Abstract
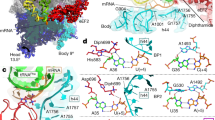
Kasugamycin (Ksg) specifically inhibits translation initiation of canonical but not of leaderless messenger RNAs. Ksg inhibition is thought to occur by direct competition with initiator transfer RNA. The 3.35-Å structure of Ksg bound to the 30S ribosomal subunit presented here provides a structural description of two Ksg-binding sites as well as a basis for understanding Ksg resistance. Notably, neither binding position overlaps with P-site tRNA; instead, Ksg mimics codon nucleotides at the P and E sites by binding within the path of the mRNA. Coupled with biochemical experiments, our results suggest that Ksg indirectly inhibits P-site tRNA binding through perturbation of the mRNA-tRNA codon-anticodon interaction during 30S canonical initiation. In contrast, for 70S-type initiation on leaderless mRNA, the overlap between mRNA and Ksg is reduced and the binding of tRNA is further stabilized by the presence of the 50S subunit, minimizing Ksg efficacy.
*NOTE: In the version of this article initially published, the author name Mikako Shirouzo was spelled incorrectly. The correct author name is Mikako Shirouzu. This error has been corrected in the HTML and PDF versions of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
19 October 2006
Changed 9th author name
References
Umezawa, H., Hamada, M., Suhara, Y., Hashimoto, T. & Ikekawa, T. Kasugamycin, a new antibiotic. Antimicrob. Agents Chemother. 5, 753–757 (1965).
Suhara, Y., Maeda, K. & Umezawa, H. Chemical studies on kasugamycin. V. The structure of kasugamycin. Tetrahedr. Lett. 12, 1239–1244 (1966).
Hamada, M. et al. Antimicrobial activity of kasugamycin. J. Antibiot. 18, 104–106 (1965).
Takeuchi, T. et al. Pharmacology of kasugamycin and the effect on Pseudomonas infection. J. Antibiot. 18, 107–110 (1965).
Tanaka, N., Yoshida, Y., Sashikata, K., Yamaguchi, H. & Umezawa, H. Inhibition of polypeptide synthesis by kasugamycin, an aminoglycosidic antibiotic. J. Antibiot. 19, 65–68 (1966).
Masukawa, H., Tanaka, N. & Umezawa, H. Inhibition by kasugamycin of protein synthesis in Piricularia oryzae. J. Antibiot. 21, 73–74 (1968).
Cassan, M., Berteaux, V., Angrand, P.O. & Rousset, J.P. Expression vectors for quantitating in vivo translational ambiguity: their potential use to analyse frameshifting at the HIV gag-pol junction. Res. Virol. 141, 597–610 (1990).
Van Buul, C.P., Visser, W. & Van Knippenberg, P.H. Increased translational fidelity caused by the antibiotic kasugamycin and ribosomal ambiguity in mutants harboring the ksgA gene. FEBS Lett. 177, 119–124 (1984).
Davies, J., Gilbert, W. & Gorini, L. Streptomycin, suppression, and the code. Proc. Natl. Acad. Sci. USA 51, 883–890 (1964).
Okuyama, A., Machiyama, N., Kinoshita, T. & Tanaka, N. Inhibition by kasugamycin of initiation complex formation on 30S ribosomes. Biochem. Biophys. Res. Commun. 43, 196–199 (1971).
Poldermans, B., Goosen, N. & Van Knippenberg, P.H. Studies on the function of two adjacent N6,N6-dimethyladenosines near the 3′ end of 16S ribosomal RNA of Escherichia coli. I. The effect of kasugamycin on initiation of protein synthesis. J. Biol. Chem. 254, 9085–9089 (1979).
Chin, K., Shean, C.S. & Gottesman, M.E. Resistance of lambda cI translation to antibiotics that inhibit translation initiation. J. Bacteriol. 175, 7471–7473 (1993).
Moll, I. & Bläsi, U. Differential inhibition of 30S and 70S translation initiation complexes on leaderless mRNA by kasugamycin. Biochem. Biophys. Res. Commun. 297, 1021–1026 (2002).
Okuyama, A., Tanaka, N. & Komai, T. The binding of kasugamycin to the Escherichia coli ribosomes. J. Antibiot. 28, 903–905 (1975).
Woodcock, J., Moazed, D., Cannon, M., Davies, J. & Noller, H.F. Interaction of antibiotics with A- and P-site-specific bases in 16S ribosomal RNA. EMBO J. 10, 3099–3103 (1991).
