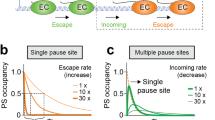
Abstract
We imaged transcription in living cells using a locus-specific reporter system, which allowed precise, single-cell kinetic measurements of promoter binding, initiation and elongation. Photobleaching of fluorescent RNA polymerase II revealed several kinetically distinct populations of the enzyme interacting with a specific gene. Photobleaching and photoactivation of fluorescent MS2 proteins used to label nascent messenger RNAs provided sensitive elongation measurements. A mechanistic kinetic model that fits our data was validated using specific inhibitors. Polymerases elongated at 4.3 kilobases min−1, much faster than previously documented, and entered a paused state for unexpectedly long times. Transcription onset was inefficient, with only 1% of polymerase-gene interactions leading to completion of an mRNA. Our systems approach, quantifying both polymerase and mRNA kinetics on a defined DNA template in vivo with high temporal resolution, opens new avenues for studying regulation of transcriptional processes in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Moore, M.J. From birth to death: the complex lives of eukaryotic mRNAs. Science 309, 1514–1518 (2005).
Shilatifard, A., Conaway, R.C. & Conaway, J.W. The RNA polymerase II elongation complex. Annu. Rev. Biochem. 72, 693–715 (2003).
Cramer, P., Bushnell, D.A. & Kornberg, R.D. Structural basis of transcription: RNA polymerase II at 2.8 angstrom resolution. Science 292, 1863–1876 (2001).
Cramer, P. RNA polymerase II structure: from core to functional complexes. Curr. Opin. Genet. Dev. 14, 218–226 (2004).
Krumm, A., Hickey, L.B. & Groudine, M. Promoter-proximal pausing of RNA polymerase II defines a general rate-limiting step after transcription initiation. Genes Dev. 9, 559–572 (1995).
Dvir, A. Promoter escape by RNA polymerase II. Biochim. Biophys. Acta 1577, 208–223 (2002).
Lescure, B., Williamson, V. & Sentenac, A. Efficient and selective initiation by yeast RNA polymerase B in a dinucleotide-primed reaction. Nucleic Acids Res. 9, 31–45 (1981).
Holstege, F.C., Fiedler, U. & Timmers, H.T. Three transitions in the RNA polymerase II transcription complex during initiation. EMBO J. 16, 7468–7480 (1997).
Kapanidis, A.N. et al. Initial transcription by RNA polymerase proceeds through a DNA-scrunching mechanism. Science 314, 1144–1147 (2006).
Margeat, E. et al. Direct observation of abortive initiation and promoter escape within single immobilized transcription complexes. Biophys. J. 90, 1419–1431 (2006).
Adelman, K. et al. Single molecule analysis of RNA polymerase elongation reveals uniform kinetic behavior. Proc. Natl. Acad. Sci. USA 99, 13538–13543 (2002).
Davenport, R.J., Wuite, G.J., Landick, R. & Bustamante, C. Single-molecule study of transcriptional pausing and arrest by E. coli RNA polymerase. Science 287, 2497–2500 (2000).
O'Brien, T. & Lis, J.T. Rapid changes in Drosophila transcription after an instantaneous heat shock. Mol. Cell. Biol. 13, 3456–3463 (1993).
Tennyson, C.N., Klamut, H.J. & Worton, R.G. The human dystrophin gene requires 16 hours to be transcribed and is cotranscriptionally spliced. Nat. Genet. 9, 184–190 (1995).
Femino, A.M., Fogarty, K., Lifshitz, L.M., Carrington, W. & Singer, R.H. Visualization of single molecules of mRNA in situ. Methods Enzymol. 361, 245–304 (2003).
Bertrand, E. et al. Localization of ASH1 mRNA particles in living yeast. Mol. Cell 2, 437–445 (1998).
Janicki, S.M. et al. From silencing to gene expression: real-time analysis in single cells. Cell 116, 683–698 (2004).
Robinett, C.C. et al. In vivo localization of DNA sequences and visualization of large-scale chromatin organization using lac operator/repressor recognition. J. Cell Biol. 135, 1685–1700 (1996).
Shav-Tal, Y. et al. Dynamics of single mRNPs in nuclei of living cells. Science 304, 1797–1800 (2004).
Femino, A.M., Fay, F.S., Fogarty, K. & Singer, R.H. Visualization of single RNA transcripts in situ. Science 280, 585–590 (1998).
Fusco, D. et al. Single mRNA molecules demonstrate probabilistic movement in living mammalian cells. Curr. Biol. 13, 161–167 (2003).
