Abstract
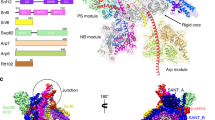
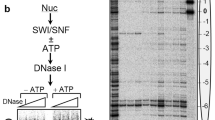
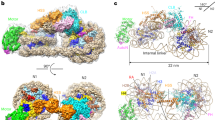
ATP-dependent chromatin-remodeling complexes, such as RSC, can reposition, evict or restructure nucleosomes. A structure of a RSC–nucleosome complex with a nucleosome determined by cryo-EM shows the nucleosome bound in a central RSC cavity. Extensive interaction of RSC with histones and DNA seems to destabilize the nucleosome and lead to an overall ATP-independent rearrangement of its structure. Nucleosomal DNA appears disordered and largely free to bulge out into solution as required for remodeling, but the structure of the RSC–nucleosome complex indicates that RSC is unlikely to displace the octamer from the nucleosome to which it is bound. Consideration of the RSC–nucleosome structure and published biochemical information suggests that ATP-dependent DNA translocation by RSC may result in the eviction of histone octamers from adjacent nucleosomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boeger, H., Griesenbeck, J., Strattan, J.S. & Kornberg, R.D. Nucleosomes unfold completely at a transcriptionally active promoter. Mol. Cell 11, 1587–1598 (2003).
Boeger, H., Griesenbeck, J., Strattan, J.S. & Kornberg, R.D. Removal of promoter nucleosomes by disassembly rather than sliding in vivo. Mol. Cell 14, 667–673 (2004).
Karagiannis, T.C. & El-Osta, A. Chromatin modifications and DNA double-strand breaks: the current state of play. Leukemia 21, 195–200 (2007).
Cairns, B.R. et al. RSC, an abundant and essential chromatin remodeling complex. Cell 87, 1249–1260 (1996).
Lorch, Y., Cairns, B.R., Zhang, M. & Kornberg, R.D. Activated RSC-nucleosome complex and persistently altered form of the nucleosome. Cell 94, 29–34 (1998).
Asturias, F.J., Chung, W.H., Kornberg, R.D. & Lorch, Y. Structural analysis of the RSC chromatin-remodeling complex. Proc. Natl. Acad. Sci. USA 99, 13477–13480 (2002).
Lorch, Y., Zhang, M. & Kornberg, R.D. RSC unravels the nucleosome. Mol. Cell 7, 89–95 (2001).
Lorch, Y., Maier-Davis, B. & Kornberg, R.D. Chromatin remodeling by nucleosome disassembly in vitro. Proc. Natl. Acad. Sci. USA 103, 3090–3093 (2006).
Lorch, Y., Zhang, M. & Kornberg, R.D. Histone octamer transfer by a chromatin-remodeling complex. Cell 96, 389–392 (1999).
Saha, A., Wittmeyer, J. & Cairns, B.R. Chromatin remodeling through directional DNA translocation from an internal nucleosomal site. Nat. Struct. Mol. Biol. 12, 747–755 (2005).
Lorch, Y., Davis, B. & Kornberg, R.D. Chromatin remodeling by DNA bending, not twisting. Proc. Natl. Acad. Sci. USA 102, 1329–1332 (2005).
Zofall, M., Persinger, J., Kassabov, S.R. & Bartholomew, B. Chromatin remodeling by ISW2 and SWI/SNF requires DNA translocation inside the nucleosome. Nat. Struct. Mol. Biol. 13, 339–346 (2006).
Lia, G. et al. Direct observation of DNA distortion by the RSC complex. Mol. Cell 21, 417–425 (2006).
Zhang, Y. et al. DNA translocation and loop formation mechanism of chromatin remodeling by SWI/SNF and RSC. Mol. Cell 24, 559–568 (2006).
Penczek, P.A., Grassucci, R.A. & Frank, J. The ribosome at improved resolution: new techniques for merging and orientation refinement in 3D cryo-electron microscopy of biological particles. Ultramicroscopy 53, 251–270 (1994).
Saxton, W.O. & Baumeister, W. The correlation averaging of a regularly arranged bacterial cell envelope protein. J. Microsc. 127, 127–138 (1982).
Skiniotis, G., Moazed, D. & Walz, T. Acetylated histone tail peptides induce structural rearrangements in the RSC chromatin remodeling complex. J. Biol. Chem. 282, 20804–20808 (2007).
Leschziner, A.E. et al. Conformational flexibility in the chromatin remodeler RSC observed by electron microscopy and the orthogonal tilt reconstruction method. Proc. Natl. Acad. Sci. USA 104, 4913–4918 (2007).
Frank, J. Three-Dimensional Electron Microscopy of Macromolecular Assemblies 342 (Academic Press, San Diego, 1996).
Craighead, J.L., Chang, W.H. & Asturias, F.J. Structure of yeast RNA polymerase II in solution. Implications for enzyme regulation and interaction with promoter DNA. Structure 10, 1117–1125 (2002).
Gao, H., Valle, M., Ehrenberg, M. & Frank, J. Dynamics of EF-G interaction with the ribosome explored by classification of a heterogeneous cryo-EM dataset. J. Struct. Biol. 147, 283–290 (2004).
