Key Points
-
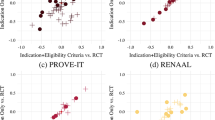
Unlike research findings on risk factors for incident conditions, the evidence on risk factors for disease sequelae among patients with rheumatic diseases have often been inconsistent or paradoxical
-
Although biological explanations for these counterintuitive results might exist, an enticing methodological explanation is a type of selection bias called index event bias, which can affect research on disease sequelae
-
Propensity score methods or active comparator analysis in pharmaco-epidemiological research helps to address confounding issues in observational studies, but does not address selection bias owing to potential differential loss to follow-up
-
The depletion of susceptibles can explain the decreasing impact of risk factors on mortality with ageing in rheumatic conditions, as well as explain the null (or inverse) associations of prevalent exposure studies
-
To avoid these issues, investigators should carefully specify the research question of interest and clarify the time sequence of exposures, mediators, and outcome variables
-
Furthermore, investigators should use incident exposures whenever possible, minimize loss to follow-up, and exercise proper inference
Abstract
The identification of modifiable risk factors for the development of rheumatic conditions and their sequelae is crucial for reducing the substantial worldwide burden of these diseases. However, the validity of such research can be threatened by sources of bias, including confounding, measurement and selection biases. In this Review, we discuss potentially major issues of selection bias—a type of bias frequently overshadowed by other bias and feasibility issues, despite being equally or more problematic—in key areas of rheumatic disease research. We present index event bias (a type of selection bias) as one of the potentially unifying reasons behind some unexpected findings, such as the 'risk factor paradox'—a phenomenon exemplified by the discrepant effects of certain risk factors on the development versus the progression of osteoarthritis (OA) or rheumatoid arthritis (RA). We also discuss potential selection biases owing to differential loss to follow-up in RA and OA research, as well as those due to the depletion of susceptibles (prevalent user bias) and immortal time bias. The lesson remains that selection bias can be ubiquitous and, therefore, has the potential to lead the field astray. Thus, we conclude with suggestions to help investigators avoid such issues and limit the impact on future rheumatology research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Reginster, J. Y. The prevalence and burden of arthritis. Rheumatology (Oxford) 41 (Suppl. 1), 3–6 (2002).
Symmons, D. P. & Gabriel, S. E. Epidemiology of CVD in rheumatic disease, with a focus on RA and SLE. Nat. Rev. Rheumatol. 7, 399–408 (2011).
Gabriel, S. E. Heart disease and rheumatoid arthritis: understanding the risks. Ann. Rheum. Dis. 69 (Suppl. 1), i61–i64 (2010).
Eder, L. et al. The association between smoking and the development of psoriatic arthritis among psoriasis patients. Ann. Rheum. Dis. 71, 219–224 (2012).
Zhang, Y. et al. Methodologic challenges in studying risk factors for progression of knee osteoarthritis. Arthritis Care Res. (Hoboken) 62, 1527–1532 (2010).
Canto, J. G. et al. Number of coronary heart disease risk factors and mortality in patients with first myocardial infarction. JAMA 306, 2120–2127 (2011).
Zhang, Y. & Jordan, J. M. Epidemiology of osteoarthritis. Rheum. Dis. Clin. North Am. 34, 515–529 (2008).
Felson, D. T. et al. Osteoarthritis: new insights. Part 1: the disease and its risk factors. Ann. Intern. Med. 133, 635–646 (2000).
Belo, J. N., Berger, M. Y., Reijman, M., Koes, B. W. & Bierma-Zeinstra, S. M. Prognostic factors of progression of osteoarthritis of the knee: a systematic review of observational studies. Arthritis Rheum. 57, 13–26 (2007).
Zhang, Y. et al. Bone mineral density and risk of incident and progressive radiographic knee osteoarthritis in women: the Framingham Study. J. Rheumatol 27, 1032–1037 (2000).
Hart, D. J. et al. The relationship of bone density and fracture to incident and progressive radiographic osteoarthritis of the knee: the Chingford Study. Arthritis Rheum. 46, 92–99 (2002).
