Abstract
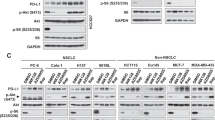
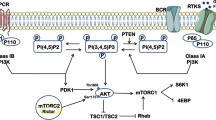
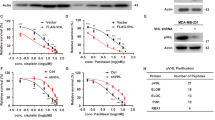
Ectopic expression of mutant forms of phosphatase and tensin homologue deleted on chromosome 10 (PTEN) lacking lipid (G129E) or lipid and protein (C124S) phosphatase activity decreased sensitivity of MCF-7 breast cancer cells, which have wild-type PTEN, to doxorubicin and increased sensitivity to the mammalian target of rapamycin (mTOR) inhibitor rapamycin. Cells transfected with a mutant PTEN gene lacking both lipid and protein phosphatase activities were more resistant to doxorubicin than cells transfected with the PTEN mutant lacking lipid phosphatase activity indicating that the protein phosphatase activity of PTEN was also important in controlling the sensitivity to doxorubicin, while no difference was observed between the lipid (G129E) and lipid and protein (C124S) phosphatase PTEN mutants in terms of sensitivity to rapamycin. A synergistic inhibitory interaction was observed when doxorubicin was combined with rapamycin in the phosphatase-deficient PTEN-transfected cells. Interference with the lipid phosphatase activity of PTEN was sufficient to activate Akt/mTOR/p70S6K signaling. These studies indicate that disruption of the normal activity of the PTEN phosphatase can have dramatic effects on the therapeutic sensitivity of breast cancer cells. Mutations in the key residues which control PTEN lipid and protein phosphatase may act as dominant-negative mutants to suppress endogenous PTEN and alter the sensitivity of breast cancer patients to chemo- and targeted therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Abdel-Rahman MH, Yang Y, Zhou XP, Craig EL, Davidorf FH, Eng C . (2006). High frequency of submicroscopic hemizygous deletion is a major mechanism of loss of expression of PTEN in uveal melanoma. J Clin Oncol 24: 288–295.
Agrawal S, Eng C . (2006). Differential expression of novel naturally occurring splice variants of PTEN and their functional consequences in Cowden syndrome and sporadic breast cancer. Hu Mol Gen 5: 777–787.
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB et al. (1997). Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol 7: 261–269.
Bose S, Crane A, Hibshoosh H, Mansukhani M, Sandweis L, Parsons R . (2002). Reduced expression of PTEN correlates with breast cancer progression. Hu Pathol 33: 405–409.
Chiang GG, Abraham RT . (2005). Phosphorylation of mammalian target of rapamycin (mTOR) at Ser-2448 is mediated by p70S6 kinase. J Biol Chem 280: 25485–25490.
Clark AS, West K, Streicher S, Dennis PA . (2002). Constitutive and inducible Akt activity promotes resistance to chemotherapy, trastuzumab, or tamoxifen in breast cancer cells. Mol Cancer Ther 1: 707–717.
Davis JM, Navolanic PM, Weinstein-Oppenheimer CR, Steelman LS, Hu W, Konopleva M et al. (2003). Raf-1 and Bcl-2 induce distinct and common pathways that contribute to breast cancer drug resistance. Clin Cancer Res 9: 1161–1170.
DeGraffenried LA, Friedrichs WE, Russell DH, Donzis EJ, Middleton AK, Silva JM et al. (2004). Inhibition of mTOR activity restores tamoxifen response in breast cancer cells with aberrant Akt Activity. Clin Cancer Res 23: 8059–8067.
Eng C . (2003). PTEN: one gene, many syndromes. Hu Mut 22: 183–198.
Franke TF, Kaplan DR, Cantley LC, Toker A . (1997). Direct regulation of the Akt proto-oncogene product by phosphatidylinositol-3,4-bisphosphate. Science 275: 665–668.
