Abstract
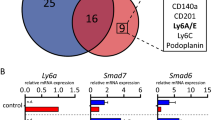
Basal-like breast cancer is an aggressive subtype of mammary carcinoma. Despite expressing basal markers, typical of mammary stem cells, this tumor has been proposed to originate from luminal progenitors, which are downstream of stem cells along the mammary epithelial hierarchy. This suggests that committed luminal progenitors may reacquire basal, stem-like characteristics, but the mechanisms that regulate this transition remain unclear. Using mouse models, we found that luminal progenitors express high levels of the Met receptor for hepatocyte growth factor (HGF), as compared with the other mammary epithelial sub-populations. Constitutive activation of Met led luminal progenitors to attain stem cell properties, including enhanced clonogenic activity in vitro and de novo ability to reconstitute mammary glands in repopulation assays in vivo. Moreover, in response to Met signaling, luminal progenitors gave rise to hyperplastic ductal morphogenesis and preferentially underwent basal lineage commitment at the expense of luminal cell-fate specification. Opposite and symmetric results were produced by systemic pharmacological inhibition of Met. Hence, Met signaling targets luminal progenitors for expansion, impairs their differentiation toward the mature luminal phenotype and enables their commitment toward the basal lineage. These results emphasize a critical role for Met in promoting deregulated proliferation and basal plasticity of normal luminal progenitors in the mammary gland, a complex of events that may be required for sustaining the functional and phenotypic properties of basal-like breast tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Shackleton M, Vaillant F, Simpson KJ, Stingl J, Smyth GK, Asselin-Labat ML et al. Generation of a functional mammary gland from a single stem cell. Nature 2006; 439: 84–88.
Sleeman KE, Kendrick H, Ashworth A, Isacke CM, Smalley MJ . CD24 staining of mouse mammary gland cells defines luminal epithelial, myoepithelial/basal and non-epithelial cells. Breast Cancer Res 2006; 8: R7.
Stingl J, Eirew P, Ricketson I, Shackleton M, Vaillant F, Choi D et al. Purification and unique properties of mammary epithelial stem cells. Nature 2006; 439: 993–997.
Taddei I, Deugnier MA, Faraldo MM, Petit V, Bouvard D, Medina D et al. Beta1 integrin deletion from the basal compartment of the mammary epithelium affects stem cells. Nat Cell Biol 2008; 10: 716–722.
Sleeman KE, Kendrick H, Robertson D, Isacke CM, Ashworth A, Smalley MJ . Dissociation of estrogen receptor expression and in vivo stem cell activity in the mammary gland. J Cell Biol 2007; 176: 19–26.
Asselin-Labat ML, Sutherland D, Barker H, Thomas R, Shackleton M, Forrest NC et al. Gata-3 is an essential regulator of mammary-gland morphogenesis and luminal-cell differentiation. Nat Cell Biol 2007; 9: 201–209.
Eirew P, Stingl J, Raouf A, Turashvili G, Aparicio S, Emerman JT et al. A method for quantifying normal human mammary epithelial stem cells with in vivo regenerative ability. Nat Med 2008; 14: 1384–1389.
Stingl J, Eaves CJ, Zandieh I, Emerman JT . Characterization of bipotent mammary epithelial progenitor cells in normal adult human breast tissue. Breast Cancer Res Treat 2001; 67: 93–109.
Villadsen R, Fridriksdottir AJ, Rønnov-Jessen L, Gudjonsson T, Rank F, LaBarge MA et al. Evidence for a stem cell hierarchy in the adult human breast. J Cell Biol 2007; 177: 87–101.
Van Keymeulen A, Rocha AS, Ousset M, Beck B, Bouvencourt G, Rock J et al. Distinct stem cells contribute to mammary gland development and maintenance. Nature 2011; 479: 189–193.
Visvader JE . Keeping abreast of the mammary epithelial hierarchy and breast tumorigenesis. Genes Dev 2009; 23: 2563–2277.
Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al. Molecular portraits of human breast tumours. Nature 2000; 406: 747–752.
Sorlie T, Tibshirani R, Parker J, Hastie T, Marron JS, Nobel A et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 2003; 100: 8418–8423.
