Abstract
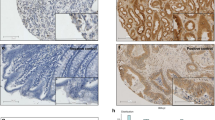
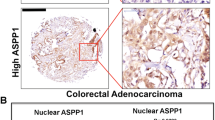
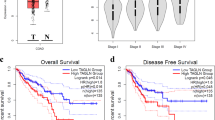
CTEN/TNS4 is an oncogene in colorectal cancer (CRC), which can induce cell motility although its mechanistic basis of activity and the clinical implications of Cten expression are unknown. As Cten is in complex with integrins at focal adhesions, we hypothesised that it may interact with integrin-linked kinase (ILK). Through forced expression and knockdown of Cten in HCT116 and SW620 (respectively, showing low and high Cten expression), we showed that Cten could regulate ILK. However, inhibition of ILK after forced expression of Cten abrogated the motility-inducing effects of Cten, thereby demonstrating that the Cten–ILK interaction was functionally relevant. Combined knockdown of Cten and ILK had no additive effects on cell motility compared with knockdown of each individually. In order to investigate the clinical implications of Cten expression, a series of 462 CRCs were evaluated by immunohistochemistry. High expression of Cten was associated with advanced Dukes’ stage (P<0.001), poor prognosis (P<0.001) and distant metastasis (P=0.008). The role of Cten in metastasis was tested by (a) intrasplenic injection of CRC cells stably transfected with a Cten expression vector into nude mice and (b) testing a series of primary human CRCs and their metastases by immunohistochemistry. Compared with controls, mice injected with cells expressing Cten developed larger tumours in the spleen (P<0.05) and liver (P<0.05). In the human cases, compared with primary tumours, the metastatic deposits had a significantly higher frequency of nuclear localisation of Cten (P=0.002). We conclude that Cten expression is of prognostic significance in CRC, and we delineate a Cten–ILK pathway controlling cell motility and possibly promoting metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ahmed MA, Al-Attar A, Kim J, Watson NF, Scholefield JH, Durrant LG et al. (2009). CD24 shows early upregulation and nuclear expression but is not a prognostic marker in colorectal cancer. J Clin Pathol 62: 1117–1122.
Albasri A, Aleskandarany M, Benhasouna A, Powe DG, Ellis IO, Ilyas M et al. (2011). CTEN (C-terminal tensin-like), a novel oncogene overexpressed in invasive breast carcinoma of poor prognosis. Breast Cancer Res Treat 126: 47–54.
Albasri A, Seth R, Jackson D, Benhasouna A, Crook S, Nateri AS et al. (2009). C-terminal tensin-like (CTEN) is an oncogene which alters cell motility possibly through repression of E-cadherin in colorectal cancer. J Pathol 218: 57–65.
Barbieri I, Pensa S, Pannellini T, Quaglino E, Maritano D, Demaria M et al. (2010). Constitutively active Stat3 enhances neu-mediated migration and metastasis in mammary tumors via upregulation of Cten. Cancer Res 70: 2558–2567.
Camp RL, Dolled-Filhart M, Rimm DL . (2004). X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res 10: 7252–7259.
Chen H, Ishii A, Wong WK, Chen LB, Lo SH . (2000). Molecular characterization of human tensin. Biochem J 351 (Part 2): 403–411.
Katz M, Amit I, Citri A, Shay T, Carvalho S, Lavi S et al. (2007). A reciprocal tensin-3-cten switch mediates EGF-driven mammary cell migration. Nat Cell Biol 9: 961–969.
Liao YC, Chen NT, Shih YP, Dong Y, Lo SH . (2009). Up-regulation of C-terminal tensin-like molecule promotes the tumorigenicity of colon cancer through beta-catenin. Cancer Res 69: 4563–4566.
Lo SH . (2004). Tensin. Int J Biochem Cell Biol 36: 31–34.