Bentley, D.L. Rules of engagement: co-transcriptional recruitment of pre-mRNA processing factors. Curr. Opin. Cell Biol. 17, 251–256 (2005).
Darzacq, X. et al. Stepwise RNP assembly at the site of H/ACA RNA transcription in human cells. J. Cell Biol. 173, 207–218 (2006).
Palancade, B. & Bensaude, O. Investigating RNA polymerase II carboxyl-terminal domain (CTD) phosphorylation. Eur. J. Biochem. 270, 3859–3870 (2003).
Phair, R.D. & Misteli, T. Kinetic modelling approaches to in vivo imaging. Nat. Rev. Mol. Cell Biol. 2, 898–907 (2001).
Nguyen, V.T. et al. In vivo degradation of RNA polymerase II largest subunit triggered by alpha-amanitin. Nucleic Acids Res. 24, 2924–2929 (1996).
Becker, M. et al. Dynamic behavior of transcription factors on a natural promoter in living cells. EMBO Rep. 3, 1188–1194 (2002).
Mason, P.B. & Struhl, K. Distinction and relationship between elongation rate and processivity of RNA polymerase II in vivo. Mol. Cell 17, 831–840 (2005).
Boeger, H. et al. Structural basis of eukaryotic gene transcription. FEBS Lett. 579, 899–903 (2005).
Phair, R.D. & Misteli, T. High mobility of proteins in the mammalian cell nucleus. Nature 404, 604–609 (2000).
Beaudouin, J., Mora-Bermudez, F., Klee, T., Daigle, N. & Ellenberg, J. Dissecting the contribution of diffusion and interactions to the mobility of nuclear proteins. Biophys. J. 90, 1878–1894 (2006).
Sprague, B.L. et al. Analysis of binding at a single spatially localized cluster of binding sites by fluorescence recovery after photobleaching. Biophys. J. 91, 1169–1191 (2006).
Hieda, M., Winstanley, H., Maini, P., Iborra, F.J. & Cook, P.R. Different populations of RNA polymerase II in living mammalian cells. Chromosome Res. 13, 135–144 (2005).
Kimura, H., Sugaya, K. & Cook, P.R. The transcription cycle of RNA polymerase II in living cells. J. Cell Biol. 159, 777–782 (2002).
Yamaguchi, Y., Wada, T. & Handa, H. Interplay between positive and negative elongation factors: drawing a new view of DRB. Genes Cells 3, 9–15 (1998).
Peterlin, B.M. & Price, D.H. Controlling the elongation phase of transcription with P-TEFb. Mol. Cell 23, 297–305 (2006).
Darzacq, X., Singer, R.H. & Shav-Tal, Y. Dynamics of transcription and mRNA export. Curr. Opin. Cell Biol. 17, 332–339 (2005).
Epshtein, V. & Nudler, E. Cooperation between RNA polymerase molecules in transcription elongation. Science 300, 801–805 (2003).
Landick, R. The regulatory roles and mechanism of transcriptional pausing. Biochem. Soc. Trans. 34, 1062–1066 (2006).
Liu, L.F. et al. Mechanism of action of camptothecin. Ann. NY Acad. Sci. 922, 1–10 (2000).
Collins, I., Weber, A. & Levens, D. Transcriptional consequences of topoisomerase inhibition. Mol. Cell. Biol. 21, 8437–8451 (2001).
Listerman, I., Sapra, A.K. & Neugebauer, K.M. Cotranscriptional coupling of splicing factor recruitment and precursor messenger RNA splicing in mammalian cells. Nat. Struct. Mol. Biol. 13, 815–822 (2006).
Cheng, C. & Sharp, P.A. RNA polymerase II accumulation in the promoter-proximal region of the dihydrofolate reductase and gamma-actin genes. Mol. Cell. Biol. 23, 1961–1967 (2003).
Eick, D. & Bornkamm, G.W. Transcriptional arrest within the first exon is a fast control mechanism in c-myc gene expression. Nucleic Acids Res. 14, 8331–8346 (1986).
Kao, S.Y., Calman, A.F., Luciw, P.A. & Peterlin, B.M. Anti-termination of transcription within the long terminal repeat of HIV-1 by tat gene product. Nature 330, 489–493 (1987).
Rougvie, A.E. & Lis, J.T. The RNA polymerase II molecule at the 5′ end of the uninduced hsp70 gene of D. melanogaster is transcriptionally engaged. Cell 54, 795–804 (1988).
Saunders, A., Core, L.J. & Lis, J.T. Breaking barriers to transcription elongation. Nat. Rev. Mol. Cell Biol. 7, 557–567 (2006).
Luo, W. & Bentley, D. A ribonucleolytic rat torpedoes RNA polymerase II. Cell 119, 911–914 (2004).