Lane, N. E. et al. Wnt signaling antagonists are potential prognostic biomarkers for the progression of radiographic hip osteoarthritis in elderly Caucasian women. Arthritis Rheum. 56, 3319–3325 (2007).
McAlindon, T. E. et al. Do antioxidant micronutrients protect against the development and progression of knee osteoarthritis? Arthritis Rheum. 39, 648–656 (1996).
Vesperini, V. et al. Tobacco exposure reduces radiographic progression in early rheumatoid arthritis. Results from the ESPOIR cohort. Arthritis Care Res. (Hoboken) 65, 1899–1906 (2013).
Harrison, B. J., Silman, A. J., Wiles, N. J., Scott, D. G. & Symmons, D. P. The association of cigarette smoking with disease outcome in patients with early inflammatory polyarthritis. Arthritis Rheum. 44, 323–330 (2001).
Finckh, A., Dehler, S., Costenbader, K. H. & Gabay, C. Cigarette smoking and radiographic progression in rheumatoid arthritis. Ann. Rheum. Dis. 66, 1066–1071 (2007).
Gonzalez, A. et al. Do cardiovascular risk factors confer the same risk for cardiovascular outcomes in rheumatoid arthritis patients as in non-rheumatoid arthritis patients? Ann. Rheum. Dis. 67, 64–69 (2008).
Naranjo, A. et al. Cardiovascular disease in patients with rheumatoid arthritis: results from the QUEST-RA study. Arthritis Res. Ther. 10, R30 (2008).
Manson, J. E. et al. A prospective study of obesity and risk of coronary heart disease in women. N. Engl. J. Med. 322, 882–889 (1990).
Escalante, A., Haas, R. W. & del Rincon, I. Paradoxical effect of body mass index on survival in rheumatoid arthritis: role of comorbidity and systemic inflammation. Arch. Intern. Med. 165, 1624–1629 (2005).
Wilson, P. W. et al. Prediction of coronary heart disease using risk factor categories. Circulation 97, 1837–1847 (1998).
Myasoedova, E. et al. Lipid paradox in rheumatoid arthritis: the impact of serum lipid measures and systemic inflammation on the risk of cardiovascular disease. Ann. Rheum. Dis. 70, 482–487 (2011).
Peters, M. J. et al. EULAR evidence-based recommendations for cardiovascular risk management in patients with rheumatoid arthritis and other forms of inflammatory arthritis. Ann. Rheum. Dis. 69, 325–331 (2009).
Solomon, D. H., Peters, M. J., Nurmohamed, M. T. & Dixon, W. Unresolved questions in rheumatology: motion for debate: the data support evidence-based management recommendations for cardiovascular disease in rheumatoid arthritis. Arthritis Rheum. 65, 1675–1683 (2013).
Li, W., Han, J. & Qureshi, A. A. Smoking and risk of incident psoriatic arthritis in US women. Ann. Rheum. Dis. 71, 804–808 (2011).
Bowcock, A. M. & Cookson, W. O. The genetics of psoriasis, psoriatic arthritis and atopic dermatitis. Hum. Mol. Genet. 13 (Suppl. 1), R43–R55 (2004).
Duffin, K. C. et al. Genetics of psoriasis and psoriatic arthritis: update and future direction. J. Rheumatol 35, 1449–1453 (2008).
Aune, E., Roislien, J., Mathisen, M., Thelle, D. S. & Otterstad, J. E. The “smoker's paradox” in patients with acute coronary syndrome: a systematic review. BMC Med. 9, 97 (2011).
Romero-Corral, A. et al. Association of bodyweight with total mortality and with cardiovascular events in coronary artery disease: a systematic review of cohort studies. Lancet 368, 666–678 (2006).
Lavie, C. J., De Schutter, A., Patel, D., Artham, S. M. & Milani, R. V. Body composition and coronary heart disease mortality—an obesity or a lean paradox? Mayo Clin. Proc. 86, 857–864 (2011).
Dahabreh, I. J. & Kent, D. M. Index event bias as an explanation for the paradoxes of recurrence risk research. JAMA 305, 822–823 (2011).