Hu Z, Fan C, Oh DS, Marron JS, He X, Qaqish BF et al. The molecular portraits of breast tumors are conserved across microarray platforms. BMC Genomics 2006; 7: 96.
Stingl J, Caldas C . Molecular heterogeneity of breast carcinomas and the cancer stem cell hypothesis. Nat Rev Cancer 2007; 7: 791–799.
Foulkes WD, Smith IE, Reis-Filho JS . Triple-negative breast cancer. N Engl J Med 2010; 363: 1938–1948.
Chaffer CL, Weinberg RA . Cancer cell of origin: spotlight on luminal progenitors. Cell Stem Cell 2010; 7: 271–272.
Turner N, Tutt A, Ashworth A . Hallmarks of 'BRCAness' in sporadic cancers. Nat Rev Cancer 2004; 4: 814–819.
Lim E, Vaillant F, Wu D, Forrest NC, Pal B, Hart AH et al. Aberrant luminal progenitors as the candidate target population for basal tumor development in BRCA1 mutation carriers. Nat Med 2009; 15: 907–913.
Molyneux G, Geyer FC, Magnay FA, McCarthy A, Kendrick H, Natrajan R et al. BRCA1 basal-like breast cancers originate from luminal epithelial progenitors and not from basal stem cells. Cell Stem Cell 2010; 7: 403–417.
Mackay A, Weigelt B, Grigoriadis A, Kreike B, Natrajan R, A'Hern R et al. Microarray-based class discovery for molecular classification of breast cancer: analysis of interobserver agreement. J Natl Cancer Inst 2011; 103: 662–673.
Boccaccio C, Comoglio PM . Invasive growth: a MET-driven genetic programme for cancer and stem cells. Nat Rev Cancer 2006; 6: 637–645.
Graveel CR, DeGroot JD, Su Y, Koeman J, Dykema K, Leung S et al. Met induces diverse mammary carcinomas i n mice and is associated with human basal breast cancer. Proc Natl Acad Sci USA 2009; 106: 12909–12914.
Ponzo MG, Lesurf R, Petkiewicz S, O'Malley FP, Pinnaduwage D, Andrulis IL et al. Met induces mammary tumors with diverse histologies and is associated with poor outcome and human basal breast cancer. Proc Natl Acad Sci USA 2009; 106: 12903–12908.
Trusolino L, Bertotti A, Comoglio PM . MET signalling: principles and functions in development, organ regeneration and cancer. Nat Rev Mol Cell Biol 2010; 11: 834–848.
Garcia S, Dalès JP, Charafe-Jauffret E, Carpentier-Meunier S, Andrac-Meyer L, Jacquemier J et al. Poor prognosis in breast carcinomas correlates with increased expression of targetable CD146 and c-Met and with proteomic basal-like phenotype. Hum Pathol 2007; 38: 830–841.
Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF . Met metastasis, motility and more. Nat Rev Mol Cell Biol 2003; 4: 915–925.
Bertotti A, Burbridge MF, Gastaldi S, Galimi F, Torti D, Medico E et al. Only a subset of Met-activated pathways are required to sustain oncogene addiction. Sci Signal 2009; 2: ra80.
Eder JP, Vande Woude GF, Boerner SA, LoRusso PM . Novel therapeutic inhibitors of the c-Met signaling pathway in cancer. Clin Cancer Res 2009; 15: 2207–2214.
Comoglio PM, Giordano S, Trusolino L . Drug development of MET inhibitors: targeting oncogene addiction and expedience. Nat Rev Drug Discov 2008; 7: 504–516.
Cepero V, Sierra JR, Corso S, Ghiso E, Casorzo L, Perera T et al. MET and KRAS gene amplification mediates acquired resistance to MET tyrosine kinase inhibitors. Cancer Res 2010; 70: 7580–7590.
Galimi F, Torti D, Sassi F, Isella C, Corà D, Gastaldi S et al. Genetic and expression analysis of MET, MACC1, and HGF in metastatic colorectal cancer: response to met inhibition in patient xenografts and pathologic correlations. Clin Cancer Res 2011; 17: 3146–3156.
