Abstract
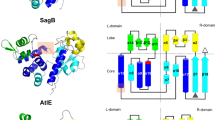
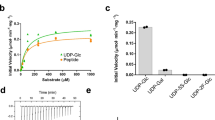
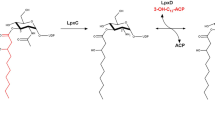
The zinc-dependent UDP-3-O-acyl-N-acetylglucosamine deacetylase (LpxC) catalyzes the first committed step in the biosynthesis of lipid A, the hydrophobic anchor of lipopolysaccharide (LPS) that constitutes the outermost monolayer of Gram-negative bacteria. As LpxC is crucial for the survival of Gram-negative organisms and has no sequence homology to known mammalian deacetylases or amidases, it is an excellent target for the design of new antibiotics. The solution structure of LpxC from Aquifex aeolicus in complex with a substrate-analog inhibitor, TU-514, reveals a novel α/β fold, a unique zinc-binding motif and a hydrophobic passage that captures the acyl chain of the inhibitor. On the basis of biochemical and structural studies, we propose a catalytic mechanism for LpxC, suggest a model for substrate binding and provide evidence that mobility and dynamics in structural motifs close to the active site have key roles in the capture of the substrate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Raetz, C.R.H. & Whitfield, C. Lipopolysaccharide endotoxins. Annu. Rev. Biochem. 71, 635–700 (2002).
Anderson, M.S., Bulawa, C.E. & Raetz, C.R.H. The biosynthesis of Gram- negative endotoxin: formation of lipid A precursors from UDP-GlcNAc in extracts of Escherichia coli. J. Biol. Chem. 260, 15536–15541 (1985).
Anderson, M.S., Robertson, A.D., Macher, I. & Raetz, C.R.H. Biosynthesis of lipid A in Escherichia coli: identification of UDP-3-O-(R-3-hydroxymyristoyl)-α-D-glucosamine as a precursor of UDP-N2,O3-bis(R-3-hydroxymyristoyl)-α-D-glucosamine. Biochemistry 27, 1908–1917 (1988).
Anderson, M.S. et al. UDP-N-acetylglucosamine acyltransferase of Escherichia coli: the first step of endotoxin biosynthesis is thermodynamically unfavorable. J. Biol. Chem. 268, 19858–19865 (1993).
Young, K. et al. The envA permeability/cell division gene of Escherichia coli encodes the second enzyme of lipid A biosynthesis. J. Biol. Chem. 270, 30384–30391 (1995).
Jackman, J.E., Raetz, C.R.H. & Fierke, C.A. UDP-3-O-(R-3-hydroxymyristoyl)-N-acetylglucosamine deacetylase of Escherichia coli is a zinc metalloamidase. Biochemistry 38, 1902–1911 (1999).
Jackman, J.E., Raetz, C.R.H. & Fierke, C.A. Site-directed mutagenesis of the bacterial metalloamidase UDP-(3-O-acyl)-N-acetylglucosamine deacetylase (LpxC). Identification of the zinc binding site. Biochemistry 40, 514–523 (2001).
Onishi, H.R. et al. Antibacterial agents that inhibit lipid A biosynthesis. Science 274, 980–982 (1996).
Jackman, J.E. et al. Antibacterial agents that target lipid A biosynthesis in Gram-negative bacteria. J. Biol. Chem. 275, 11002–11009 (2000).
Pirrung, M. et al. Inhibition of the antibacterial target UDP-(3-O-acyl)-N-acetylglucosamine deacetylase (LpxC): isoxazoline zinc amidase inhibitors bearing diverse metal binding groups. J. Med. Chem. 45, 4359–4370 (2002).
Li, X., Uchiyama, T., Raetz, C.R.H. & Hindsgaul, O. Synthesis of a carbohydrate-derived hydroxamic acid inhibitor of the bacterial enzyme (LpxC) involved in lipid A biosynthesis. Organic Letters 4, 539–541 (2003).
Holm, L. & Sander, C. Protein structure comparison by alignment of distance matrices. J. Mol. Biol. 233, 123–138 (1993).
