Abstract
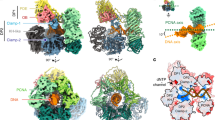
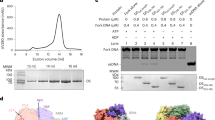
DNA polymerase X (Pol X) from the African swine fever virus (ASFV) specifically binds intermediates in the single-nucleotide base-excision repair process, an activity indicative of repair function. In addition, Pol X catalyzes DNA polymerization with low nucleotide-insertion fidelity. The structural mechanisms by which DNA polymerases confer high or low fidelity in DNA polymerization remain to be elucidated. The three-dimensional structure of Pol X has been determined. Unlike other DNA polymerases, Pol X is formed from only a palm and a C-terminal subdomain. Pol X has a novel palm subdomain fold, containing a positively charged helix at the DNA binding surface. Purine deoxynucleoside triphosphate (dNTP) substrates bind between the palm and C-terminal subdomain, at a dNTP-binding helix, and induce a unique conformation in Pol X. The purine dNTP–bound conformation and high binding affinity for dGTP–Mg2+ of Pol X may contribute to its low fidelity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Oliveros, M. et al. J. Biol. Chem. 272, 30899–30910 (1997).
Aravind, L. & Koonin, E.V. Nucleic Acids Res. 27, 1609–1618 (1999).
Showalter, A.K. & Tsai, M.-D. J. Am. Chem. Soc. 123, 1776–1777 (2001).
Odemuyiwa, S.O. et al. Virus Genes 20, 139–142 (2000).
Ruiz, J.F. et al. Philos. Trans. R. Soc. Lond. B 356, 99–109 (2001).
Dominguez, O. et al. EMBO J. 19, 1731–1742 (2000).
Maciejewski, M.W., Pan, B., Shin, R., Denninger, A. & Mullen, G.P. J. Biomol. NMR In the press (2001).
Ollis, D.L., Brick, P., Hamlin, R., Xuong, N.G. & Steitz, T.A. Nature 313, 762–766 (1985).
Pedersen, L.C., Benning, M.M. & Holden, H.M. Biochemistry 34, 13305–13311 (1995).
Sawaya, M.R., Pelletier, H., Kumar, A., Wilson, S.H. & Kraut, J. Science 264, 1930–1935 (1994).
Martin, G., Keller, W. & Doublie, S. EMBO J . 19, 4193–4203 (2000).
Bard, J. et al. Science 289, 1346–1349 (2000).
Srivastava, D.K. et al. J. Biol. Chem. 273, 21203–21209 (1998).
Dianov, G.L., Prasad, R., Wilson, S.H. & Bohr, V.A. J. Biol. Chem. 274, 13741–13743 (1999).
Liu, D.-J., Prasad, R., Wilson, S.H., DeRose, E.F. & Mullen, G.P. Biochemistry 35, 6188–6200 (1996).
Sawaya, M.R., Prasad, R., Wilson, S.H., Kraut, J. & Pelletier, H. Biochemistry 36, 11205–11215 (1997).
Pelletier, H., Sawaya, M.R., Kumar, A., Wilson, S.H. & Kraut, J. Science 264, 1891–1903 (1994).
Beese, L.S., Friedman, J.M. & Steitz, T.A. Biochemistry 32, 14095–14101 (1993).
Doublie, S., Tabor, S., Long, A.M., Richardson, C.C. & Ellenberger, T. Nature 391, 251–258 (1998).
Bennett, R.A.O., Wilson III, D.M., Wong, D. & Demple, B. Proc. Natl Acad. Sci. USA 94, 7166–7169 (1997).
Kubota, Y. et al. EMBO J. 15, 6662–6670 (1996).
Marintchev, A. et al. Nature Struct. Biol. 6, 884–893 (1999).
Marintchev, A. et al. Nucleic Acids Res. 28, 2049–2059 (2000).
Thompson, L.H. & West, M. G. Mutat. Res. 459, 1–18 (2000).
Showalter, A., Byeon, I.-J.L., Su, M.-I. & Tsai, M.-D. Nature Struct. Biol. 8, 942–946 (2001).
Mullen, G.P., Shenbagamurthi, P. & Mildvan, A.S. J. Biol. Chem. 264, 19637–19647 (1989).
Delaglio, F., et al. J. Biomol. NMR 6, 277–293 (1995).
Bartels, C., Xia, T., Billeter, M., Güntert, P. & Wütrich, K. J. Biomol. NMR 6, 1–10 (1995).
Güntert, P., Mumenthaler, C. & Wüthrich, K. J. Mol. Biol. 273, 283–298 (1997).
Mumenthaler, C., Güntert, P., Braun, W. & Wüthrich, K. J. Biomol. NMR 10, 351–362 (1997).
Cornilescu, G., Delaglio, F. & Bax, A. J. Biomol. NMR 13, 289–302 (1999).
Brünger, A.T. X-PLOR Manual, Version 3.1: A System for X-ray Crystallography and NMR (Yale University Press, New Haven; 1992).
Koradi, R., Billeter, M. & Wüthrich, K. J. Mol. Graphics 14, 51–55 (1996).
Acknowledgements
This research was supported by grants from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maciejewski, M., Shin, R., Pan, B. et al. Solution structure of a viral DNA repair polymerase. Nat Struct Mol Biol 8, 936–941 (2001). https://doi.org/10.1038/nsb1101-936
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb1101-936
This article is cited by
-
A unique DNA-binding mode of African swine fever virus AP endonuclease
Cell Discovery (2020)
-
Structure of the error-prone DNA ligase of African swine fever virus identifies critical active site residues
Nature Communications (2019)
-
How DNA polymerases catalyse replication and repair with contrasting fidelity
Nature Reviews Chemistry (2017)
-
The thumb domain is not essential for the catalytic action of HoLaMa DNA polymerase
The Protein Journal (2017)
-
An automated tool for maximum entropy reconstruction of biomolecular NMR spectra
Nature Methods (2007)