Torti D, Sassi F, Galimi F, Gastaldi S, Perera T, Comoglio PM et al. A preclinical algorithm of soluble surrogate biomarkers that correlate with therapeutic inhibition of the MET oncogene in gastric tumors. Int J Cancer 2012; 130: 1357–1366.
Bublil EM, Yarden Y . The EGF receptor family: spearheading a merger of signaling and therapeutics. Curr Opin Cell Biol 2007; 19: 124–134.
Niranjan B, Buluwela L, Yant J, Perusinghe N, Atherton A, Phippard D et al. HGF/SF: a potent cytokine for mammary growth, morphogenesis and development. Development 1995; 121: 2897–2908.
Yang Y, Spitzer E, Meyer D, Sachs M, Niemann C, Hartmann G et al. Sequential requirement of hepatocyte growth factor and neuregulin in the morphogenesis and differentiation of the mammary gland. J Cell Biol 1995; 131: 215–226.
Sebastian J, Richards RG, Walker MP, Wiesen JF, Werb Z, Derynck R et al. Activation and function of the epidermal growth factor receptor and erbB-2 during mammary gland morphogenesis. Cell Growth Differ 1998; 9: 777–785.
Wiesen JF, Young P, Werb Z, Cunha GR . Signaling through the stromal epidermal growth factor receptor is necessary for mammary ductal development. Development 1999; 126: 335–344.
Schroeder JA, Lee DC . Transgenic mice reveal roles for TGFalpha and EGF receptor in mammary gland development and neoplasia. J Mammary Gland Biol Neoplasia 1997; 2: 119–129.
Luetteke NC, Qiu TH, Fenton SE, Troyer KL, Riedel RF, Chang A et al. Targeted inactivation of the EGF and amphiregulin genes reveals distinct roles for EGF receptor ligands in mouse mammary gland development. Development 1999; 126: 2739–2750.
Sternlicht MD, Sunnarborg SW, Kouros-Mehr H, Yu Y, Lee DC, Werb Z . Mammary ductal morphogenesis requires paracrine activation of stromal EGFR via ADAM17-dependent shedding of epithelial amphiregulin. Development 2005; 132: 3923–3933.
Pao W, Chmielecki J . Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer. Nat Rev Cancer 2010; 10: 760–774.
Cataldo VD, Gibbons DL, Pérez-Soler R, Quintás-Cardama A . Treatment of non-small-cell lung cancer with erlotinib or gefitinib. N Engl J Med 2011; 364: 947–955.
Bozec A, Formento P, Lassalle S, Lippens C, Hofman P, Milano G . Dual inhibition of EGFR and VEGFR pathways in combination with irradiation: antitumour supra-additive effects on human head and neck cancer xenografts. Br J Cancer 2007; 97: 65–72.
Shimoyama T, Koizumi F, Fukumoto H, Kiura K, Tanimoto M, Saijo N et al. Effects of different combinations of gefitinib and irinotecan in lung cancer cell lines expressing wild or deletional EGFR. Lung Cancer 2006; 53: 13–21.
Kendrick H, Regan JL, Magnay FA, Grigoriadis A, Mitsopoulos C, Zvelebil M et al. Transcriptome analysis of mammary epithelial subpopulations identifies novel determinants of lineage commitment and cell fate. BMC Genomics 2008; 9: 591.
Birchmeier C, Gherardi E . Developmental roles of HGF/SF and its receptor, the c-Met tyrosine kinase. Trends Cell Biol 1998; 8: 404–410.
Rosário M, Birchmeier W . How to make tubes: signaling by the Met receptor tyrosine kinase. Trends Cell Biol 2003; 13: 328–335.
Gjorevski N, Nelson CM . Integrated morphodynamic signalling of the mammary gland. Nat Rev Mol Cell Biol 2012; 12: 581–593.
Jackson-Fisher AJ, Bellinger G, Ramabhadran R, Morris JK, Lee KF, Stern DF . ErbB2 is required for ductal morphogenesis of the mammary gland. Proc Natl Acad Sci USA 2004; 101: 17138–17143.
Andrechek ER, White D, Muller WJ . Targeted disruption of ErbB2/Neu in the mammary epithelium results in impaired ductal outgrowth. Oncogene 2005; 24: 932–937.