Williamson, J.M., Anderson, M.S. & Raetz, C.R.H. Acyl–acyl carrier protein specificity of UDP-GlcNAc acyltransferases from Gram-negative bacteria: relationship to lipid A structure. J. Bacteriol. 173, 3591–3596 (1991).
Hyland, S.A., Eveland, S.S. & Anderson, M.S. Cloning, expression, and purification of UDP-3-O-acyl-GlcNAc deacetylase from Pseudomonas aeruginosa: a metalloamidase of the lipid A biosynthesis pathway. J. Bacteriol. 179, 2029–2037 (1997).
Vallee, B.L. & Auld, D.S. Active-site zinc ligands and activated H2O of zinc enzymes. Proc. Natl. Acad. Sci. USA 87, 220–224 (1990).
Vallee, B.L. & Auld, D.S. Zinc coordination, function, and structure of zinc enzymes and other proteins. Biochemistry 29, 5647–5659 (1990).
Lipscomb, W.N. & Sträter, N. Recent advances in zinc enzymology. Chem. Rev. 96, 2375–2433 (1996).
Alberts, I.L., Nadassy, K. & Wodak, S.J. Analysis of zinc binding sites in protein crystal structures. Protein Sci. 7, 1700–1716 (1998).
Christianson, D.W. Carboxypeptidase A. Acc. Chem. Res. 22, 62–69 (1989).
Christianson, D.W. & Cox, J.D. Catalysis by metal-activated hydroxide in zinc and manganese metalloenzymes. Annu. Rev. Biochem. 68, 33–57 (1999).
Cheng, X., Zhang, X., Pflugrath, J.W. & Studier, F.W. The structure of bacteriophage T7 lysozyme, a zinc amidase and an inhibitor of T7 RNA polymerase. Proc. Natl. Acad. Sci. USA 91, 4034–4038 (1994).
Finnin, M.S. et al. Structures of a histone deacetylase homologue bound to the TSA and SAHA inhibitors. Nature 401, 188–193 (1999).
Christianson, D.W. & Alexander, R.S. Carboxylate–histidine–zinc interactions in protein structure and function. J. Am. Chem. Soc. 111, 6412–6419 (1989).
Larsen, K.S. & Auld, D.S. Carboxypeptidase A: mechanism of zinc inhibition. Biochemistry 28, 9620–9625 (1989).
Larsen, K.S. & Auld, D.S. Characterization of an inhibitory metal binding site in carboxypeptidase A. Biochemistry 30, 2613–2618 (1991).
Holland, D.H., Hausrath, A.C., Juers, D. & Matthews, B.W. Structural analysis of zinc substitutions in the active site of thermolysin. Protein Sci. 4, 1955–1965 (1995).
Gomez-Ortiz, M., Gomis-Rüth, F.X., Huber, R. & Aviles, F.X. Inhibition of carboxypeptidase A by excess zinc: analysis of the structural determinants by X-ray crystallography. FEBS Lett. 400, 336–340 (1997).
Brunger, A.T. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998).
LeMaster, D.M. & Richards, F.M. 1H-15N heteronuclear NMR studies of Escherichia coli thioredoxin in samples isotopically labeled by residue type. Biochemistry 24, 7263–7268 (1985).
LeMaster, D.M. & Richards, F.M. NMR sequential assignment of Escherichia coli thioredoxin utilizing random fractional deuteration. Biochemistry 27, 142–150 (1988).
Goto, N.K., Gardner, K.H., Mueller, G.A., Willis, R.C. & Kay, L.E. A robust and cost-effective method for the production of Val, Leu, Ile (δ1) methyl-protonated 15N-, 13C-, 2H-labeled proteins. J. Biomol. NMR 13, 369–374 (1999).
Bartels, C., Xia, T.-H., Billeter, M., Güntert, P. & Wüthrich, K. The program XEASY for computer-supported NMR spectral analysis of biological macromolecules. J. Biomol. NMR 5, 1–10 (1995).
Yamazaki, T., Lee, W., Arrowsmith, C.H., Muhandiram, D.R. & Kay, L.E. A suite of triple resonance NMR experiments for the backbone assignment of 15N, 13C, 2H labeled proteins with high sensitivity. J. Am. Chem. Soc. 116, 11655–11666 (1994).