Helser, T.L., Davies, J.E. & Dahlberg, J.E. Change in methylation of 16S ribosomal RNA associated with mutation to kasugamycin resistance in Escherichia coli. Nat. New Biol. 233, 12–14 (1971).
Helser, T.L., Davies, J.E. & Dahlberg, J.E. Mechanism of kasugamycin resistance in Escherichia coli. Nat. New Biol. 235, 6–9 (1972).
Vila-Sanjurjo, A., Squires, C.L. & Dahlberg, A.E. Isolation of kasugamycin resistant mutants in the 16 S ribosomal RNA of Escherichia coli. J. Mol. Biol. 293, 1–8 (1999).
Carter, A.P. et al. Functional insights from the structure of the 30S ribosomal subunit and its interactions with antibiotics. Nature 407, 340–348 (2000).
Pioletti, M. et al. Crystal structures of complexes of the small ribosomal subunit with tetracycline, edeine and IF3. EMBO J. 20, 1829–1839 (2001).
Wilson, D.N. Antibiotics and the inhibition of ribosome function. in Protein Synthesis and Ribosome Structure (eds. Nierhaus, K.H. & Wilson, D.N.) 449–527 (Wiley-VCH, Weinheim, Germany, 2004).
Schuwirth, B.S. et al. Structures of the bacterial ribosome at 3.5 Å resolution. Science 310, 827–834 (2005).
Moazed, D. & Noller, H.F. Interaction of antibiotics with functional sites in 16S ribosomal RNA. Nature 327, 389–394 (1987).
Yusupov, M.M. et al. Crystal structure of the ribosome at 5.5 Å resolution. Science 292, 883–896 (2001).
Jenner, L. et al. Translational operator of mRNA on the ribosome: how repressor proteins exclude ribosome binding. Science 308, 120–123 (2005).
Gnirke, A. & Nierhaus, K.H. tRNA binding sites on the subunits of Escherichia coli ribosomes. J. Biol. Chem. 261, 14506–14514 (1986).
Heus, H.A., Formenoy, L.J. & Van Knippenberg, P.H. Conformational and thermodynamic effects of naturally occurring base methylations in a ribosomal RNA hairpin of Bacillus stearothermophilus. Eur. J. Biochem. 188, 275–281 (1990).
Van Charldorp, R., Verhoeven, J.J., Van Knippenberg, P.H., Haasnoot, C.A. & Hilbers, C.W. A carbon-13 nuclear magnetic resonance study of the 3′-terminus of 16S ribosomal RNA of Escherichia coli specifically labeled with carbon-13 in the methylgroups of the m26Am26A sequence. Nucleic Acids Res. 10, 4237–4245 (1982).
Hobartner, C., Ebert, M., Jaun, B. & Micura, R. RNA two-state conformation equilibria and the effect of nucleobase methylation. Angew. Chem. Int. Edn Engl. 41, 605–609 (2002).
Vila-Sanjurjo, A. & Dahlberg, A.E. Mutational analysis of the conserved bases C1402 and A1500 in the center of the decoding domain of Escherichia coli 16 S rRNA reveals an important tertiary interaction. J. Mol. Biol. 308, 457–463 (2001).
Hirashima, A., Childs, G. & Inouye, M. Differential inhibitory effects of antibiotics on the biosynthesis of envelope proteins of Escherichia coli. J. Mol. Biol. 79, 373–389 (1973).
Tanaka, N., Yamaguchi, H. & Umezawa, H. Mechanism of kasugamycin action on polypeptide synthesis. J. Biochem. 60, 429–434 (1966).
Okuyama, A. & Tanaka, N. Differential effects of aminoglycosides on cistron-specific initiation of protein synthesis. Biochem. Biophys. Res. Commun. 49, 951–957 (1972).
Kozak, M. & Nathans, D. Differential inhibition of coliphage MS2 protein synthesis by ribosome- directed antibiotics. J. Mol. Biol. 70, 41–55 (1972).
Studer, S. & Joseph, S. Unfolding of mRNA secondary structure by the bacterial translation initiation complex. Mol. Cell 22, 105–115 (2006).
de Smit, M.H. & van Duin, J. Control of translation by mRNA secondary structure in Escherichia coli. A quantitative analysis of literature data. J. Mol. Biol. 244, 144–150 (1994).
