2408–2412 (2010).
Hagihara, K., Kawase, I., Tanaka, T. & Kishimoto, T. Tocilizumab ameliorates clinical symptoms in polymyalgia rheumatica. J. Rheumatol. 37, 1075–1076 (2010).
Illei, G. G. et al. Tocilizumab in systemic lupus erythematosus: data on safety, preliminary efficacy, and impact on circulating plasma cells from an open-label phase I dosage-escalation study. Arthritis Rheum. 62, 542–552 (2010).
Kieseier, B. C. et al. Disease amelioration with tocilizumab in a treatment-resistant patient with neuromyelitis optica: implication for cellular immune responses. JAMA Neurol. 70, 390–393 (2013).
Weyand, C. M. & Goronzy, J. J. Immune mechanisms in medium and large-vessel vasculitis. Nat. Rev. Rheumatol. 9, 731–740 (2013).
Unizony, S. et al. Tocilizumab for the treatment of large-vessel vasculitis (giant cell arteritis, Takayasu arteritis) and polymyalgia rheumatica. Arthritis Care Res. 64, 1720–1729 (2012).
Salvarani, C. et al. Tocilizumab: a novel therapy for patients with large-vessel vasculitis. Rheumatology (Oxford) 51, 151–156 (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Sieper, J. et al. Sarilumab for the treatment of ankylosing spondylitis: results of a phase II, randomised, double-blind, placebo-controlled study (ALIGN). Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-204963.
Sieper, J., Porter-Brown, B., Thompson, L., Harari, O. & Dougados, M. Assessment of short-term symptomatic efficacy of tocilizumab in ankylosing spondylitis: results of randomised, placebo-controlled trials. Ann. Rheum. Dis. 73, 95–100 (2014).
Navarro, G., Taroumian, S., Barroso, N., Duan, L. & Furst, D. Tocilizumab in rheumatoid arthritis: A meta-analysis of efficacy and selected clinical conundrums. Semin. Arthritis Rheum. 43, 458–469 (2014).
Schiff, M. H. et al. Integrated safety in tocilizumab clinical trials. Arthritis Res. Ther. 13, R141 (2011).
Furst, D. E. et al. Updated consensus statement on biological agents for the treatment of rheumatic diseases, 2012. Ann. Rheum. Dis. 72 (Suppl. 2), ii2–ii34 (2013).
Huizinga, T. W. et al. Sarilumab, a fully human monoclonal antibody against IL-6Rα in patients with rheumatoid arthritis and an inadequate response to methotrexate: efficacy and safety results from the randomised SARIL-RA-MOBILITY Part A trial. Ann. Rheum. Dis. 73, 1626–1634 (2014).
Szepietowski, J. C. et al. Phase I, randomized, double-blind, placebo-controlled, multiple intravenous, dose-ascending study of sirukumab in cutaneous or systemic lupus erythematosus. Arthritis Rheum. 65, 2661–2671 (2013).
Jain, A. & Singh, J. A. Harms of TNF inhibitors in rheumatic diseases: a focused review of the literature. Immunotherapy 5, 265–299 (2013).
Ladel, C. H. et al. Lethal tuberculosis in interleukin-6-deficient mutant mice. Infect. Immun. 65, 4843–4849 (1997).
Kitas, G. D. & Gabriel, S. E. Cardiovascular disease in rheumatoid arthritis: state of the art and future perspectives. Ann. Rheum. Dis. 70, 8–14 (2011).
Robertson, J., Peters, M. J., McInnes, I. B. & Sattar, N. Changes in lipid levels with inflammation and therapy in RA: a maturing paradigm. Nat. Rev. Rheumatol. 9, 513–523 (2013).
Schultz, O. et al. Effects of inhibition of interleukin-6 signalling on insulin sensitivity and lipoprotein (a) levels in human subjects with rheumatoid diseases. PLoS ONE 5, e14328 (2010).
McInnes, I. B. et al. Effect of interleukin-6 receptor blockade on surrogates of vascular risk in rheumatoid arthritis: MEASURE, a randomised, placebo-controlled study. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-204345.
Acknowledgements
The work of S.R.-J. is supported by grants from the Deutsche Forschungsgemeinschaft Bonn, Germany (SFB654, project C5; SFB841, project C1; SFB877 project A1) and by the Cluster of Excellence 'Inflammation at Interfaces'.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
L.H.C. declares that he has acted as a consultant to Genentech Roche, Sanofi-Aventis, UCB, Bristol–Myers Squib and Pfizer, and has acted as a speaker for Genentech and Bristol–Myers Squib. S.R. J. declares that he is an inventor on patents owned by CONARIS Research Institute, which develops the sgp130Fc protein together with Ferring Pharmaceuticals, and he has stock ownership in CONARIS.
Rights and permissions
About this article
Cite this article
Calabrese, L., Rose-John, S. IL-6 biology: implications for clinical targeting in rheumatic disease. Nat Rev Rheumatol 10, 720–727 (2014). https://doi.org/10.1038/nrrheum.2014.127
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.127
This article is cited by
-
Unveiling the molecular Hallmarks of Peyronie’s disease: a comprehensive narrative review
International Journal of Impotence Research (2024)
-
Complete spectrum of adverse events associated with chimeric antigen receptor (CAR)-T cell therapies
Journal of Biomedical Science (2023)
-
Multi-proteomic Biomarker Risk Scores for Predicting Risk and Guiding Therapy in Patients with Coronary Artery Disease
Current Cardiology Reports (2023)
-
STAT-3 signaling role in an experimental model of nephropathy induced by doxorubicin
Molecular and Cellular Biochemistry (2023)
-
Role of interleukin-6-mediated inflammation in the pathogenesis of inflammatory bowel disease: focus on the available therapeutic approaches and gut microbiome
Journal of Cell Communication and Signaling (2023)