Abstract
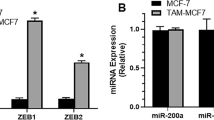
Many estrogen receptor (ER)-positive breast cancers respond well initially to endocrine therapies, but often develop resistance during treatment with selective ER modulators (SERMs) such as tamoxifen. We have reported that the 14-3-3 family member and conserved protein, 14-3-3ζ, is upregulated by tamoxifen and that high expression correlated with an early time to disease recurrence. However, the mechanism by which tamoxifen upregulates 14-3-3ζ and may promote the development of endocrine resistance is not known. Our findings herein reveal that the tamoxifen upregulation of 14-3-3ζ results from its ability to rapidly downregulate microRNA (miR)-451 that specifically targets 14-3-3ζ. The levels of 14-3-3ζ and miR-451 were inversely correlated, with 14-3-3ζ being elevated and miR-451 being at a greatly reduced level in tamoxifen-resistant breast cancer cells. Of note, downregulation of miR-451 was selectively elicited by tamoxifen but not by other SERMs, such as raloxifene or ICI182,780 (Fulvestrant). Increasing the level of miR-451 by overexpression, which decreased 14-3-3ζ, suppressed cell proliferation and colony formation, markedly reduced activation of HER2, EGFR and MAPK signaling, increased apoptosis, and, importantly, restored the growth-inhibitory effectiveness of SERMs in endocrine-resistant cells. Opposite effects were elicited by miR-451 knockdown. Thus, we identify tamoxifen downregulation of miR-451, and consequent elevation of the key survival factor 14-3-3ζ, as a mechanistic basis of tamoxifen-associated development of endocrine resistance. These findings suggest that therapeutic approaches to increase expression of this tumor suppressor-like miR should be considered to downregulate 14-3-3ζ and enhance the effectiveness of endocrine therapies. Furthermore, the selective ability of the SERM tamoxifen but not raloxifene to regulate miR-451 and 14-3-3ζ may assist in understanding differences in their activities, as seen in the STAR (Study of Tamoxifen and Raloxifene) breast cancer prevention trial and in other clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- ER:
-
estrogen receptor
- ICI:
-
ICI 182,780, fulvestrant
- miR:
-
microRNA
- Ral:
-
raloxifene
- SERM:
-
selective estrogen receptor modulator
- Tam:
-
tamoxifen
References
Allgood VE, Zhang Y, O'Malley BW, Weigel NL . (1997). Analysis of chicken progesterone receptor function and phosphorylation using an adenovirus-mediated procedure for high-efficiency DNA transfer. Biochemistry 36: 224–232.
Ando K, Ozaki T, Yamamoto H, Furuya K, Hosoda M, Hayashi S et al. (2004). Polo-like kinase 1 (Plk1) inhibits p53 function by physical interaction and phosphorylation. J Biol Chem 279: 25549–25561.
Arpino G, Wiechmann L, Osborne CK, Schiff R . (2008). Crosstalk between the estrogen receptor and the HER tyrosine kinase receptor family: molecular mechanism and clinical implications for endocrine therapy resistance. Endocr Rev 29: 217–233.
Bandres E, Bitarte N, Arias F, Agorreta J, Fortes P, Agirre X et al. (2009). microRNA-451 regulates macrophage migration inhibitory factor production and proliferation of gastrointestinal cancer cells. Clin Cancer Res 15: 2281–2290.
Bartel DP . (2004). MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116: 281–297.
Bergamaschi A, Frasor J, Kieser K, Wiley E, Katzenellenbogen B . (2009). A breast cancer gene signature associated with high expression of 14-3-3z predicts poor clinical outcome on endocrine therapy. Proc Am Assoc Cancer Res Annu Meet 2: 2854.
Chang EC, Charn TH, Park SH, Helferich WG, Komm B, Katzenellenbogen JA et al. (2008). Estrogen receptors alpha and beta as determinants of gene expression: influence of ligand, dose, and chromatin binding. Mol Endocrinol 22: 1032–1043.