Jones FE, Jerry DJ, Guarino BC, Andrews GC, Stern DF . Heregulin induces in vivo proliferation and differentiation of mammary epithelium into secretory lobuloalveoli. Cell Growth Differ 1996; 7: 1031–1038.
Jones FE, Welte T, Fu XY, Stern DF . ErbB4 signaling in the mammary gland is required for lobuloalveolar development and Stat5 activation during lactation. J Cell Biol 1999; 147: 77–88.
Brisken C, Heineman A, Chavarria T, Elenbaas B, Tan J, Dey SK et al. Essential function of Wnt-4 in mammary gland development downstream of progesterone signaling. Genes Dev 2000; 14: 650–654.
Simian M, Hirai Y, Navre M, Werb Z, Lochter A, Bissell MJ . The interplay of matrix metalloproteinases, morphogens and growth factors is necessary for branching of mammary epithelial cells. Development 2001; 128: 3117–3131.
Finkbeiner MR, Astanehe A, To K, Fotovati A, Davies AH, Zhao Y et al. Profiling YB-1 target genes uncovers a new mechanism for MET receptor regulation in normal and malignant human mammary cells. Oncogene 2009; 28: 1421–1431.
Gastaldi S, Comoglio PM, Trusolino L . The Met oncogene and basal-like breast cancer: another culprit to watch out for? Breast Cancer Res 2010; 12: 208.
Ponzo MG, Park M . The Met receptor tyrosine kinase and basal breast cancer. Cell Cycle 2010; 9: 1043–1050.
Regan JL, Kendrick H, Magnay FA, Vafaizadeh V, Groner B, Smalley MJ . c-Kit is required for growth and survival of the cells of origin of Brca1-mutation-associated breast cancer. Oncogene 2012; 31: 869–883.
Michieli P, Mazzone M, Basilico C, Cavassa S, Sottile A, Naldini L et al. Targeting the tumor and its microenvironment by a dual-function decoy Met receptor. Cancer Cell 2004; 6: 61–73.
Acknowledgements
We thank Erica Lantelme for help with FACS sorting; Noemi Cavalera, Claudio Isella, Howard Kendrick, Simonetta Leto, Barbara Lupo, Paolo Luraghi, Fiona-Ann Magnay, Barbara Martinoglio, Enzo Medico, Roberta Porporato and Eugenia Zanella for help with experiments, discussion and sharing reagents; Fabrizio Maina, Laura Tarditi and Tuti Werdiningsih for animal husbandry; Raffaella Albano, Stefania Giove and Laura Palmas for technical assistance; Antonella Cignetto, Daniela Gramaglia and Francesca Natale for secretarial assistance. This work was supported by AIRC, Associazione Italiana per la Ricerca sul Cancro (IG project 10116 to LT); MIUR-FIRB, Fondo per gli Investimenti della Ricerca di Base – Futuro in Ricerca (AB); FPRC, Fondazione Piemontese per la Ricerca sul Cancro (LT); Breakthrough Breast Cancer and United Kingdom National Health Service funding to the National Institute for Health Research Biomedical Research Centre (MJS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
T Perera is an employee of Janssen pharmaceutical companies of Johnson & Johnson. PM Comoglio receives research grants from Janssen pharmaceutical companies. The other authors declare no potential conflicts of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Gastaldi, S., Sassi, F., Accornero, P. et al. Met signaling regulates growth, repopulating potential and basal cell-fate commitment of mammary luminal progenitors: implications for basal-like breast cancer. Oncogene 32, 1428–1440 (2013). https://doi.org/10.1038/onc.2012.154
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.154
Keywords
This article is cited by
-
Activation of CD44 signaling in leader cells induced by tumor-associated macrophages drives collective detachment in luminal breast carcinomas
Cell Death & Disease (2022)
-
Lineage plasticity in cancer: a shared pathway of therapeutic resistance
Nature Reviews Clinical Oncology (2020)
-
p53 controls the plasticity of mammary luminal progenitor cells downstream of Met signaling
Breast Cancer Research (2019)
-
Hormones induce the formation of luminal-derived basal cells in the mammary gland
Cell Research (2019)
-
Known and novel roles of the MET oncogene in cancer: a coherent approach to targeted therapy
Nature Reviews Cancer (2018)