Abstract
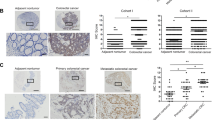
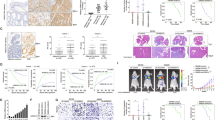
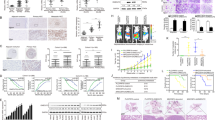
Metastasis is the major cause of death in colorectal cancer (CRC). Although multiple genes have been identified to be responsible for the development of CRC, the molecular changes that enable CRC cells to undergo early local invasion and to form distant metastatic colonies still remain largely unknown. Herein, we investigated the role of Forkhead box protein C2 (FOXC2) and explored the underlying mechanisms in invasion and metastasis of CRC. We show that both high FOXC2 expression and nuclear localization of FOXC2 are significantly correlated with advanced TNM (T=primary tumor; N=regional lymph nodes; M=distant metastasis) stages. FOXC2 enhanced the invasive abilities of CRC cells in vitro and promoted local invasion and distant metastasis in an orthotopic mouse metastatic model of CRC. Microarray analysis revealed that overexpression of FOXC2 increased the proto-oncogene MET tyrosine kinase expression and activated the hepatocyte growth factor (HGF)-MET signaling pathway. Furthermore, luciferase reporter assays and chromatin immunoprecipitation assays revealed that FOXC2 directly associated with MET promoter to increase the transcriptional activity of MET. Inhibition of MET attenuates the invasive phenotype and metastatic potential of FOXC2-overexpressing CRC cells, indicating that MET is a major mediator of FOXC2-promoted metastasis. In addition, FOXC2 expression was positively correlated with MET expression in CRC tissue samples. Our findings suggest that FOXC2 has a crucial role in CRC metastasis by regulating HGF-MET signaling via inducing MET expression, highlighting FOXC2 as a potential therapeutic target for preventing or reducing metastasis in CRC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
09 April 2022
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1038/s41388-022-02287-w
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D . Global cancer statistics. CA Cancer J Clin 2011; 61: 69–90.
Van Cutsem E, Grothey A . Regorafenib for metastatic colorectal cancer—authors' reply. Lancet 2013; 381: 1538–1539.
Duffy MJ, van Dalen A, Haglund C, Hansson L, Holinski-Feder E, Klapdor R et al. Tumour markers in colorectal cancer: European Group on Tumour Markers (EGTM) guidelines for clinical use. Eur J Cancer 2007; 43: 1348–1360.
Markowitz SD, Bertagnolli MM . Molecular origins of cancer: molecular basis of colorectal cancer. New Engl J Med 2009; 361: 2449–2460.
Hayashi H, Sano H, Seo S, Kume T . The Foxc2 transcription factor regulates angiogenesis via induction of integrin beta3 expression. J Biol Chem 2008; 283: 23791–23800.
Kume T . The role of FoxC2 transcription factor in tumor angiogenesis. J Oncol 2012; 2012: 204593.
Sano H, Leboeuf JP, Novitskiy SV, Seo S, Zaja-Milatovic S, Dikov MM et al. The Foxc2 transcription factor regulates tumor angiogenesis. Biochem Biophys Res Commun 2010; 392: 201–206.
Mani SA, Yang J, Brooks M, Schwaninger G, Zhou A, Miura N et al. Mesenchyme Forkhead 1 (FOXC2) plays a key role in metastasis and is associated with aggressive basal-like breast cancers. Proc Natl Acad Sci USA 2007; 104: 10069–10074.
Hollier BG, Tinnirello AA, Werden SJ, Evans KW, Taube JH, Sarkar TR et al. FOXC2 expression links epithelial-mesenchymal transition and stem cell properties in breast cancer. Cancer Res 2013; 73: 1981–1992.
Nishida N, Mimori K, Yokobori T, Sudo T, Tanaka F, Shibata K et al. FOXC2 is a novel prognostic factor in human esophageal squamous cell carcinoma. Ann Surg Oncol 2011; 18: 535–542.
Zhu JL, Song YX, Wang ZN, Gao P, Wang MX, Dong YL et al. The clinical significance of mesenchyme forkhead 1 (FoxC2) in gastric carcinoma. Histopathology 2013; 62: 1038–1048.