Costa RH . (2005). FoxM1 dances with mitosis. Nat Cell Biol 7: 108–110.
Creighton CJ, Hilger AM, Murthy S, Rae JM, Chinnaiyan AM, El-Ashry D . (2006). Activation of mitogen-activated protein kinase in estrogen receptor alpha-positive breast cancer cells in vitro induces an in vivo molecular phenotype of estrogen receptor alpha-negative human breast tumors. Cancer Res 66: 3903–3911.
Croce CM . (2009). Causes and consequences of microRNA dysregulation in cancer. Nat Rev Genet 10: 704–714.
Dore LC, Amigo JD, Dos Santos CO, Zhang Z, Gai X, Tobias JW et al. (2008). A GATA-1-regulated microRNA locus essential for erythropoiesis. Proc Natl Acad Sci USA 105: 3333–3338.
Dunn BK, Ford LG . (2001). From adjuvant therapy to breast cancer prevention: BCPT and STAR. Breast J 7: 144–157.
EBCTCG (2005). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365: 1687–1717.
Filipowicz W, Bhattacharyya SN, Sonenberg N . (2008). Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9: 102–114.
Frasor J, Chang EC, Komm B, Lin CY, Vega VB, Liu ET et al. (2006). Gene expression preferentially regulated by tamoxifen in breast cancer cells and correlations with clinical outcome. Cancer Res 66: 7334–7340.
Frasor J, Stossi F, Danes JM, Komm B, Lyttle CR, Katzenellenbogen BS . (2004). Selective estrogen receptor modulators: discrimination of agonistic versus antagonistic activities by gene expression profiling in breast cancer cells. Cancer Res 64: 1522–1533.
Green KA, Carroll JS . (2007). Oestrogen-receptor-mediated transcription and the influence of co-factors and chromatin state. Nat Rev 7: 713–722.
Guo H, Ingolia NT, Weissman JS, Bartel DP . (2010). Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 466: 835–840.
Harfe BD . (2005). MicroRNAs in vertebrate development. Curr Opin Genet Dev 15: 410–415.
Herman ME, Katzenellenbogen BS . (1996). Response-specific antiestrogen resistance in a newly characterized MCF-7 human breast cancer cell line resulting from long-term exposure to trans-hydroxytamoxifen. J Steroid Biochem Mol Biol 59: 121–134.
Jaiyesimi IA, Buzdar AU, Decker DA, Hortobagyi GN . (1995). Use of tamoxifen for breast cancer: twenty-eight years later. J Clin Oncol 13: 513–529.
Katzenellenbogen BS, Katzenellenbogen JA . (2002). Biomedicine. Defining the ‘S’ in SERMs. Science 295: 2380–2381.
Katzenellenbogen JA, O'Malley BW, Katzenellenbogen BS . (1996). Tripartite steroid hormone receptor pharmacology: interaction with multiple effector sites as a basis for the cell- and promoter-specific action of these hormones. Mol Endocrinol 10: 119–131.
Li Z, Zhao J, Du Y, Park HR, Sun SY, Bernal-Mizrachi L et al. (2008). Down-regulation of 14-3-3zeta suppresses anchorage-independent growth of lung cancer cells through anoikis activation. Proc Natl Acad Sci USA 105: 162–167.
Lowery AJ, Miller N, McNeill RE, Kerin MJ . (2008). MicroRNAs as prognostic indicators and therapeutic targets: potential effect on breast cancer management. Clin Cancer Res 14: 360–365.
Masaki S, Ohtsuka R, Abe Y, Muta K, Umemura T . (2007). Expression patterns of microRNAs 155 and 451 during normal human erythropoiesis. Biochem Biophys Res Commun 364: 509–514.
Massarweh S, Osborne CK, Creighton CJ, Qin L, Tsimelzon A, Huang S et al. (2008). Tamoxifen resistance in breast tumors is driven by growth factor receptor signaling with repression of classic estrogen receptor genomic function. Cancer Res 68: 826–833.
McPherson RA, Harding A, Roy S, Lane A, Hancock JF . (1999). Interactions of c-Raf-1 with phosphatidylserine and 14-3-3. Oncogene 18: 3862–3869.
