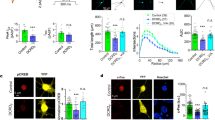
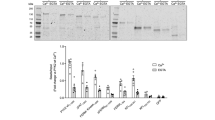
Abstract
In hippocampal neurons, the scaffold protein AKAP79 recruits the phosphatase calcineurin to L-type Ca2+ channels and couples Ca2+ influx to activation of calcineurin and of its substrate, the transcription factor NFAT. Here we show that an IAIIIT anchoring site in human AKAP79 binds the same surface of calcineurin as the PxIxIT recognition peptide of NFAT, albeit more strongly. A modest decrease in calcineurin-AKAP affinity due to an altered anchoring sequence is compatible with NFAT activation, whereas a further decrease impairs activation. Counterintuitively, increasing calcineurin-AKAP affinity increases recruitment of calcineurin to the scaffold but impairs NFAT activation; this is probably due to both slower release of active calcineurin from the scaffold and sequestration of active calcineurin by 'decoy' AKAP sites. We propose that calcineurin-AKAP79 scaffolding promotes NFAT signaling by balancing strong recruitment of calcineurin with its efficient release to communicate with NFAT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Zeke, A., Lukacs, M., Lim, W.A. & Remenyi, A. Scaffolds: interaction platforms for cellular signalling circuits. Trends Cell Biol. 19, 364–374 (2009).
Scott, J.D. & Pawson, T. Cell signaling in space and time: where proteins come together and when they're apart. Science 326, 1220–1224 (2009).
Good, M.C., Zalatan, J.G. & Lim, W.A. Scaffold proteins: hubs for controlling the flow of cellular information. Science 332, 680–686 (2011).
Coghlan, V.M. et al. Association of protein kinase A and protein phosphatase 2B with a common anchoring protein. Science 267, 108–111 (1995).
Logue, J.S. & Scott, J.D. Organizing signal transduction through A-kinase anchoring proteins (AKAPs). FEBS J. 277, 4370–4375 (2010).
Hogan, P.G., Chen, L., Nardone, J. & Rao, A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 17, 2205–2232 (2003).
Molkentin, J.D. & Dorn, G.W. II. Cytoplasmic signaling pathways that regulate cardiac hypertrophy. Annu. Rev. Physiol. 63, 391–426 (2001).
Heineke, J. & Molkentin, J.D. Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat. Rev. Mol. Cell Biol. 7, 589–600 (2006).
Groth, R.D., Dunbar, R.L. & Mermelstein, P.G. Calcineurin regulation of neuronal plasticity. Biochem. Biophys. Res. Commun. 311, 1159–1171 (2003).
Li, H., Rao, A. & Hogan, P.G. Interaction of calcineurin with substrates and targeting proteins. Trends Cell Biol. 21, 91–103 (2011).
Feske, S., Okamura, H., Hogan, P.G. & Rao, A. Ca2+/calcineurin signalling in cells of the immune system. Biochem. Biophys. Res. Commun. 311, 1117–1132 (2003).
Macian, F. NFAT proteins: key regulators of T-cell development and function. Nat. Rev. Immunol. 5, 472–484 (2005).
Aramburu, J. et al. Selective inhibition of NFAT activation by a peptide spanning the calcineurin targeting site of NFAT. Mol. Cell 1, 627–637 (1998).
Aramburu, J. et al. Affinity-driven peptide selection of an NFAT inhibitor more selective than cyclosporin A. Science 285, 2129–2133 (1999).
Li, H., Rao, A. & Hogan, P.G. Structural delineation of the calcineurin–NFAT interaction and its parallels to PP1 targeting interactions. J. Mol. Biol. 342, 1659–1674 (2004).
Li, H., Zhang, L., Rao, A., Harrison, S.C. & Hogan, P.G. Structure of calcineurin in complex with PVIVIT peptide: portrait of a low-affinity signalling interaction. J. Mol. Biol. 369, 1296–1306 (2007).
Takeuchi, K., Roehrl, M.H., Sun, Z.Y. & Wagner, G. Structure of the calcineurin-NFAT complex: defining a T cell activation switch using solution NMR and crystal coordinates. Structure 15, 587–597 (2007).
Roy, J., Li, H., Hogan, P.G. & Cyert, M.S. A conserved docking site modulates substrate affinity for calcineurin, signaling output, and in vivo function. Mol. Cell 25, 889–901 (2007).