Frech M, Andjelkovic M, Ingley E, Reddy KK, Falck JR, Hemmings BA . (1997). High affinity binding of inositol phosphates and phosphoinositides to the pleckstrin homology domain of RAC/protein kinase B and their influence on kinase activity. J Biol Chem 272: 8474–8881.
Garcia JM, Silva J, Pena C, Garcia V, Rodríguez R, Cruz MA et al. (2004). Promoter methylation of the PTEN gene is a common molecular change in breast cancer. Genes Chrom Can 41: 117–124.
Inoki K, Corradetti MN, Guan KL . (2005). Dysregulation of the TSC–mTOR pathway in human disease. Nature Gen 37: 19–24.
Jin J, Woodgett JR . (2005). Chronic activation of protein kinase Bbeta/Akt2 leads to multinucleation and cell fusion in human epithelial kidney cells: events associated with tumorigenesis. Oncogene 24: 5459–5470.
Klos KS, Wyszomierski SL, Sun M, Tan M, Zhou X, Li P et al. (2006). ErbB2 increases vascular endothelial growth factor protein synthesis via activation of mammalian target of rapamycin/p70S6K leading to increased angiogenesis and spontaneous metastasis of human breast cancer cells. Cancer Res 66: 2028–2037.
Lee JS, Kim HS, Kim YB, Lee MC, Park CS, Min KW . (2004). Reduced PTEN expression is associated with poor outcome and angiogenesis in invasive ductal carcinoma of the breast. Appl Immunohistochem Mol Morph 12: 205–210.
Lehmann BD, McCubrey JA, Jefferson HS, Paine MS, Chappell WH, Terrian DM . (2007). A dominant role for p53-dependent cellular senescence in radiosensitization of human prostate cancer cells. Cell Cycle 6: 595–605.
Leslie NR, Gray A, Pass I, Orchiston EA, Downes CP . (2000). Analysis of the cellular functions of PTEN using catalytic domain and C-terminal mutations: differential effects of C-terminal deletion on signalling pathways downstream of phosphoinositide 3-kinase. Biochem J 346: 827–833.
Lin HJ, Hsieh FC, Song H, Lin J . (2005). Elevated phosphorylation and activation of PDK-1/AKT pathway in human breast cancer. Br J Cancer 12: 1372–1381.
Luo J, Cantley LC . (2005). The negative regulation of phosphoinositide 3-kinase signaling by p85 and it's implication in cancer. Cell Cycle 4: 1309–1312.
Lynch ED, Ostermeyer EA, Lee MK, Arena JF, Ji H, Dann J et al. (1997). Inherited mutations in PTEN that are associated with breast cancer, Cowden disease, and juvenile polyposis. Am J Hum Genet 61: 1254–1260.
Maehama T, Dixon JE . (1998). The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem 273: 13375–13378.
Marsh DJ, Coulon V, Lunetta KL, Rocca-Serra P, Dahia PL, Zheng Z et al. (1998). Mutation spectrum and genotype–phenotype analyses in Cowden disease and Bannayan–Zonana syndrome, two hamartoma syndromes with germline PTEN mutation. Hu Mol Genet 7: 507–515.
Martelli AM, Nyakern M, Tabellini G, Bortul R, Tazzari PL, Evangelisti C et al. (2006). Phosphoinositide 3-kinase/Akt signaling pathway and its therapeutical implications for human acute myeloid leukemia. Leukemia 20: 911–928.
Martelli AM, Tazzari PL, Evangelisti C, Chiarini F, Blalock WL, Billi AM et al. (2007). Targeting the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin module for acute myelogenous leukemia therapy: from bench to bedside. Curr Med Chem 14: 2009–2023.
Nagata Y, Lan KH, Zhou X, Tan M, Esteva FJ, Sahin AA et al. (2004). PTEN activation contributes to tumor inhibition by trastuzumab, and loss of PTEN predicts trastuzumab resistance in patients. Cancer Cell 6: 117–127.
