Abstract
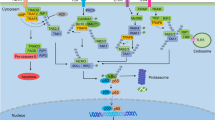
The membrane bound receptor tyrosine kinase Her2 is overexpressed in approximately 30% of human breast cancers, which correlates with poor prognosis. Her2-induced signaling pathways include MAPK and PI3K/Akt, of which the latter has been shown to be critical for Her2+ breast cancer cell growth and survival. In addition, the NF-κB pathway has been shown to be activated downstream of Her2 overexpression; however, the mechanisms leading to this activation are not currently clear. Using Her2+/ER− breast cancer cells, we show that Her2 activates NF-κB through the canonical pathway which, surprisingly, involves IKKα. Knockdown of IKKα led to a significant decrease in transcription levels of multiple NF-κB-regulated cytokine and chemokine genes. siRNA-mediated knockdown of IKKα resulted in a decrease in cancer cell invasion, but had no effect on cell proliferation. Inhibition of the PI3K/Akt pathway had no effect on NF-κB activation, but significantly inhibited cell proliferation. Our study suggests different roles for the NF-κB and PI3K pathways downstream of Her2, leading to changes in invasion and proliferation of breast cancer cells. In addition this work indicates the importance of IKKα as a mediator of Her2-induced tumor progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Arihiro K, Oda H, Kaneko M, Inai K . (2000). Cytokines facilitate chemotactic motility of breast carcinoma cells. Breast Cancer 7: 221–230.
Arora P, Cuevas BD, Russo A, Johnson GL, Trejo J . (2008). Persistent transactivation of EGFR and ErbB2/HER2 by protease-activated receptor-1 promotes breast carcinoma cell invasion. Oncogene 27: 4434–4445.
Baselga J, Tripathy D, Mendelsohn J, Baughman S, Benz CC, Dantis L et al. (1999). Phase II study of weekly intravenous trastuzumab (Herceptin) in patients with HER2/neu-overexpressing metastatic breast cancer. Semin Oncol 26: 78–83.
Basseres DS, Baldwin AS . (2006). Nuclear factor-kappaB and inhibitor of kappaB kinase pathways in oncogenic initiation and progression. Oncogene 25: 6817–6830.
Belguise K, Sonenshein GE. . (2007). PKCtheta promotes c-Rel-driven mammary tumorigenesis in mice and humans by repressing estrogen receptor alpha synthesis. J Clin Invest 117: 4009–4021.
Bhaumik D, Scott GK, Schokrpur S, Patil CK, Campisi J, Benz CC . (2008). Expression of microRNA-146 suppresses NF-kappaB activity with reduction of metastatic potential in breast cancer cells. Oncogene 27: 5643–5647.
Biswas DK, Shi Q, Baily S, Strickland I, Ghosh S, Pardee AB et al. (2004). NF-kappa B activation in human breast cancer specimens and its role in cell proliferation and apoptosis. Proc Natl Acad Sci USA 101: 10137–10142.
Bonizzi G, Karin M . (2004). The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol 25: 280–288.
Cao Y, Luo JL, Karin M . (2007). I{kappa}B kinase {alpha} kinase activity is required for self-renewal of ErbB2/Her2-transformed mammary tumor-initiating cells. Proc Natl Acad Sci USA 104: 15852–15857.
Chavey C, Bibeau F, Gourgou-Bourgade S, Burlinchon S, Boissiere F, Laune D et al. (2007). Oestrogen receptor negative breast cancers exhibit high cytokine content. Breast Cancer Res 9: R15.
Chen D, Xu LG, Chen L, Li L, Zhai Z, Shu HB . (2003). NIK is a component of the EGF/heregulin receptor signaling complexes. Oncogene 22: 4348–4355.
Cogswell PC, Guttridge DC, Funkhouser WK, Baldwin Jr AS . (2000). Selective activation of NF-kappa B subunits in human breast cancer: potential roles for NF-kappa B2/p52 and for Bcl-3. Oncogene 19: 1123–1131.
Dan HC, Cooper MJ, Cogswell PC, Duncan JA, Ting JP, Baldwin AS . (2008). Akt-dependent regulation of NF-{kappa}B is controlled by mTOR and Raptor in association with IKK. Genes Dev 22: 1490–1500.
Dillon RL, White DE, Muller WJ . (2007). The phosphatidyl inositol 3-kinase signaling network: implications for human breast cancer. Oncogene 26: 1338–1345.