Roy, J. & Cyert, M.S. Cracking the phosphatase code: docking interactions determine substrate specificity. Sci. Signal. 2, re9 (2009).
Shi, Y. Serine/threonine phosphatases: mechanism through structure. Cell 139, 468–484 (2009).
Oliveria, S.F., Dell'Acqua, M.L. & Sather, W.A. AKAP79/150 anchoring of calcineurin controls neuronal L-type Ca2+ channel activity and nuclear signaling. Neuron 55, 261–275 (2007).
Dell'Acqua, M.L. et al. Regulation of neuronal PKA signaling through AKAP targeting dynamics. Eur. J. Cell Biol. 85, 627–633 (2006).
Wong, W. & Scott, J.D. AKAP signalling complexes: focal points in space and time. Nat. Rev. Mol. Cell Biol. 5, 959–970 (2004).
Hoshi, N. et al. AKAP150 signaling complex promotes suppression of the M-current by muscarinic agonists. Nat. Neurosci. 6, 564–571 (2003).
Hoshi, N., Langeberg, L.K. & Scott, J.D. Distinct enzyme combinations in AKAP signalling complexes permit functional diversity. Nat. Cell Biol. 7, 1066–1073 (2005).
Tavalin, S.J. et al. Regulation of GluR1 by the A-kinase anchoring protein 79 (AKAP79) signaling complex shares properties with long-term depression. J. Neurosci. 22, 3044–3051 (2002).
Liu, G. et al. Assembly of a Ca2+-dependent BK channel signaling complex by binding to β2 adrenergic receptor. EMBO J. 23, 2196–2205 (2004).
Tunquist, B.J. et al. Loss of AKAP150 perturbs distinct neuronal processes in mice. Proc. Natl. Acad. Sci. USA 105, 12557–12562 (2008).
Graef, I.A. et al. L-type calcium channels and GSK-3 regulate the activity of NF-ATc4 in hippocampal neurons. Nature 401, 703–708 (1999).
Dell'Acqua, M.L., Dodge, K.L., Tavalin, S.J. & Scott, J.D. Mapping the protein phosphatase-2B anchoring site on AKAP79. Binding and inhibition of phosphatase activity are mediated by residues 315–360. J. Biol. Chem. 277, 48796–48802 (2002).
Oliveria, S.F., Gomez, L.L. & Dell'Acqua, M.L. Imaging kinase–AKAP79–phosphatase scaffold complexes at the plasma membrane in living cells using FRET microscopy. J. Cell Biol. 160, 101–112 (2003).
Gorski, J.A., Gomez, L.L., Scott, J.D. & Dell'Acqua, M.L. Association of an A-kinase-anchoring protein signaling scaffold with cadherin adhesion molecules in neurons and epithelial cells. Mol. Biol. Cell 16, 3574–3590 (2005).
Gold, M.G. et al. Molecular basis of AKAP specificity for PKA regulatory subunits. Mol. Cell 24, 383–395 (2006).
Gold, M.G. et al. Architecture and dynamics of an A-kinase anchoring protein 79 (AKAP79) signaling complex. Proc. Natl. Acad. Sci. USA 108, 6426–6431 (2011).
Stemmer, P. & Klee, C.B. Serine/threonine phosphatases in the nervous system. Curr. Opin. Neurobiol. 1, 53–64 (1991).
Salazar, C. & Hofer, T. Allosteric regulation of the transcription factor NFAT1 by multiple phosphorylation sites: a mathematical analysis. J. Mol. Biol. 327, 31–45 (2003).
Bashor, C.J., Horwitz, A.A., Peisajovich, S.G. & Lim, W.A. Rewiring cells: synthetic biology as a tool to interrogate the organizational principles of living systems. Annu. Rev. Biophys. 39, 515–537 (2010).
Pettersen, E.F. et al. UCSF Chimera--a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Alto, N.M. et al. Bioinformatic design of A-kinase anchoring protein-in silico: a potent and selective peptide antagonist of type II protein kinase A anchoring. Proc. Natl. Acad. Sci. USA 100, 4445–4450 (2003).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Brünger, A.T. et al. Crystallography & NMR system: a new software system for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Wang, Z.X. An exact mathematical expression for describing competitive binding of two different ligands to a protein molecule. FEBS Lett. 360, 111–114 (1995).