Jiang W, Pang XG, Wang Q, Shen YX, Chen XK, Xi JJ . Prognostic role of Twist, Slug, and Foxc2 expression in stage I non-small-cell lung cancer after curative resection. Clin Lung Cancer 2012; 13: 280–287.
Yu YH, Chen HA, Chen PS, Cheng YJ, Hsu WH, Chang YW et al. MiR-520h-mediated FOXC2 regulation is critical for inhibition of lung cancer progression by resveratrol. Oncogene 2013; 32: 431–443.
Mortazavi F, An J, Dubinett S, Rettig M . p120-catenin is transcriptionally downregulated by FOXC2 in non-small cell lung cancer cells. Mol Cancer Res 2010; 8: 762–774.
Nguyen DX, Bos PD, Massague J . Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer 2009; 9: 274–284.
Valastyan S, Weinberg RA . Tumor metastasis: molecular insights and evolving paradigms. Cell 2011; 147: 275–292.
Xue Y, Cao R, Nilsson D, Chen S, Westergren R, Hedlund EM et al. FOXC2 controls Ang-2 expression and modulates angiogenesis, vascular patterning, remodeling, and functions in adipose tissue. Proc Natl Acad Sci USA 2008; 105: 10167–10172.
Di Renzo MF, Olivero M, Giacomini A, Porte H, Chastre E, Mirossay L et al. Overexpression and amplification of the met/HGF receptor gene during the progression of colorectal cancer. Clin Cancer Res 1995; 1: 147–154.
Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF . Met, metastasis, motility and more. Nat Rev Mol Cell Biol 2003; 4: 915–925.
Sattler M, Salgia R . c-Met and hepatocyte growth factor: potential as novel targets in cancer therapy. Curr Oncol Rep 2007; 9: 102–108.
Mazzone M, Comoglio PM . The Met pathway: master switch and drug target in cancer progression. FASEB J 2006; 20: 1611–1621.
Bottaro DP, Rubin JS, Faletto DL, Chan AM, Kmiecik TE, Vande Woude GF et al. Identification of the hepatocyte growth factor receptor as the c-met proto-oncogene product. Science 1991; 251: 802–804.
Chmielowiec J, Borowiak M, Morkel M, Stradal T, Munz B, Werner S et al. c-Met is essential for wound healing in the skin. J Cell Biol 2007; 177: 151–162.
Danilkovitch-Miagkova A, Zbar B . Dysregulation of Met receptor tyrosine kinase activity in invasive tumors. J Clin Invest 2002; 109: 863–867.
De Oliveira AT, Matos D, Logullo AF, DA Silva SR, Neto RA, Filho AL et al. MET Is highly expressed in advanced stages of colorectal cancer and indicates worse prognosis and mortality. Anticancer Res 2009; 29: 4807–4811.
Takeuchi H, Bilchik A, Saha S, Turner R, Wiese D, Tanaka M et al. c-MET expression level in primary colon cancer: a predictor of tumor invasion and lymph node metastases. Clin Cancer Res 2003; 9: 1480–1488.
Kammula US, Kuntz EJ, Francone TD, Zeng Z, Shia J, Landmann RG et al. Molecular co-expression of the c-Met oncogene and hepatocyte growth factor in primary colon cancer predicts tumor stage and clinical outcome. Cancer Lett 2007; 248: 219–228.
Liu C, Park M, Tsao MS . Overexpression of c-met proto-oncogene but not epidermal growth factor receptor or c-erbB-2 in primary human colorectal carcinomas. Oncogene 1992; 7: 181–185.
Zeng ZS, Weiser MR, Kuntz E, Chen CT, Khan SA, Forslund A et al. c-Met gene amplification is associated with advanced stage colorectal cancer and liver metastases. Cancer Lett 2008; 265: 258–269.
Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, Giordano S, Comoglio PM . Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer cell 2003; 3: 347–361.
Boon EM, van der Neut R, van de Wetering M, Clevers H, Pals ST . Wnt signaling regulates expression of the receptor tyrosine kinase met in colorectal cancer. Cancer Res 2002; 62: 5126–5128.
Rasola A, Fassetta M, De Bacco F, D'Alessandro L, Gramaglia D, Di Renzo MF et al. A positive feedback loop between hepatocyte growth factor receptor and beta-catenin sustains colorectal cancer cell invasive growth. Oncogene 2007; 26: 1078–1087.