Kent, D. M. & Thaler, D. E. Is patent foramen ovale a modifiable risk factor for stroke recurrence? Stroke 41, S26–S30 (2010).
Tyas, S. L. et al. Transitions to mild cognitive impairments, dementia, and death: findings from the Nun Study. Am. J. Epidemiol. 165, 1231–1238 (2007).
Glymour, M. M. Invited commentary: when bad genes look good—APOE*E4, cognitive decline, and diagnostic thresholds. Am. J. Epidemiol. 165, 1239–1246; author reply 1247 (2007).
Baglin, T. Unraveling the thrombophilia paradox: from hypercoagulability to the prothrombotic state. J. Thromb. Haemost. 8, 228–233 (2010).
Hernandez-Diaz, S., Schisterman, E. F. & Hernan, M. A. The birth weight “paradox” uncovered? Am. J. Epidemiol. 164, 1115–1120 (2006).
Myers, J. et al. The obesity paradox and weight loss. Am. J. Med. 124, 924–930 (2011).
VanderWeele, T. J., Mumford, S. L. & Schisterman, E. F. Conditioning on intermediates in perinatal epidemiology. Epidemiology 23, 1–9 (2011).
VanderWeele, T. J. & Robins, J. M. Directed acyclic graphs, sufficient causes, and the properties of conditioning on a common effect. Am. J. Epidemiol. 166, 1096–1104 (2007).
Smits, L. J. et al. Index event bias—a numerical example. J. Clin. Epidemiol. 66, 192–196 (2013).
Westreich, D. & Greenland, S. The Table 2 fallacy: presenting and interpreting confounder and modifier coefficients. Am. J. Epidemiol. 177, 292–298 (2013).
Valeri, L. & Vanderweele, T. J. Mediation analysis allowing for exposure-mediator interactions and causal interpretation: theoretical assumptions and implementation with SAS and SPSS macros. Psychol. Methods 18, 137–150 (2013).
Zhang, Y. et al. What effect is really being measured? An alternative explanation of paradoxical phenomenon in studies of osteoarthritis progression. Arthritis Care & Res (Hoboken) http://dx.doi.org/10.1002/acr.22213.
Felson, D. T. et al. Risk factors for incident radiographic knee osteoarthritis in the elderly: the Framingham Study. Arthritis Rheum. 40, 728–733 (1997).
Cooper, C. et al. Risk factors for the incidence and progression of radiographic knee osteoarthritis. Arthritis Rheum. 43, 995–1000 (2000).
Grijalva, C. G. et al. Initiation of tumor necrosis factor-alpha antagonists and the risk of hospitalization for infection in patients with autoimmune diseases. JAMA 306, 2331–2339 (2011).
Solomon, D. H. et al. Association between disease-modifying antirheumatic drugs and diabetes risk in patients with rheumatoid arthritis and psoriasis. JAMA 305, 2525–2531 (2011).
O'Dell, J. R. et al. Therapies for active rheumatoid arthritis after methotrexate failure. N. Engl. J. Med. 369, 307–318 (2013).
O'Dell, J. R. et al. Treatment of rheumatoid arthritis with methotrexate and hydroxychloroquine, methotrexate and sulfasalazine, or a combination of the three medications: results of a two-year, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 46, 1164–1170 (2002).
O'Dell, J. R. et al. Treatment of rheumatoid arthritis with methotrexate alone, sulfasalazine and hydroxychloroquine, or a combination of all three medications. N. Engl. J. Med. 334, 1287–1291 (1996).
Dixon, W. & Felson, D. T. Is anti-TNF therapy safer than previously thought? JAMA 306, 2380–2381 (2011).
Hernan, M. A., Hernandez-Diaz, S. & Robins, J. M. Randomized trials analyzed as observational studies. Ann. Intern. Med. http://dx.doi.org/10.7326/0003-4819-159-8-201310150-00709.
Bongartz, T. et al. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 295, 2275–2285 (2006).