Ethier SP, Mahacek ML, Gullick WJ, Frank TS, Weber BL . (1993). Differential isolation of normal luminal mammary epithelial cells and breast cancer cells from primary and metastatic sites using selective media. Cancer Res 53: 627–635.
Galang CK, Garcia-Ramirez J, Solski PA, Westwick JK, Der CJ, Neznanov NN et al. (1996). Oncogenic Neu/ErbB-2 increases ets, AP-1, and NF-kappaB-dependent gene expression, and inhibiting ets activation blocks Neu-mediated cellular transformation. J Biol Chem 271: 7992–7998.
Ghosh S, Karin M . (2002). Missing pieces in the NF-kappaB puzzle. Cell 109 (Suppl): S81–S96.
Gilmore TD . (2006). Introduction to NF-kappaB: players, pathways, perspectives. Oncogene 25: 6680–6684.
Gum R, Wang SW, Lengyel E, Yu D, Hung MC, Juarez J et al. (1995). Upregulation of urokinase-type plasminogen activator expression by the HER2/neu proto-oncogene. Anticancer Res 15: 1167–1172.
Habib AA, Chatterjee S, Park SK, Ratan RR, Lefebvre S, Vartanian T . (2001). The epidermal growth factor receptor engages receptor interacting protein and nuclear factor-kappa B (NF-kappa B)-inducing kinase to activate NF-kappa B. Identification of a novel receptor-tyrosine kinase signalosome. J Biol Chem 276: 8865–8874.
Hayden MS, Ghosh S . (2004). Signaling to NF-kappaB. Genes Dev 18: 2195–2224.
Hegde PS, Rusnak D, Bertiaux M, Alligood K, Strum J, Gagnon R et al. (2007). Delineation of molecular mechanisms of sensitivity to lapatinib in breast cancer cell lines using global gene expression profiles. Mol Cancer Ther 6: 1629–1640.
Hynes NE, Stern DF . (1994). The biology of erbB-2/neu/HER-2 and its role in cancer. Biochim Biophys Acta 1198: 165–184.
Klapper LN, Kirschbaum MH, Sela M, Yarden Y . (2000). Biochemical and clinical implications of the ErbB/HER signaling network of growth factor receptors. Adv Cancer Res 77: 25–79.
Knuefermann C, Lu Y, Liu B, Jin W, Liang K, Wu L et al. (2003). HER2/PI-3K/Akt activation leads to a multidrug resistance in human breast adenocarcinoma cells. Oncogene 22: 3205–3212.
Lam LT, Davis RE, Ngo VN, Lenz G, Wright G, Xu W et al. (2008). Compensatory IKKalpha activation of classical NF-kappaB signaling during IKKbeta inhibition identified by an RNA interference sensitization screen. Proc Natl Acad Sci USA 105: 20798–20803.
Li YM, Pan Y, Wei Y, Cheng X, Zhou BP, Tan M et al. (2004). Upregulation of CXCR4 is essential for HER2-mediated tumor metastasis. Cancer Cell 6: 459–469.
Ma L, Teruya-Feldstein J, Weinberg RA . (2007). Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature 449: 682–688.
Makino K, Day CP, Wang SC, Li YM, Hung MC . (2004). Upregulation of IKKalpha/IKKbeta by integrin-linked kinase is required for HER2/neu-induced NF-kappaB antiapoptotic pathway. Oncogene 23: 3883–3887.
Mayo MW, Wang CY, Cogswell PC, Rogers-Graham KS, Lowe SW, Der CJ et al. (1997). Requirement of NF-kappaB activation to suppress p53-independent apoptosis induced by oncogenic Ras. Science 278: 1812–1815.
Papa S, Bubici C, Zazzeroni F, Pham CG, Kuntzen C, Knabb JR et al. (2006). The NF-kappaB-mediated control of the JNK cascade in the antagonism of programmed cell death in health and disease. Cell Death Differ 13: 712–729.
Pegram MD, Lipton A, Hayes DF, Weber BL, Baselga JM, Tripathy D et al. (1998). Phase II study of receptor-enhanced chemosensitivity using recombinant humanized anti-p185HER2/neu monoclonal antibody plus cisplatin in patients with HER2/neu-overexpressing metastatic breast cancer refractory to chemotherapy treatment. J Clin Oncol 16: 2659–2671.
Pianetti S, Arsura M, Romieu-Mourez R, Coffey RJ, Sonenshein GE . (2001). Her-2/neu overexpression induces NF-kappaB via a PI3-kinase/Akt pathway involving calpain-mediated degradation of IkappaB-alpha that can be inhibited by the tumor suppressor PTEN. Oncogene 20: 1287–1299.
