Abstract
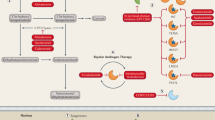
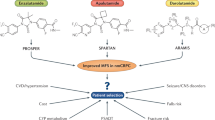
The majority of prostate cancers are hormone-dependent at diagnosis highlighting the central role of androgen signalling in this disease. Surprisingly, most forms of castration-resistant prostate cancer (CRPC) are still dependent on the androgen receptor (AR) for survival. Therefore, the advent of new AR-targeting drugs, such as enzalutamide, is certainly beneficial for the many patients with metastatic CRPC. Indeed, this compound provides a substantial survival benefit—but it is not curative. This Perspectives article describes the different ways through which cancer cells can become resistant to enzalutamide, such as AR truncation and other mutations, as well as by-pass of the AR dependence of prostate cancer cells through expression of the glucocorticoid receptor. The clinical relevance of these mechanisms and emerging questions concerning new therapeutic regimens in the treatment of metastatic CRPC are being discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
van Soest, R. J. et al. Cross-resistance between taxanes and new hormonal agents abiraterone and enzalutamide may affect drug sequence choices in metastatic castration-resistant prostate cancer. Eur. J. Cancer 49, 3821–3830 (2013).
Rathkopf, D. E. et al. Updated interim efficacy analysis and long-term safety of abiraterone acetate in metastatic castration-resistant prostate cancer patients without prior chemotherapy (COU-AA-302). Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2014.02.056.
Scher, H. I. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 367, 1187–1197 (2012).
Zong, Y. & Goldstein, A. S. Adaptation or selection—mechanisms of castration-resistant prostate cancer. Nat. Rev. Urol. 10, 90–98 (2013).
Claessens, F. et al. Diverse roles of androgen receptor (AR) domains in AR-mediated signaling. Nucl. Recept. Signal. 6, e008 (2008).
Hu, R. et al. Ligand-independent androgen receptor variants derived from splicing of cryptic exons signify hormone-refractory prostate cancer. Cancer Res. 69, 16–22 (2009).
Guo, Z. et al. A novel androgen receptor splice variant is up-regulated during prostate cancer progression and promotes androgen depletion-resistant growth. Cancer Res. 69, 2305–2313 (2009).
Ware, K. E., Garcia-Blanco, M. A., Armstrong, A. J. & Dehm, S. M. Biologic and clinical significance of androgen receptor variants in castration resistant prostate cancer. Endocr. Relat Cancer 21, T87–T103 (2014).
Liu, G. et al. AR variant ARv567es induces carcinogenesis in a novel transgenic mouse model of prostate cancer. Neoplasia 15, 1009–1017 (2013).
Li, Y. et al. Androgen receptor splice variants mediate enzalutamide resistance in castration-resistant prostate cancer cell lines. Cancer Res. 73, 483–489 (2013).
Efstathiou, E. et al. Molecular characterization of enzalutamide-treated bone metastatic castration-resistant prostate cancer. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2014.05.005.
Yu, Z. et al. Rapid induction of androgen receptor splice variants by androgen deprivation in prostate cancer. Clin. Cancer Res. 20, 1590–1600 (2014).
Sun, S. et al. Castration resistance in human prostate cancer is conferred by a frequently occurring androgen receptor splice variant. J. Clin. Invest. 120, 2715–2730 (2010).
Tanner, T. M. et al. A 629RKLKK633 motif in the hinge region controls the androgen receptor at multiple levels. Cell. Mol. Life Sci. 67, 1919–1927 (2010).
Watson, P. A. et al. Constitutively active androgen receptor splice variants expressed in castration-resistant prostate cancer require full-length androgen receptor. Proc. Natl Acad. Sci. USA 107, 16759–16765 (2010).
Helsen, C. et al. Evidence for DNA-binding domain–ligand-binding domain communications in the androgen receptor. Mol. Cell Biol. 32, 3033–3043 (2012).
Waltering, K. K., Urbanucci, A. & Visakorpi, T. Androgen receptor (AR) aberrations in castration-resistant prostate cancer. Mol. Cell Endocrinol. 360, 38–43 (2012).
Suzuki, H. et al. Codon 877 mutation in the androgen receptor gene in advanced prostate cancer: relation to antiandrogen withdrawal syndrome. Prostate 29, 153–158 (1996).
Yoshida, T. et al. Antiandrogen bicalutamide promotes tumor growth in a novel androgen-dependent prostate cancer xenograft model derived from a bicalutamide-treated patient. Cancer Res. 65, 9611–9616 (2005).
Balbas, M. D. et al. Overcoming mutation-based resistance to antiandrogens with rational drug design. Elife 2, e00499 (2013).
Joseph, J. D. et al. A clinically relevant androgen receptor mutation confers resistance to second-generation antiandrogens enzalutamide and ARN-509. Cancer Discov. 3, 1020–1029 (2013).
Korpal, M. et al. An F876L mutation in androgen receptor confers genetic and phenotypic resistance to MDV3100 (enzalutamide). Cancer Discov. 3, 1030–43 (2013).