Stein U, Clinical Schlag PM . biological, and molecular aspects of metastasis in colorectal cancer. Recent Results Cancer Res 2007; 176: 61–80.
Stein U, Walther W, Arlt F, Schwabe H, Smith J, Fichtner I et al. MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis. Nat Med 2009; 15: 59–67.
Liu Y . The human hepatocyte growth factor receptor gene: complete structural organization and promoter characterization. Gene 1998; 215: 159–169.
Liang H, O'Reilly S, Liu Y, Abounader R, Laterra J, Maher VM et al. Sp1 regulates expression of MET, and ribozyme-induced down-regulation of MET in fibrosarcoma-derived human cells reduces or eliminates their tumorigenicity. Int J Oncol 2004; 24: 1057–1067.
Liu X, Yao W, Newton RC, Scherle PA . Targeting the c-MET signaling pathway for cancer therapy. Expert Opin Investig Drugs 2008; 17: 997–1011.
Zhang SH, Wang CJ, Shi L, Li XH, Zhou J, Song LB et al. High Expression of FLOT1 is associated with progression and poor prognosis in hepatocellular carcinoma. PLoS ONE 2013; 8: e64709.
Park SJ, Gadi J, Cho KW, Kim KJ, Kim SH, Jung HS et al. The forkhead transcription factor Foxc2 promotes osteoblastogenesis via up-regulation of integrin beta1 expression. Bone 2011; 49: 428–438.
Song LB, Zeng MS, Liao WT, Zhang L, Mo HY, Liu WL et al. Bmi-1 is a novel molecular marker of nasopharyngeal carcinoma progression and immortalizes primary human nasopharyngeal epithelial cells. Cancer Res 2006; 66: 6225–6232.
Liao WT, Jiang D, Yuan J, Cui YM, Shi XW, Chen CM et al. HOXB7 as a prognostic factor and mediator of colorectal cancer progression. Clin Cancer Res 2011; 17: 3569–3578.
Shibue T, Brooks MW, Inan MF, Reinhardt F, Weinberg RA . The outgrowth of micrometastases is enabled by the formation of filopodium-like protrusions. Cancer Discov 2012; 2: 706–721.
Tseng W, Leong X, Engleman E . Orthotopic mouse model of colorectal cancer. J Vis Exp 2007; 10: 484.
Acknowledgements
This work was supported by the National Basic Research Program of China (973 program, nos. 2015CB554002 and 2010CB529403), the National Natural Science Foundation of China (NSFC)-Guangdong Joint Fund (U1201226), the Major Projects of the National Natural Science Foundation of China (no. 81090422), the National Natural Science Foundation of China (nos. 30901791, 81172055, 81472313, 81472710 and 81071735), Guangdong Provincial Natural Science Foundation of China (no. S2012010009643), Zhu Jiang Science & Technology New Star Foundation in Guangzhou city (2012J2200052 and 2012J2200044), the Science and Technology Innovation Foundation of Guangdong Higher Education (CXZD1016), the Key Program of the National Natural Science Foundation of Guangdong, China (2010B031500012), and Guangzhou Science & Technology Plan Project (201300000056).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
This article has been retracted. Please see the retraction notice for more detail:https://doi.org/10.1038/s41388-022-02287-w
Supplementary information
About this article
Cite this article
Cui, YM., Jiao, HL., Ye, YP. et al. RETRACTED ARTICLE: FOXC2 promotes colorectal cancer metastasis by directly targeting MET. Oncogene 34, 4379–4390 (2015). https://doi.org/10.1038/onc.2014.368
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.368
This article is cited by
-
MicroRNAs as the critical regulators of Forkhead box protein family during gynecological and breast tumor progression and metastasis
European Journal of Medical Research (2023)
-
HSF4 promotes tumor progression of colorectal cancer by transactivating c-MET
Molecular and Cellular Biochemistry (2023)
-
Colorectal liver metastasis: molecular mechanism and interventional therapy
Signal Transduction and Targeted Therapy (2022)
-
Oncogenic functions of the FOXC2 transcription factor: a hallmarks of cancer perspective
Cancer and Metastasis Reviews (2022)
-
MicroRNAs and angiogenesis: a new era for the management of colorectal cancer
Cancer Cell International (2021)