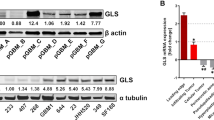
Abstract
Malignant glioma represents one of the most aggressive and lethal human neoplasias. A hallmark of gliomas is their rapid proliferation and destruction of vital brain tissue, a process in which excessive glutamate release by glioma cells takes center stage. Pharmacologic antagonism with glutamate signaling through ionotropic glutamate receptors attenuates glioma progression in vivo, indicating that glutamate release by glioma cells is a prerequisite for rapid glioma growth. Glutamate has been suggested to promote glioma cell proliferation in an autocrine or paracrine manner, in particular by activation of the (RS)-α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid hydrate (AMPA) subtype of glutamate receptors. Here, we dissect the effects of glutamate secretion on glioma progression. Glioma cells release glutamate through the amino-acid antiporter system Xc−, a process that is mechanistically linked with cystine incorporation. We show that disrupting glutamate secretion by interfering with the system Xc− activity attenuates glioma cell proliferation solely cystine dependently, whereas glutamate itself does not augment glioma cell growth in vitro. Neither AMPA receptor agonism nor antagonism affects glioma growth in vitro. On a molecular level, AMPA insensitivity is concordant with a pronounced transcriptional downregulation of AMPA receptor subunits or overexpression of the fully edited GluR2 subunit, both of which block receptor activity. Strikingly, AMPA receptor inhibition in tumor-implanted brain slices resulted in markedly reduced tumor progression associated with alleviated neuronal cell death, suggesting that the ability of glutamate to promote glioma progression strictly requires the tumor microenvironment. Concerning a potential pharmacotherapy, targeting system Xc− activity disrupts two major pathophysiological properties of glioma cells, that is, the induction of excitotoxic neuronal cell death and incorporation of cystine required for rapid proliferation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
22 February 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41388-023-02634-5
References
Behrens PF, Langemann H, Strohschein R, Draeger J, Hennig J . (2000). Extracellular glutamate and other metabolites in and around RG2 rat glioma: an intracerebral microdialysis study. J Neurooncol 47: 11–22.
Brusa R, Zimmermann F, Koh DS, Feldmeyer D, Gass P, Seeburg PH et al. (1995). Early-onset epilepsy and postnatal lethality associated with an editing-deficient GluR-B allele in mice. Science 270: 1677–1680.
Chung WJ, Lyons SA, Nelson GM, Hamza H, Gladson CL, Gillespie GY et al. (2005). Inhibition of cystine uptake disrupts the growth of primary brain tumors. J Neurosci 25: 7101–7110.
Chung WJ, Sontheimer H . (2009). Sulfasalazine inhibits the growth of primary brain tumors independent of nuclear factor-kappaB. J Neurochem 110: 182–193.
de Groot JF, Piao Y, Lu L, Fuller GN, Yung WK . (2008). Knockdown of GluR1 expression by RNA interference inhibits glioma proliferation. J Neurooncol 88: 121–133.
DeAngelis LM . (2001). Brain tumors. N Engl J Med 344: 114–123.
Donevan SD, Rogawski MA . (1993). GYKI 52466, a 2,3-benzodiazepine, is a highly selective, noncompetitive antagonist of AMPA/kainate receptor responses. Neuron 10: 51–59.
Eyupoglu IY, Hahnen E, Buslei R, Siebzehnrubl FA, Savaskan NE, Luders M et al. (2005a). Suberoylanilide hydroxamic acid (SAHA) has potent anti-glioma properties in vitro, ex vivo and in vivo. J Neurochem 93: 992–999.
Eyupoglu IY, Hahnen E, Heckel A, Siebzehnrubl FA, Buslei R, Fahlbusch R et al. (2005b). Malignant glioma-induced neuronal cell death in an organotypic glioma invasion model. Technical note. J Neurosurg 102: 738–744.
Eyupoglu IY, Hahnen E, Trankle C, Savaskan NE, Siebzehnrubl FA, Buslei R et al. (2006). Experimental therapy of malignant gliomas using the inhibitor of histone deacetylase MS-275. Mol Cancer Ther 5: 1248–1255.
Furnari FB, Fenton T, Bachoo RM, Mukasa A, Stommel JM, Stegh A et al. (2007). Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes Dev 21: 2683–2710.