Little, R. J. et al. The prevention and treatment of missing data in clinical trials. N. Engl. J. Med. 367, 1355–1360 (2012).
Doll, R. & Hill, A. B. Mortality of British doctors in relation to smoking: observations on coronary thrombosis. Natl Cancer Inst. Monogr. 19, 205–268 (1966).
Wolfe, F. & Michaud, K. Effect of body mass index on mortality and clinical status in rheumatoid arthritis. Arthritis Care Res. (Hoboken) 64, 1471–1479 (2012).
Nguyen, U. S., Niu, J., Choi, H. K. & Zhang, Y. Body mass index and mortality: comment on article by Wolfe and Michaud. Arthritis Care Res. (Hoboken) 65, 834–835 (2013).
Choi, H. K. et al. The risk of pulmonary embolism and deep vein thrombosis in rheumatoid arthritis: a UK population-based outpatient cohort study. Ann. Rheum. Dis. 72, 1182–1187 (2013).
Zoller, B., Li, X., Sundquist, J. & Sundquist, K. Risk of pulmonary embolism in patients with autoimmune disorders: a nationwide follow-up study from Sweden. Lancet 379, 244–249 (2012).
Grodstein, F. & Stampfer, M. The epidemiology of coronary heart disease and estrogen replacement in postmenopausal women. Prog. Cardiovasc. Dis. 38, 199–210 (1995).
Grady, D. et al. Hormone therapy to prevent disease and prolong life in postmenopausal women. Ann. Intern. Med. 117, 1016–1037 (1992).
Manson, J. E. et al. Estrogen plus progestin and the risk of coronary heart disease. N. Engl. J. Med. 349, 523–534 (2003).
Hernan, M. A. et al. Observational studies analyzed like randomized experiments: an application to postmenopausal hormone therapy and coronary heart disease. Epidemiology 19, 766–779 (2008).
Levesque, L. E., Hanley, J. A., Kezouh, A. & Suissa, S. Problem of immortal time bias in cohort studies: example using statins for preventing progression of diabetes. BMJ 340, b5087 (2010).
Tsai, C. L. & Camargo, C. A. Jr. Methodological considerations, such as directed acyclic graphs, for studying “acute on chronic” disease epidemiology: chronic obstructive pulmonary disease example. J. Clin. Epidemiol. 62, 982–990 (2009).
Rich, J. D. et al. Prior aspirin use and outcomes in acute coronary syndromes. J. Am. Coll. Cardiol. 56, 1376–1385 (2010).
Acknowledgements
This work was partly supported by grants from the NIH (NIAMS): grants R01-AR056291, R01-AR065944, K01AR064351 and P60AR047785.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to researching the data for the article, discussions of the content, writing the article and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Let us consider an initial cohort of 30,000 participants without rheumatoid arthritis (RA) at baseline and assume that there are four risk factors involved in the aetiology of RA incidence (E1) or progression (E2), namely the risk factor of interest (R) and three other unmeasured risk factors (U1, U2, and U3). (DOC 92 kb)
Rights and permissions
About this article
Cite this article
Choi, H., Nguyen, US., Niu, J. et al. Selection bias in rheumatic disease research. Nat Rev Rheumatol 10, 403–412 (2014). https://doi.org/10.1038/nrrheum.2014.36
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.36
This article is cited by
-
Obesity and the risk of multiple or severe frequent knee pain episodes: a 4-year follow-up of the ELSA-Brasil MSK cohort
International Journal of Obesity (2024)
-
Prevalence of H. pylori among patients undergoing coronary angiography (The HP-DAPT prevalence study)
Scientific Reports (2022)
-
The comparative effect of exposure to various risk factors on the risk of hyperuricaemia: diet has a weak causal effect
Arthritis Research & Therapy (2021)
-
Increased risk of malignancy in patients with systemic lupus erythematosus: population-based cohort study in Korea
Arthritis Research & Therapy (2021)
-
Prevalence of ultrasound-detected knee synovial abnormalities in a middle-aged and older general population—the Xiangya Osteoarthritis Study
Arthritis Research & Therapy (2021)