Yamazaki, T. et al. An HNCA pulse scheme for the backbone assignment of 15N, 13C, 2H-labeled proteins: application to a 37-kDa Trp repressor–DNA complex. J. Am. Chem. Soc. 116, 6464–6465 (1994).
Coggins, B.E. & Zhou, P. PACES: protein sequential assignment by computer-assisted exhaustive search. J. Biomol. NMR 26, 93–111 (2003).
Clore, G.M. & Gronenborn, A.M. Determining the structures of large proteins and protein complexes by NMR. Trends Biotechnol. 16, 22–34 (1998).
Olejniczak, E.T., Xu, R.X., Petros, A.M. & Fesik, S.W. Optimized constant-time 4D HNCAHA and HN(CO)CAHA experiments: applications to the backbone assignments of the FKBP/ascomycin complex. J. Magn. Reson. 100, 444–450 (1992).
Lin, Y. & Wagner, G. Efficient side-chain and backbone assignment in large proteins: application to tGCN5. J. Biomol. NMR 15, 227–239 (1999).
Szyperski, T., Neri, D., Leiting, B., Otting, G. & Wüthrich, K. Support of 1H NMR assignments in proteins by biosynthetically directed fractional 13C-labeling. J. Biomol. NMR 2, 323–334 (1992).
Breeze, A.L. Isotope-filtered NMR methods for the study of biomolecular structure and interactions. Prog. Nucl. Magn. Reson. Spectrosc. 36, 323–372 (2000).
Güntert, P., Mumenthaler, C. & Wüthrich, K. Torsion angle dynamics for NMR structure calculation with the new program DYANA. J. Mol. Biol. 273, 283–298 (1997).
Zwahlen, C. et al. An NMR experiment for measuring methyl–methyl NOEs in 13C-labeled proteins with high resolution. J. Am. Chem. Soc. 120, 7617–7625 (1998).
Cornilescu, G., Delaglio, F. & Bax, A. Protein backbone angle restraints from searching a database for chemical shift and sequence homology. J. Biomol. NMR 13, 289–302 (1999).
Kelly, T.M., Stachula, S.A., Raetz, C.R.H. & Anderson, M.S. The firA gene of Escherichia coli encodes UDP-3-O-(R-3-hydroxymyristoyl)-glucosamine N-acyltransferase: the third step of endotoxin biosynthesis. J. Biol. Chem. 268, 19866–19874 (1993).
Koradi, R., Billeter, M. & Wüthrich, K. MOLMOL: A program for display and analysis of macromolecular structures. J. Mol. Graph. 14, 51–55, 29–32 (1996).
Laskowski, R.A., Rullmann, J.A., MacArthur, M.W., Kaptein, R. & Thornton, J.M. AQUA and PROCHECK-NMR: programs for checking the quality of protein structures solved by NMR. J. Biol. Chem. 8, 477–486 (1996).
Lovell, S.C. et al. Structure validation by Cα geometry: φ, ψ and Cβ deviation. Proteins 50, 437–450 (2003).
Acknowledgements
We thank J. Rudolph for helpful and stimulating discussions. This work was supported by grants from the US National Institutes of Health (NIH) and National Institute of General Medical Sciences (to C.R.H.R.), the NIH and National Institute of Allergy and Infectious Diseases (to P.Z.), the Natural Science and Engineering Research Council of Canada (to O.H.) and the Whitehead Institute (to P.Z.). X.L. is the recipient of a graduate scholarship in carbohydrate chemistry from the Alberta Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Coggins, B., Li, X., McClerren, A. et al. Structure of the LpxC deacetylase with a bound substrate-analog inhibitor. Nat Struct Mol Biol 10, 645–651 (2003). https://doi.org/10.1038/nsb948
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb948
This article is cited by
-
Crystal structure of lipid A disaccharide synthase LpxB from Escherichia coli
Nature Communications (2018)
-
Drug design from the cryptic inhibitor envelope
Nature Communications (2016)
-
Assignment of 1H, 13C and 15N backbone resonances of Escherichia coli LpxC bound to L-161,240
Biomolecular NMR Assignments (2010)