Dundr, M. et al. A kinetic framework for a mammalian RNA polymerase in vivo. Science 298, 1623–1626 (2002).
Miller, O.L., Jr. & Hamkalo, B.A. Visualization of RNA synthesis on chromosomes. Int. Rev. Cytol. 33, 1–25 (1972).
Jackson, D.A., Iborra, F.J., Manders, E.M. & Cook, P.R. Numbers and organization of RNA polymerases, nascent transcripts, and transcription units in HeLa nuclei. Mol. Biol. Cell 9, 1523–1536 (1998).
Struhl, K. Transcriptional noise and the fidelity of initiation by RNA polymerase II. Nat. Struct. Mol. Biol. 14, 103–105 (2007).
Yao, J., Munson, K.M., Webb, W.W. & Lis, J.T. Dynamics of heat shock factor association with native gene loci in living cells. Nature 442, 1050–1053 (2006).
Chubb, J.R., Trcek, T., Shenoy, S.M. & Singer, R.H. Transcriptional pulsing of a developmental gene. Curr. Biol. 16, 1018–1025 (2006).
Shav-Tal, Y., Singer, R.H. & Darzacq, X. Imaging gene expression in single living cells. Nat. Rev. Mol. Cell Biol. 5, 855–861 (2004).
Chartrand, P., Bertrand, E., Singer, R.H. & Long, R.M. Sensitive and high-resolution detection of RNA in situ. Methods Enzymol. 318, 493–506 (2000).
Bregman, D.B., Du, L., van der Zee, S. & Warren, S.L. Transcription-dependent redistribution of the large subunit of RNA polymerase II to discrete nuclear domains. J. Cell Biol. 129, 287–298 (1995).
Bonnet, F., Vigneron, M., Bensaude, O. & Dubois, M.F. Transcription-independent phosphorylation of the RNA polymerase II C-terminal domain (CTD) involves ERK kinases (MEK1/2). Nucleic Acids Res. 27, 4399–4404 (1999).
Akaike, H. A new look at the statistical model identification. IEEE Trans. Automat. Contr. 19, 716–723 (1974).
Schwarz, G. Estimating the dimension of a model. Ann. Stat. 6, 461–464 (1978).
Acknowledgements
We acknowledge the seminal contributions of S. Janicki and D. Spector in developing the cell line. We thank D. Larson and O. Bensaude for discussions on the kinetic modeling, K. Neugebauer for suggesting the use of camptothecin, S. Buhl (Albert Einstein College of Medicine) for hybridoma supernatants containing the H14 and H5 antibodies, and E. Bertrand for sharing unpublished data. This work was online in open peer review in July 2006 on the Nature website. This work was supported by the US National Institutes of Health, National Institute of Biomedical Imaging and Bioengineering, grant EB-002060 to R.H.S. Y.S.-T. is the Jane Stern Lebell Family Fellow in Life Sciences at Bar-Ilan University.
Author information
Authors and Affiliations
Contributions
All data were initially acquired by X.D. and Y.S.-T. Subsequent data were obtained by V.d.T. (Fig. 4a,b and Fig. 5a) and Y.B. (Fig. 9b). S.M.S. was responsible for the microscopy, built the wide-field microscope for live-cell imaging and wrote analysis software. X.D. performed the kinetic modeling. R.D.P. provided consultation on model formulation and testing, and training in the use of the ProcessDB software. R.H.S. supervised the project.
Corresponding author
Ethics declarations
Competing interests
R.D.P. is cofounder and chief science officer of Integrative Bioinformatics, Inc. (IBI) and holds an equity position in IBI.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1, Supplementary Discussion, Supplementary Methods (PDF 194 kb)
Rights and permissions
About this article
Cite this article
Darzacq, X., Shav-Tal, Y., de Turris, V. et al. In vivo dynamics of RNA polymerase II transcription. Nat Struct Mol Biol 14, 796–806 (2007). https://doi.org/10.1038/nsmb1280
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1280
This article is cited by
-
RNA polymerase II depletion from the inactive X chromosome territory is not mediated by physical compartmentalization
Nature Structural & Molecular Biology (2023)
-
Combined SPT and FCS methods reveal a mechanism of RNAP II oversampling in cell nuclei
Scientific Reports (2023)
-
Stepwise modifications of transcriptional hubs link pioneer factor activity to a burst of transcription
Nature Communications (2023)
-
RNA export through the nuclear pore complex is directional
Nature Communications (2022)
-
Direct imaging of intracellular RNA, DNA, and liquid–liquid phase separated membraneless organelles with Raman microspectroscopy
Communications Biology (2022)