Sakurai H, Chiba H, Miyoshi H, Sugita T, Toriumi W . (1999). IkappaB kinases phosphorylate NF-kappaB p65 subunit on serine 536 in the transactivation domain. J Biol Chem 274: 30353–30356.
She QB, Chandarlapaty S, Ye Q, Lobo J, Haskell KM, Leander KR et al. (2008). Breast tumor cells with PI3K mutation or HER2 amplification are selectively addicted to Akt signaling. PLoS ONE 3: e3065.
Singh S, Shi Q, Bailey ST, Palczewski MJ, Pardee AB, Iglehart JD et al. (2007). Nuclear factor-kappaB activation: a molecular therapeutic target for estrogen receptor-negative and epidermal growth factor receptor family receptor-positive human breast cancer. Mol Cancer Ther 6: 1973–1982.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL . (1987). Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235: 177–182.
Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE et al. (1989). Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244: 707–712.
Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A et al. (2001). Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344: 783–792.
Solt LA, May MJ . (2008). The IkappaB kinase complex: master regulator of NF-kappaB signaling. Immunol Res 42: 3–18.
Steinbrecher KA, Wilson III W, Cogswell PC, Baldwin AS . (2005). Glycogen synthase kinase 3beta functions to specify gene-specific, NF-kappaB-dependent transcription. Mol Cell Biol 25: 8444–8455.
Vazquez-Martin A, Colomer R, Menendez JA . (2008). Her-2/neu-induced ‘cytokine signature’ in breast cancer. Adv Exp Med Biol 617: 311–319.
Verma IM, Stevenson JK, Schwarz EM, Van Antwerp D, Miyamoto S . (1995). Rel/NF-kappa B/I kappa B family: intimate tales of association and dissociation. Genes Dev 9: 2723–2735.
Wang W, Abbruzzese JL, Evans DB, Chiao PJ . (1999). Overexpression of urokinase-type plasminogen activator in pancreatic adenocarcinoma is regulated by constitutively activated RelA. Oncogene 18: 4554–4563.
Weih F, Caamano J . (2003). Regulation of secondary lymphoid organ development by the nuclear factor-kappaB signal transduction pathway. Immunol Rev 195: 91–105.
Wilson III W, Baldwin AS . (2008). Maintenance of constitutive IkappaB kinase activity by glycogen synthase kinase-3alpha/beta in pancreatic cancer. Cancer Res 68: 8156–8163.
Xia W, Mullin RJ, Keith BR, Liu LH, Ma H, Rusnak DW et al. (2002). Anti-tumor activity of GW572016: a dual tyrosine kinase inhibitor blocks EGF activation of EGFR/erbB2 and downstream Erk1/2 and AKT pathways. Oncogene 21: 6255–6263.
Zhou H, Kim YS, Peletier A, McCall W, Earp HS, Sartor CI . (2004). Effects of the EGFR/HER2 kinase inhibitor GW572016 on EGFR- and HER2-overexpressing breast cancer cell line proliferation, radiosensitization, and resistance. Int J Radiat Oncol Biol Phys 58: 344–352.
Acknowledgements
We thank William Comb for his assistance in the preparation of this paper, as well as Dr H Shelton Earp and Dr Carolyn Sartor for generously providing reagents. This work is funded by NIH RO1CA73756 and RO1CA75080, and Department of Defense Grant BC074027. Support is also provided by the Samuel Waxman Cancer Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Merkhofer, E., Cogswell, P. & Baldwin, A. Her2 activates NF-κB and induces invasion through the canonical pathway involving IKKα. Oncogene 29, 1238–1248 (2010). https://doi.org/10.1038/onc.2009.410
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.410
Keywords
This article is cited by
-
KCTD15 deregulation is associated with alterations of the NF-κB signaling in both pathological and physiological model systems
Scientific Reports (2021)
-
RANK signaling increases after anti-HER2 therapy contributing to the emergence of resistance in HER2-positive breast cancer
Breast Cancer Research (2021)
-
RANKL/RANK promotes the migration of gastric cancer cells by interacting with EGFR
Clinical and Translational Medicine (2020)
-
ETS1 is associated with cisplatin resistance through IKKα/NF-κB pathway in cell line MDA-MB-231
Cancer Cell International (2018)
-
A dual role of miR-22 modulated by RelA/p65 in resensitizing fulvestrant-resistant breast cancer cells to fulvestrant by targeting FOXP1 and HDAC4 and constitutive acetylation of p53 at Lys382
Oncogenesis (2018)