Huang Y, Dai Z, Barbacioru C, Sadee W . (2005). Cystine-glutamate transporter SLC7A11 in cancer chemosensitivity and chemoresistance. Cancer Res 65: 7446–7454.
Ishiuchi S, Tsuzuki K, Yoshida Y, Yamada N, Hagimura N, Okado H et al. (2002). Blockage of Ca(2+)-permeable AMPA receptors suppresses migration and induces apoptosis in human glioblastoma cells. Nat Med 8: 971–978.
Jones DP, Go YM, Anderson CL, Ziegler TR, Kinkade Jr JM, Kirlin WG . (2004). Cysteine/cystine couple is a newly recognized node in the circuitry for biologic redox signaling and control. FASEB J 18: 1246–1248.
Liu RR, Brown CE, Murphy TH . (2007). Differential regulation of cell proliferation in neurogenic zones in mice lacking cystine transport by xCT. Biochem Biophys Res Commun 364: 528–533.
Lomeli H, Mosbacher J, Melcher T, Hoger T, Geiger JR, Kuner T et al. (1994). Control of kinetic properties of AMPA receptor channels by nuclear RNA editing. Science 266: 1709–1713.
Lu W, Shi Y, Jackson AC, Bjorgan K, During MJ, Sprengel R et al. (2009). Subunit composition of synaptic AMPA receptors revealed by a single-cell genetic approach. Neuron 62: 254–268.
Lyons SA, Chung WJ, Weaver AK, Ogunrinu T, Sontheimer H . (2007). Autocrine glutamate signaling promotes glioma cell invasion. Cancer Res 67: 9463–9471.
Maas S, Patt S, Schrey M, Rich A . (2001). Underediting of glutamate receptor GluR-B mRNA in malignant gliomas. Proc Natl Acad Sci USA 98: 14687–14692.
Marcus HJ, Carpenter KL, Price SJ, Hutchinson PJ . (2010). In vivo assessment of high-grade glioma biochemistry using microdialysis: a study of energy-related molecules, growth factors and cytokines. J Neurooncol 97: 11–23.
Mawatari K, Yasui Y, Sugitani K, Takadera T, Kato S . (1996). Reactive oxygen species involved in the glutamate toxicity of C6 glioma cells via xc antiporter system. Neuroscience 73: 201–208.
Mosbacher J, Schoepfer R, Monyer H, Burnashev N, Seeburg PH, Ruppersberg JP . (1994). A molecular determinant for submillisecond desensitization in glutamate receptors. Science 266: 1059–1062.
Ohgaki H, Kleihues P . (2005). Epidemiology and etiology of gliomas. Acta Neuropathol (Berl) 109: 93–108.
Patel SA, Warren BA, Rhoderick JF, Bridges RJ . (2004). Differentiation of substrate and non-substrate inhibitors of transport system xc(–): an obligate exchanger of L-glutamate and L-cystine. Neuropharmacology 46: 273–284.
Pham AN, Blower PE, Alvarado O, Ravula R, Gout PW, Huang Y . (2010). Pharmacogenomic approach reveals a role for the x(c)– cystine/glutamate antiporter in growth and celastrol resistance of glioma cell lines. J Pharmacol Exp Ther 332: 949–958.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D et al. (2004). ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6: 1–6.
Robe PA, Martin DH, Nguyen-Khac MT, Artesi M, Deprez M, Albert A et al. (2009). Early termination of ISRCTN45828668, a phase 1/2 prospective, randomized study of sulfasalazine for the treatment of progressing malignant gliomas in adults. BMC Cancer 9: 372.
Sato H, Shiiya A, Kimata M, Maebara K, Tamba M, Sakakura Y et al. (2005). Redox imbalance in cystine/glutamate transporter-deficient mice. J Biol Chem 280: 37423–37429.
Savaskan NE, Hahnen E, Eyupoglu IY . (2009). The x(c)(–) cystine/glutamate antiporter (xCT) as a potential target for therapy of cancer: yet another cytotoxic anticancer approach? J Cell Physiol 220: 531–532; author reply 533–534.
Savaskan NE, Heckel A, Hahnen E, Engelhorn T, Doerfler A, Ganslandt O et al. (2008). Small interfering RNA-mediated xCT silencing in gliomas inhibits neurodegeneration and alleviates brain edema. Nat Med 14: 629–632.