Moazed, D. & Noller, H.F. Interaction of tRNA with 23S rRNA in the ribosomal A, P, and E sites. Cell 57, 585–597 (1989).
Schäfer, M.A. et al. Codon-anticodon interaction at the P site is a prerequisite for tRNA interaction with the small ribosomal subunit. J. Biol. Chem. 277, 19095–19105 (2002).
Gualerzi, C.O. & Pon, C.L. Initiation of messenger-RNA translation in prokaryotes. Biochemistry 29, 5881–5889 (1990).
Moll, I. et al. Translation initiation with 70S ribosomes: an alternative pathway for leaderless mRNAs. Nucleic Acids Res. 32, 3354–3363 (2004).
Udagawa, T. et al. Evidence for the translation initiation of leaderless mRNAs by the intact 70 S ribosome without its dissociation into subunits in eubacteria. J. Biol. Chem. 279, 8539–8546 (2004).
Blaha, G. et al. Preparation of functional ribosomal complexes and the effect of buffer conditions on tRNA positions observed by cryoelectron microscopy. Methods Enzymol. 317, 292–309 (2000).
Rheinberger, H.-J., Geigenmüller, U., Wedde, M. & Nierhaus, K.H. Parameters for the preparation of Escherichia coli ribosomes and ribosomal subunits active in tRNA binding. Methods Enzymol. 164, 658–670 (1988).
Sharma, M.R. et al. Interaction of Era with the 30S ribosomal subunit: implications for 30S subunit assembly. Mol. Cell 18, 319–329 (2005).
Schluenzen, F. et al. Structure of functionally activated small ribosomal subunit at 3.3 Å resolution. Cell 102, 615–623 (2000).
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Crystallogr. 26, 795–800 (1993).
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D Biol. Crystallogr. 50, 760–763 (1994).
Brunger, A. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Diaconu, M. et al. Structural basis for the function of the ribosomal L7/12 stalk in factor binding and GTPase activation. Cell 121, 991–1004 (2005).
Ogle, J.M. et al. Recognition of cognate transfer RNA by the 30S ribosomal subunit. Science 292, 897–902 (2001).
Acknowledgements
We would like to thank R. Albrecht, J. Buerger, B. Schmidt and M. Nomura for technical assistance and S. Connell for helpful discussions. These studies could not have been performed without the expert assistance of the staff, especially T. Tomizaki and C. Schulze-Briese, at the X06SA beamline (Swiss Light Source). This work was funded by the RIKEN Structural Genomics/Proteomics Initiative and the National Project on Protein Structural and Functional Analyses, Ministry of Education, Culture, Sports, Science and Technology of Japan (S.Y.) and by the Deutsche Forschungs Gemeinschaft (FU579 to P.F.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Stereo image of density for Ksg bound within the primary and secondary sites. (PDF 202 kb)
Supplementary Fig. 2
Phylogenetic conservation of 16S rRNA and comparison of Ksg2 binding site on 30S and 70S ribosomes. (PDF 150 kb)
Supplementary Fig. 3
Position of Ksg relative to Spur-ASL, tRNA and mRNA. (PDF 134 kb)
Supplementary Table 1
Data collection and refinement statistics. (PDF 38 kb)
Rights and permissions
About this article
Cite this article
Schluenzen, F., Takemoto, C., Wilson, D. et al. The antibiotic kasugamycin mimics mRNA nucleotides to destabilize tRNA binding and inhibit canonical translation initiation. Nat Struct Mol Biol 13, 871–878 (2006). https://doi.org/10.1038/nsmb1145
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1145
This article is cited by
-
Direct measurements of mRNA translation kinetics in living cells
Nature Communications (2022)
-
Mechanisms of drug interactions between translation-inhibiting antibiotics
Nature Communications (2020)
-
NMR assignments of the N-terminal domain of Staphylococcus aureus hibernation promoting factor (SaHPF)
Biomolecular NMR Assignments (2018)
-
A Novel Method to Evaluate Ribosomal Performance in Cell-Free Protein Synthesis Systems
Scientific Reports (2017)
-
Heterologous production of kasugamycin, an aminoglycoside antibiotic from Streptomyces kasugaensis, in Streptomyces lividans and Rhodococcus erythropolis L-88 by constitutive expression of the biosynthetic gene cluster
Applied Microbiology and Biotechnology (2017)