Shelton JG, Steelman LS, Abrams SL, Bertrand FE, Franklin RA, McMahon M et al. (2005a). Conditional EGFR promotes cell cycle progression and prevention of apoptosis in the absence of autocrine cytokines. Cell Cycle 4: 822–830.
Shelton JG, Steelman LS, Abrams SL, Bertrand FE, Franklin RA, McMahon M et al. (2005b). The epidermal growth factor receptor gene family as a target for therapeutic intervention in numerous cancers: what's genetics got to do with it? Expert Opin Ther Targets 9: 1009–1030.
Singh B, Ittmann MM, Krolewski JJ . (1998). Sporadic breast cancers exhibit loss of heterozygosity on chromosome segment 10q23 close to the Cowden disease locus. Genes Chrom Cancer 21: 166–171.
Steelman LS, Bertrand FE, McCubrey JA . (2004). The complexity of PTEN: mutation, marker and potential target for therapeutic intervention. Expert Opin Ther Targets 8: 537–550.
Stokoe D, Stephens LR, Copeland T, Gaffney PR, Reese CB, Painter GF et al. (1997). Dual role of phosphatidylinositol-3,4,5-trisphosphate in the activation of protein kinase B. Science 277: 567–570.
Tokunaga E, Kimura Y, Mashino K, Oki E, Oki E, Kataoka A et al. (2006). Activation of PI3K/Akt signaling and hormone resistance in breast cancer. Breast Cancer 13: 137–144.
Tsutsui S, Inoue H, Yasuda K, Suzuki K, Higashi H, Era S et al. (2005a). Reduced expression of PTEN protein and its prognostic implications in invasive ductal carcinoma of the breast. Oncology 68: 398–404.
Tsutsui S, Inoue H, Yasuda K, Suzuki K, Tahara K, Higashi H et al. (2005b). Inactivation of PTEN is associated with a low p27Kip1 protein expression in breast carcinoma. Cancer 104: 2048–2053.
Van Nimwegen MJ, Huigsloot H, Camier A, Tijdens IB, van de Water B . (2006). Focal adhesion kinase and protein kinase B cooperate to suppress doxorubicin-induced apoptosis of breast tumor cells. Mol Phamacol 70: 1330–1339.
Weinstein-Oppenheimer CR, Henriquez-Roldan CF, Davis JM, Navolanic PM, Saleh OA, Steelman LS et al. (2001). Role of the Raf signal transduction cascade in the in vitro resistance to the anticancer drug doxorubicin. Clin Cancer Res 7: 2898–2907.
Acknowledgements
This work was supported in part by a grant from the US National Institutes of Health to JAM (R01CA098195). AMM has been supported in part by grants from the CARISBO Foundation and the Progetti Strategici Università di Bologna EF2006.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steelman, L., Navolanic, P., Sokolosky, M. et al. Suppression of PTEN function increases breast cancer chemotherapeutic drug resistance while conferring sensitivity to mTOR inhibitors. Oncogene 27, 4086–4095 (2008). https://doi.org/10.1038/onc.2008.49
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.49
Keywords
This article is cited by
-
Phospho PTEN mediated dephosphorylation of mitotic kinase PLK1 and Aurora Kinase A prevents aneuploidy and preserves genomic stability
Medical Oncology (2023)
-
Macropinocytosis confers resistance to therapies targeting cancer anabolism
Nature Communications (2020)
-
Prospective phase II trial of everolimus in PIK3CA amplification/mutation and/or PTEN loss patients with advanced solid tumors refractory to standard therapy
BMC Cancer (2017)
-
MicroRNA-130b targets PTEN to mediate drug resistance and proliferation of breast cancer cells via the PI3K/Akt signaling pathway
Scientific Reports (2017)
-
Doxorubicin Conjugated to Immunomodulatory Anticancer Lactoferrin Displays Improved Cytotoxicity Overcoming Prostate Cancer Chemo resistance and Inhibits Tumour Development in TRAMP Mice
Scientific Reports (2016)