Abstract
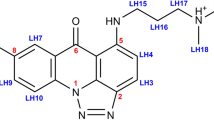
Cryptolepine, a naturally occurring indoloquinoline alkaloid used as an antimalarial drug in Central and Western Africa, has been found to bind to DNA in a formerly unknown intercalation mode. Evidence from competition dialysis assays demonstrates that cryptolepine is able to bind CG-rich sequences containing nonalternating CC sites. Here we show that cryptolepine interacts with the CC sites of the DNA fragment d(CCTAGG)2 in a base-stacking intercalation mode. This is the first DNA intercalator complex, from ∼90 solved by X-ray crystallography, to bind a nonalternating (pyrimidine-pyrimidine) DNA sequence. The asymmetry of the drug induces a perfect stacking with the asymmetric site, allowing for the stability of the complex in the absence of hydrogen bonding interactions. The crystal structure of this antimalarial drug–DNA complex provides evidence for the first nonalternating intercalation and, as such, provides a basis for the design of new anticancer or antimalarial drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rivas, L., Murza, A., Sanchez-Cortes, S. & Garcia-Ramos, J.V. J. Biomol. Struct. Dyn. 18, 371–383 (2000).
Boye, G.I. & Ampofo, O. Proceedings of the first international seminar on cryptolepine (eds Boakye-Yiadom, K. & Bamgbose S.O.A.) 37 (University of Kumasi, Ghana; 1983).
Bonjean, K. et al. Biochemistry 37, 5136–5146 (1998).
Dassonneville, L. et al. Biochemistry 38, 7719–7726 (1999).
Wright, C.W. et al. J. Med. Chem. 44, 3187–3194 (2001).
Arzel, E. et al. J. Med. Chem. 44, 949–960 (2001).
Frederick, C.A. et al. Biochemistry 29, 2538–2549 (1990).
Aymami, J., Nunn, C.M. & Neidle, S. Nucleic Acids Res. 27, 2691–2698 (1999).
White, S., Szewczyk, J.W., Turner, J.M., David, E.E. & Dervan, P.B. Nature 391, 468–471 (1998).
Portugal, J. et al. Curr. Med. Chem. 8, 1–8 (2001).
Hu, G.G. et al. Biochemistry 36, 5940–5946 (1997).
Takenaka, S., Nishira, S., Kondo, H. & Takagi, M. Nucleic Acid Symp. Ser. 27, 71–72 (1992).
Lokey, R.S. et al. J. Am. Chem. Soc. 119,7202–7210 (1997).
Murr, M.M. et al. Bioorg. Med. Chem. 9, 1141–1148 (2001).
Shinomiya, M., Chu, W., Carlson, R.G., Weaver, R.F. & Takusagawa, F. Biochemistry 34, 8481–8491 (1995).
Adams, A., Guss, J.M., Collyer, C.A., Denny, W.A. & Wakelin, L.P.G. Biochemistry 38, 9221–9233 (1999).
Berman, H.M. et al. Biophys. J. 63, 751 (1992).
Kumar, R.A., Ikemoto, N. & Patel. D.J. J. Mol. Biol. 265, 173–186 (1997)
Kumar, R.A., Ikemoto, N. & Patel. D.J. J. Mol. Biol. 265, 187–199 (1997)
Ren, J. & Chaires, J.B. Biochemistry 38, 16067–16075 (1999).
Wright, C.W., Phillipson, J.D., Lisgarten, J.N. & Palmer, R.A. J. Chem. Crystallogr. 29, 449–455 (1999).
Bailly, C. et al. Anticancer Drug Des. 15, 191–201 (2000).
Dean, P.M. & Wakelin, L.P.G. Proc. R. Soc. Lond. B 209, 453–471 (1980).
Haq, I., Ladbury, J.E., Chowdhry, B.Z., Jenkins, T.C. & Chaires, J.B. J. Mol. Biol. 271, 244–257 (1997).
Shieh, H.-S., Berman, H.M., Dabrow, M. & Neidle, S. Nucleic Acids Res. 8, 85–97 (1980).
Boser, P.N. Handbook of biochemistry and molecular biology 3rd edn, Vol 1 Nucleic acids (ed. Fasman, B.G.D.) 589 (CRC Press, Boca Raton, Florida; 1976)
Navaza, J. Acta Crystallogr. A 50, 157 (1994).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905 (1998).
Sheldrick, G.M. The SHELX-97 manual (University of Gottingen, Germany; 1997).
Roussel, A. & Campillau, C. Turbo-Frodo. In Silicon graphics geometry partners directory. 77–79 (Silicon Graphics, Mountain View, California; 1989).
Acknowledgements
This work was supported by grants from the Ministerio de Educacion y Cultura of Spain and the Generalitat de Catalunya. J.N.L. acknowledges support from MEyC of Spain. Synchrotron data collection was supported by the ESRF and EU grants to the EMBL-DESY.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lisgarten, J., Coll, M., Portugal, J. et al. The antimalarial and cytotoxic drug cryptolepine intercalates into DNA at cytosine-cytosine sites. Nat Struct Mol Biol 9, 57–60 (2002). https://doi.org/10.1038/nsb729
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb729
This article is cited by
-
In silico study of HASDI (high-affinity selective DNA intercalator) as a new agent capable of highly selective recognition of the DNA sequence
Scientific Reports (2023)
-
Unravelling the pharmacological properties of cryptolepine and its derivatives: a mini-review insight
Naunyn-Schmiedeberg's Archives of Pharmacology (2023)
-
Alkaloids Rich Extracts from Brown Algae Against Multidrug-Resistant Bacteria by Distinctive Mode of Action
Arabian Journal for Science and Engineering (2022)
-
Silver Nanocrystals Bio-Fabricated Using Rhizobium rhizogenes-Transformed In Vitro Root Extracts Demonstrate Health Proactive Properties
BioNanoScience (2022)
-
Cryptolepine inhibits hepatocellular carcinoma growth through inhibiting interleukin-6/STAT3 signalling
BMC Complementary Medicine and Therapies (2021)