Clegg, N. J. et al. ARN-509: a novel antiandrogen for prostate cancer treatment. Cancer Res. 72, 1494–1503 (2012).
Sartor, A. O. et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group trial (SWOG 9426). Cancer 112, 2393–2400 (2008).
von Klot, C. A. et al. Is there an anti-androgen withdrawal syndrome for enzalutamide? World J. Urol. http://dx.doi.org/10.1007/s00345-014-1288-3.
Yano, A. et al. Glucocorticoids suppress tumor lymphangiogenesis of prostate cancer cells. Clin. Cancer Res. 12, 6012–6017 (2006).
Nishimura, K. et al. Potential mechanism for the effects of dexamethasone on growth of androgen-independent prostate cancer. J. Natl Cancer Inst. 93, 1739–1746 (2001).
de Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011).
Fosså, S. D. et al. Flutamide versus prednisone in patients with prostate cancer symptomatically progressing after androgen-ablative therapy: a phase III study of the European organization for research and treatment of cancer genitourinary group. J. Clin. Oncol. 19, 62–71 (2001).
Storlie, J. A. et al. Prostate specific antigen levels and clinical response to low dose dexamethasone for hormone-refractory metastatic prostate carcinoma. Cancer 76, 96–100 (1995).
Zhao, X. Y. et al. Glucocorticoids can promote androgen-independent growth of prostate cancer cells through a mutated androgen receptor. Nat. Med. 6, 703–706 (2000).
Yan, T. Z., Jin, F. S., Xie, L. P. & Li, L. C. Relationship between glucocorticoid receptor signal pathway and androgen-independent prostate cancer. Urol. Int. 81, 228–233 (2008).
Isikbay, M. et al. Glucocorticoid receptor activity contributes to resistance to androgen-targeted therapy in prostate cancer. Horm. Cancer 5, 72–89 (2014).
Yemelyanov, A. et al. Differential targeting of androgen and glucocorticoid receptors induces ER stress and apoptosis in prostate cancer cells: a novel therapeutic modality. Cell Cycle 11, 395–406 (2012).
Arora, V. K. et al. Glucocorticoid receptor confers resistance to antiandrogens by bypassing androgen receptor blockade. Cell 155, 1309–1322 (2013).
Sharifi, N. Steroid receptors aplenty in prostate cancer. N. Engl. J. Med. 370, 970–971 (2014).
Denayer, S., Helsen, C., Thorrez, L., Haelens, A. & Claessens, F. The rules of DNA recognition by the androgen receptor. Mol. Endocrinol. 24, 898–913 (2010).
Sahu, B. et al. FoxA1 specifies unique androgen and glucocorticoid receptor binding events in prostate cancer cells. Cancer Res. 73, 1570–1580 (2013).
Mottet, N. et al. Implementing newer agents for the management of castrate-resistant prostate cancer: what is known and what is needed? BJU Int. http://dx.doi.org/10.1111/bju.12736.
Noonan, K. L. et al. Clinical activity of abiraterone acetate in patients with metastatic castration-resistant prostate cancer progressing after enzalutamide. Ann. Oncol. 24, 1802–1807 (2013).
Schrader, A. J. et al. Enzalutamide in castration-resistant prostate cancer patients progressing after docetaxel and abiraterone. Eur. Urol. 65, 30–36 (2014).
Sweeney, C. et al. Impact on overall survival (OS) with chemohormonal therapy versus hormonal therapy for hormone-sensitive newly metastatic prostate cancer (mPrCa): An ECOG-led phase III randomized trial [abstract]. J. Clin. Oncol. 32 (Suppl.), LBA2 (2014).
Acknowledgements
F.C. and S.J. hold grants from Fonds Wetenschappelijk Onderzoek-Vlaanderen (G.0684.12N and G.0830.13N), the Belgian federal government (National Cancer Plan KPC_29_023) and a Concerted Research Action of the KU Leuven (GOA/15/017).
Author information
Authors and Affiliations
Contributions
F.C., C.H., S.P. and T.V.d.B. researched the data for the article, provided substantial contributions to discussions of its content, wrote the article and undertook review and/or editing of the manuscript before submission. L.S., H.V.P. and S.J. contributed to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Claessens, F., Helsen, C., Prekovic, S. et al. Emerging mechanisms of enzalutamide resistance in prostate cancer. Nat Rev Urol 11, 712–716 (2014). https://doi.org/10.1038/nrurol.2014.243
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2014.243
This article is cited by
-
New advances of the androgen receptor in prostate cancer: report from the 1st International Androgen Receptor Symposium
Journal of Translational Medicine (2024)
-
RNF8 up-regulates AR/ARV7 action to contribute to advanced prostate cancer progression
Cell Death & Disease (2022)
-
AR-regulated ZIC5 contributes to the aggressiveness of prostate cancer
Cell Death Discovery (2022)
-
Androgen receptor antagonists produced by Streptomyces overcome resistance to enzalutamide
The Journal of Antibiotics (2021)
-
Nuclear βArrestin1 regulates androgen receptor function in castration resistant prostate cancer
Oncogene (2021)