Roehrl, M.H. et al. Selective inhibition of calcineurin-NFAT signaling by blocking protein–protein interaction with small organic molecules. Proc. Natl. Acad. Sci. USA 101, 7554–7559 (2004).
Gomez, L.L., Alam, S., Smith, K.E., Horne, E. & Dell'Acqua, M.L. Regulation of A-kinase anchoring protein 79/150–cAMP-dependent protein kinase postsynaptic targeting by NMDA receptor activation of calcineurin and remodeling of dendritic actin. J. Neurosci. 22, 7027–7044 (2002).
Robertson, H.R., Gibson, E.S., Benke, T.A. & Dell'Acqua, M.L. Regulation of postsynaptic structure and function by an A-kinase anchoring protein–membrane-associated guanylate kinase scaffolding complex. J. Neurosci. 29, 7929–7943 (2009).
Lippincott-Schwartz, J., Snapp, E. & Kenworthy, A. Studying protein dynamics in living cells. Nat. Rev. Mol. Cell Biol. 2, 444–456 (2001).
Sharma, K., Fong, D.K. & Craig, A.M. Postsynaptic protein mobility in dendritic spines: long-term regulation by synaptic NMDA receptor activation. Mol. Cell. Neurosci. 31, 702–712 (2006).
Acknowledgements
We are grateful to the Department of Neurobiology, Harvard Medical School, for use of its spectrofluorometer, and to S. Lehrer, Boston Biomedical Research Institute, for access to the stopped-flow instrument. Diffraction data were collected at the Advanced Photon Source on Northeastern Collaborative Access Team beamline 24-ID-C, which is supported by award RR15-301 from the National Center for Research Resources at the US National Institutes of Health. Use of the Advanced Photon Source, an Office of Science User Facility operated for the US Department of Energy (DOE) Office of Science by Argonne National Laboratory, was supported by the US DOE under Contract No. DE-AC02-06CH11357. We thank A. Sorkin, University of Pittsburgh, for providing the tandem YFP-CFP construct. The work was supported by US National Institutes of Health grants AI40127 (to A. Rao and P.G.H.), MH080291 (to M.L.D.) and AI090428 (to H.L.). M.D.P. was supported in part by T32NS007083 and by an American Heart Association Predoctoral Fellowship from the Pacific Mountain Affiliate. J.G.M. was supported in part by T32HD041697.
Author information
Authors and Affiliations
Contributions
H.L. prepared the recombinant calcineurin and AKAP79 proteins, carried out the X-ray crystallography, developed the FRET assay for calcineurin-AKAP interaction and performed equilibrium binding measurements. H.L. and P.G.H. analyzed data from these experiments. H.L. and A.S. carried out SEC-MALS measurements. H.L. and P.G.H. performed and analyzed the stopped-flow kinetic experiments. M.D.P., J.G.M. and M.L.D. carried out and analyzed the FRET and YFP/CFP ratio measurements in MDCK cells. M.D.P. and M.L.D. carried out and analyzed the NFAT localization and FRAP experiments with hippocampal neurons. J.G.M. and M.L.D. carried out and analyzed the transcriptional reporter experiments with hippocampal neurons. P.G.H. and H.L. drafted the manuscript, with significant contributions from M.L.D., M.D.P. and J.G.M.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Methods (PDF 1522 kb)
Rights and permissions
About this article
Cite this article
Li, H., Pink, M., Murphy, J. et al. Balanced interactions of calcineurin with AKAP79 regulate Ca2+–calcineurin–NFAT signaling. Nat Struct Mol Biol 19, 337–345 (2012). https://doi.org/10.1038/nsmb.2238
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2238
This article is cited by
-
ATP sulfurylase atypical leucine zipper interacts with Cys3 and calcineurin A in the regulation of sulfur amino acid biosynthesis in Cryptococcus neoformans
Scientific Reports (2023)
-
Calcineurin
Cell Communication and Signaling (2020)
-
A calcineurin–Hoxb13 axis regulates growth mode of mammalian cardiomyocytes
Nature (2020)
-
Selective recruitment of different Ca2+-dependent transcription factors by STIM1-Orai1 channel clusters
Nature Communications (2019)
-
Molecular basis of AKAP79 regulation by calmodulin
Nature Communications (2017)