Leforestier, A., Dubochet, J. & Livolant, F. Bilayers of nucleosome core particles. Biophys. J. 81, 2414–2421 (2001).
Sengupta, S.M. et al. The interactions of yeast SWI/SNF and RSC with the nucleosome before and after chromatin remodeling. J. Biol. Chem. 276, 12636–12644 (2001).
Durr, H., Korner, C., Muller, M., Hickmann, V. & Hopfner, K.P. X-ray structures of the Sulfolobus solfataricus SWI2/SNF2 ATPase core and its complex with DNA. Cell 121, 363–373 (2005).
Dechassa, M.L. et al. Architecture of the SWI/SNF-nucleosome complex. Mol. Cell Biol. 28, 6010–6021 (2008).
Strohner, R. et al. A 'loop recapture' mechanism for ACF-dependent nucleosome remodeling. Nat. Struct. Mol. Biol. 12, 683–690 (2005).
Saeki, H. et al. Linker histone variants control chromatin dynamics during early embryogenesis. Proc. Natl. Acad. Sci. USA 102, 5697–5702 (2005).
Horn, P.J. et al. Phosphorylation of linker histones regulates ATP-dependent chromatin remodeling enzymes. Nat. Struct. Biol. 9, 263–267 (2002).
Boeger, H., Griesenbeck, J. & Kornberg, R.D. Nucleosome retention and the stochastic nature of promoter chromatin remodeling for transcription. Cell 133, 716–726 (2008).
Cairns, B.R. Chromatin remodeling: insights and intrigue from single-molecule studies. Nat. Struct. Mol. Biol. 14, 989–996 (2007).
Dubochet, J. et al. Cryo-electron microscopy of vitrified specimens. Q. Rev. Biophys. 21, 129–228 (1988).
Frank, J. et al. SPIDER and WEB: processing and visualization of images in 3D electron microscopy and related fields. J. Struct. Biol. 116, 190–199 (1996).
Radermacher, M. Three-dimensional reconstruction of single particles from random and nonrandom tilt series. J. Electron Microsc. Tech. 9, 359–394 (1988).
Penczek, P., Radermacher, M. & Frank, J. Three-dimensional reconstruction of single particles embedded in ice. Ultramicroscopy 40, 33–53 (1992).
Borland, L. & Vanheel, M. Classification of image data in conjugate representation spaces. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 7, 601–610 (1990).
Ward, J.H. Hierarchical grouping to optimize an objective function. Am. Stat. Assoc. J. 58, 236–244 (1963).
Pettersen, E.F. et al. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Wriggers, W. & Birmanns, S. Using situs for flexible and rigid-body fitting of multiresolution single-molecule data. J. Struct. Biol. 133, 193–202 (2001).
Luger, K., Mader, A.W., Richmond, R.K., Sargent, D.F. & Richmond, T.J. Crystal structure of the nucleosome core particle at 2.8Å resolution. Nature 389, 251–260 (1997).
Volkmann, N. & Hanein, D. Docking of atomic models into reconstructions from electron microscopy. Methods Enzymol. 374, 204–225 (2003).
Author information
Authors and Affiliations
Contributions
B.M.-D. purified RSC and nucleosomes; Y.L. established optimal conditions and determined affinity for RSC–nucleosome interaction; C.E. and W.-H.C. worked on the initial RSC and RSC–nucleosome cryo–reconstructions; F.Z. collected a portion of the RSC cryo-EM data; Y.C. collected additional cryo-EM data and was responsible for refinement and analysis of the final cryo-EM reconstructions; R.D.K. and F.J.A. interpreted the results and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Methods (PDF 322 kb)
Supplementary Movie 1
Cryo-EM reconstruction of the RSC chromatin remodeling complex. (MOV 556 kb)
Supplementary Movie 2
RSC bottom domain movement viewed from the position close to the front view. (MOV 136 kb)
Supplementary Movie 3
Cryo-EM reconstruction of the RSC-nucleosome complex and comparison with the reconstruction of RSC alone. (MOV 1032 kb)
Supplementary Movie 4
Docking of a model of the histones into the central portion of the density apparent in the RSC-nucleosome reconstruction. (MOV 7913 kb)
Rights and permissions
About this article
Cite this article
Chaban, Y., Ezeokonkwo, C., Chung, WH. et al. Structure of a RSC–nucleosome complex and insights into chromatin remodeling. Nat Struct Mol Biol 15, 1272–1277 (2008). https://doi.org/10.1038/nsmb.1524
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1524
This article is cited by
-
The BAF chromatin remodeler synergizes with RNA polymerase II and transcription factors to evict nucleosomes
Nature Genetics (2024)
-
SWI/SNF Chromatin Remodelers: Structural, Functional and Mechanistic Implications
Cell Biochemistry and Biophysics (2023)
-
Structure of SWI/SNF chromatin remodeller RSC bound to a nucleosome
Nature (2020)
-
Generation of Remosomes by the SWI/SNF Chromatin Remodeler Family
Scientific Reports (2019)
-
Opposing chromatin remodelers control transcription initiation frequency and start site selection
Nature Structural & Molecular Biology (2019)