Neal CL, Yao J, Yang W, Zhou X, Nguyen NT, Lu J et al. (2009). 14-3-3 {zeta} overexpression defines high risk for breast. Cancer Res 69: 3425–3432.
Oksvold MP, Huitfeldt HS, Langdon WY . (2004). Identification of 14-3-3[zeta] as an EGF receptor interacting protein. FEBS Lett 569: 207–210.
Osborne CK . (1998). Tamoxifen in the treatment of breast cancer. New Engl J Med 339: 1609–1618.
Patrick DM, Zhang CC, Tao Y, Yao H, Qi X, Schwartz RJ et al. (2010). Defective erythroid differentiation in miR-. Genes Dev 24: 1614–1619.
Riccardi C, Nicoletti I . (2006). Analysis of apoptosis by propidium iodide staining and flow cytometry. Nat Protocols 1: 1458–1461.
Shan G, Li Y, Zhang J, Li W, Szulwach KE, Duan R et al. (2008). A small molecule enhances RNA interference and promotes microRNA processing. Nat Biotechnol 26: 933–940.
Shang Y, Brown M . (2002). Molecular determinants for the tissue specificity of SERMs. Science 295: 2465–2468.
Tzivion G, Gupta VS, Kaplun L, Balan V . (2006). 14-3-3 proteins as potential oncogenes. Semin Cancer Biol 16: 203–213.
Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN et al. (2006). Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial. JAMA 295: 2727–2741.
Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN et al. (2010). Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 trial: preventing breast cancer. Cancer Prev Res 3: 696–706.
Welboren WJ, van Driel MA, Janssen-Megens EM, van Heeringen SJ, Sweep FC, Span PN et al. (2009). ChIP-Seq of ERalpha and RNA polymerase II defines genes differentially responding to ligands. Embo J 28: 1418–1428.
Williams AE, Perry MM, Moschos SA, Lindsay MA . (2007). microRNA expression in the aging mouse lung. BMC Genomics 8: 172.
Yang JS, Maurin T, Robine N, Rasmussen KD, Jeffrey KL, Chandwani R et al. (2010). Conserved vertebrate mir-451 provides a platform for Dicer-independent, Ago2-mediated microRNA biogenesis. Proc Natl Acad Sci USA 107: 15163–15168.
Yu D, Dos Santos CO, Zhao G, Jiang J, Amigo JD, Khandros E et al. (2010). miR-451 protects against erythroid oxidant stress by repressing 14-3-3 {zeta}. Genes Dev 24: 1620–1633.
Acknowledgements
This research was supported by grants from the Breast Cancer Research Foundation (BSK) and the NIH (P01AG024387 and P50 AT006268, BSK), and a Postdoctoral Fellowship from the Department of Defense (W81XWH-09-1-0398, AB). We thank Dr Nancy Weigel, Baylor College of Medicine, for providing lysine-coated adenovirus.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Bergamaschi, A., Katzenellenbogen, B. Tamoxifen downregulation of miR-451 increases 14-3-3ζ and promotes breast cancer cell survival and endocrine resistance. Oncogene 31, 39–47 (2012). https://doi.org/10.1038/onc.2011.223
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.223
Keywords
This article is cited by
-
Multi-time scale transcriptomic analysis on the dynamic process of tamoxifen resistance development in breast cancer cell lines
Breast Cancer (2022)
-
Resistance to endocrine therapy in HR + and/or HER2 + breast cancer: the most promising predictive biomarkers
Molecular Biology Reports (2022)
-
Small non-coding RNA profiling in breast cancer: plasma U6 snRNA, miR-451a and miR-548b-5p as novel diagnostic and prognostic biomarkers
Molecular Biology Reports (2022)
-
High expression of eIF4E is associated with tumor macrophage infiltration and leads to poor prognosis in breast cancer
BMC Cancer (2021)
-
Lnc-DC promotes estrogen independent growth and tamoxifen resistance in breast cancer
Cell Death & Disease (2021)