Lo SH, Janmey PA, Hartwig JH, Chen LB . (1994). Interactions of tensin with actin and identification of its three distinct actin-binding domains. J Cell Biol 125: 1067–1075.
Lo SH, Lo TB . (2002). Cten, a COOH-terminal tensin-like protein with prostate restricted expression, is down-regulated in prostate cancer. Cancer Res 62: 4217–4221.
McPhee TR, McDonald PC, Oloumi A, Dedhar S . (2008). Integrin-linked kinase regulates E-cadherin expression through PARP-1. Dev Dyn 237: 2737–2747.
Oloumi A, McPhee T, Dedhar S . (2004). Regulation of E-cadherin expression and beta-catenin/Tcf transcriptional activity by the integrin-linked kinase. Biochim Biophys Acta 1691: 1–15.
Sakashita K, Mimori K, Tanaka F, Kamohara Y, Inoue H, Sawada T et al. (2008). Prognostic relevance of tensin4 expression in human gastric cancer. Ann Surg Oncol 15: 2606–2613.
Sasaki H, Moriyama S, Mizuno K, Yukiue H, Konishi A, Yano M et al. (2003a). Cten mRNA expression was correlated with tumor progression in lung cancers. Lung Cancer 40: 151–155.
Sasaki H, Yukiue H, Kobayashi Y, Fukai I, Fujii Y . (2003b). Cten mRNA expression is correlated with tumor progression in thymoma. Tumour Biol 24: 271–274.
Somasiri A, Howarth A, Goswami D, Dedhar S, Roskelley CD . (2001). Overexpression of the integrin-linked kinase mesenchymally transforms mammary epithelial cells. J Cell Sci 114: 1125–1136.
Ullenhag GJ, Mukheriee A, Watson NFS, Ai-Attar AH, Scholefield JH, Durrant LG . (2007). Overexpression of FLIPL is an independent marker of poor prognosis in colorectal cancer patients. Clin Cancer Res 13: 5070–5075.
Acknowledgements
We thank Professor I Tomlinson for kindly donating cell lines, Miss Carol Dunn for helping with the preparation of the histology sections and Miss Anne Kane for helping with slide scanning. Image analysis was supported by Cost action IC0604.
Funding: The study was funded by the University of Nottingham and Cancer Research Nottingham. KBK is funded by the National Association for Colitis and Crohn’s Disease (NACC).
Author contribution: Study concept and design: MI, SW, LD, KBK. Acquisition of data: AA, SA, WF, MA, YL, DJ, RK. Analysis and interpretation of data: MI, SW, KBK. Drafting of the paper & critical revision of the manuscript: MI, SW, KBK, DNL, SL. Technical/material support: DNL, SL.
Declaration of originality
It is declared that this paper has not been published elsewhere and is not being considered by any other journal.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Albasri, A., Al-Ghamdi, S., Fadhil, W. et al. Cten signals through integrin-linked kinase (ILK) and may promote metastasis in colorectal cancer. Oncogene 30, 2997–3002 (2011). https://doi.org/10.1038/onc.2011.26
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.26
Keywords
This article is cited by
-
Nuclear-localized CTEN is a novel transcriptional regulator and promotes cancer cell migration through its downstream target CDC27
Journal of Physiology and Biochemistry (2023)
-
Identification of an Immune-Related BAT Signature for Predicting Adjuvant Chemotherapy Response and Overall Survival in Patients with Resected Ductal Adenocarcinoma of the Pancreas
Journal of Gastrointestinal Surgery (2022)
-
RETRACTED ARTICLE: Identification of core miRNA based on small RNA-seq and RNA-seq for colorectal cancer by bioinformatics
Tumor Biology (2015)
-
Epidermal growth factor receptor (EGFR) and Stat3 signal through Kras and have mutually opposite effects on Cten
BMC Genomics (2014)
-
Biomarkers for predicting future metastasis of human gastrointestinal tumors
Cellular and Molecular Life Sciences (2013)