Savaskan NE, Rocha L, Kotter MR, Baer A, Lubec G, van Meeteren LA et al. (2007). Autotaxin (NPP-2) in the brain: cell type-specific expression and regulation during development and after neurotrauma. Cell Mol Life Sci 64: 230–243.
Schwenk J, Harmel N, Zolles G, Bildl W, Kulik A, Heimrich B et al. (2009). Functional proteomics identify cornichon proteins as auxiliary subunits of AMPA receptors. Science 323: 1313–1319.
Shih AY, Erb H, Sun X, Toda S, Kalivas PW, Murphy TH . (2006). Cystine/glutamate exchange modulates glutathione supply for neuroprotection from oxidative stress and cell proliferation. J Neurosci 26: 10514–10523.
Sun L, Hui AM, Su Q, Vortmeyer A, Kotliarov Y, Pastorino S et al. (2006). Neuronal and glioma-derived stem cell factor induces angiogenesis within the brain. Cancer Cell 9: 287–300.
Takano T, Lin JH, Arcuino G, Gao Q, Yang J, Nedergaard M . (2001). Glutamate release promotes growth of malignant gliomas. Nat Med 7: 1010–1015.
Turski L, Huth A, Sheardown M, McDonald F, Neuhaus R, Schneider HH et al. (1998). ZK200775: a phosphonate quinoxalinedione AMPA antagonist for neuroprotection in stroke and trauma. Proc Natl Acad Sci USA 95: 10960–10965.
van Vuurden DG, Yazdani M, Bosma I, Broekhuizen AJ, Postma TJ, Heimans JJ et al. (2009). Attenuated AMPA receptor expression allows glioblastoma cell survival in glutamate-rich environment. PLoS One 4: e5953.
Vanhoutte N, Abarca-Quinones J, Jordan BF, Gallez B, Maloteaux JM, Hermans E . (2009). Enhanced expression of the high affinity glutamate transporter GLT-1 in C6 glioma cells delays tumour progression in rat. Exp Neurol 218: 56–63.
Ye ZC, Rothstein JD, Sontheimer H . (1999). Compromised glutamate transport in human glioma cells: reduction-mislocalization of sodium-dependent glutamate transporters and enhanced activity of cystine-glutamate exchange. J Neurosci 19: 10767–10777.
Ye ZC, Sontheimer H . (1999). Glioma cells release excitotoxic concentrations of glutamate. Cancer Res 59: 4383–4391.
Yoshida Y, Tsuzuki K, Ishiuchi S, Ozawa S . (2006). Serum-dependence of AMPA receptor-mediated proliferation in glioma cells. Pathol Int 56: 262–271.
Zhuge J, Cederbaum AI . (2006). Serum deprivation-induced HepG2 cell death is potentiated by CYP2E1. Free Radic Biol Med 40: 63–74.
Acknowledgements
We thank Nadine Scheufler and Philip Rummel (both Erlangen) for experimental support. Jan Csupor (Berlin) is acknowledged for technical assistance. This study was supported by the ‘Wilhelm Sander-Stiftung’ (to EH), the ‘Institut Danone’ (to NES, IYE and EH), the ‘International Human Frontiers Science Program’ (to NES), the ‘ELAN-Programm’ (to IYE), the ‘Köln Fortune Programm’ (to EH) and the ‘Center for Molecular Medicine Cologne (CMMC)’ (to EH).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Savaskan, N., Seufert, S., Hauke, J. et al. Dissection of mitogenic and neurodegenerative actions of cystine and glutamate in malignant gliomas. Oncogene 30, 43–53 (2011). https://doi.org/10.1038/onc.2010.391
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.391
Keywords
This article is cited by
-
ATF4 promotes angiogenesis and neuronal cell death and confers ferroptosis in a xCT-dependent manner
Oncogene (2017)
-
MIF-CD74 signaling impedes microglial M1 polarization and facilitates brain tumorigenesis
Oncogene (2016)
-
Histone deacetylases inhibition by SAHA/Vorinostat normalizes the glioma microenvironment via xCT equilibration
Scientific Reports (2014)
-
Glutamate transporters in the biology of malignant gliomas
Cellular and Molecular Life Sciences (2014)
-
Activation of NMDA receptor of glutamate influences MMP-2 activity and proliferation of glioma cells
Neurological Sciences (2014)