Abstract
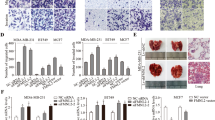
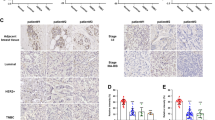
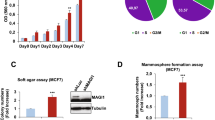
Increased CLP36 expression has been found to be closely associated with breast cancer progression. However, whether and how it contributes to malignant behavior of breast cancer cells were not known. We show here that CLP36 is critical for promoting breast cancer cell migration and invasion in vitro and metastasis in vivo, whereas it is dispensable for breast cell proliferation and anchorage-independent growth in vitro and tumor growth in vivo. CLP36 interacted with both α-actinin-1 and -4 in breast cancer cells. Depletion of either α-actinin-1 or -4 inhibited breast cancer cell migration. Furthermore, mutations inhibiting the α-actinin-binding activity abolished the ability of CLP36 to promote breast cancer cell migration. Finally, depletion of CLP36 or disruption of the CLP36–α-actinin complex in breast cancer cells substantially inhibited Cdc42 activation, cell polarization and migration. Our results identify CLP36 as an important regulator of breast cancer cell migration and metastasis, and shed light on how increased CLP36 expression contributes to the progression of breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Cardiff RD, Anver MR, Gusterson BA, Hennighausen L, Jensen RA, Merino MJ et al. The mammary pathology of genetically engineered mice: the consensus report and recommendations from the Annapolis meeting. Oncogene 2000; 19: 968–988.
Pitteri SJ, Kelly-Spratt KS, Gurley KE, Kennedy J, Buson TB, Chin A et al. Tumor microenvironment-derived proteins dominate the plasma proteome response during breast cancer induction and progression. Cancer Res 2011; 71: 5090–5100.
Kotaka M, Ngai SM, Garcia-Barcelo M, Tsui SK, Fung KP, Lee CY et al. Characterization of the human 36-kDa carboxyl terminal LIM domain protein (hCLIM1). J Cell Biochem 1999; 72: 279–285.
Bauer K, Kratzer M, Otte M, de Quintana KL, Hagmann J, Arnold GJ et al. Human CLP36, a PDZ-domain and LIM-domain protein, binds to alpha-actinin-1 and associates with actin filaments and stress fibers in activated platelets and endothelial cells. Blood 2000; 96: 4236–4245.
Vallenius T, Luukko K, Makela TP . CLP-36 PDZ-LIM protein associates with nonmuscle alpha-actinin-1 and alpha-actinin-4. J Biol Chem 2000; 275: 11100–11105.
Kotaka M, Kostin S, Ngai S, Chan K, Lau Y, Lee SM et al. Interaction of hCLIM1, an enigma family protein, with alpha-actinin 2. J Cell Biochem 2000; 78: 558–565.
Vallenius T, Scharm B, Vesikansa A, Luukko K, Schafer R, Makela TP . The PDZ-LIM protein RIL modulates actin stress fiber turnover and enhances the association of alpha-actinin with F-actin. Exp Cell Res 2004; 293: 117–128.
Tamura N, Ohno K, Katayama T, Kanayama N, Sato K . The PDZ-LIM protein CLP36 is required for actin stress fiber formation and focal adhesion assembly in BeWo cells. Biochem Biophys Res Commun 2007; 364: 589–594.
Liu Z, Blattner SM, Tu Y, Tisherman R, Wang JH, Rastaldi MP et al. Alpha-actinin-4 and CLP36 protein deficiencies contribute to podocyte defects in multiple human glomerulopathies. J Biol Chem 2011; 286: 30795–30805.
Miyazaki K, Ohno K, Tamura N, Sasaki T, Sato K . CLP36 and RIL recruit alpha-actinin-1 to stress fibers and differentially regulate stress fiber dynamics in F2408 fibroblasts. Exp Cell Res 2012; 318: 1716–1725.
Honda K, Yamada T, Endo R, Ino Y, Gotoh M, Tsuda H et al. Actinin-4, a novel actin-bundling protein associated with cell motility and cancer invasion. J Cell Biol 1998; 140: 1383–1393.
Quick Q, Skalli O . Alpha-actinin 1 and alpha-actinin 4: contrasting roles in the survival, motility, and RhoA signaling of astrocytoma cells. Exp Cell Res 2010; 316: 1137–1147.
Honda K, Yamada T, Hayashida Y, Idogawa M, Sato S, Hasegawa F et al. Actinin-4 increases cell motility and promotes lymph node metastasis of colorectal cancer. Gastroenterology 2005; 128: 51–62.
Kikuchi S, Honda K, Tsuda H, Hiraoka N, Imoto I, Kosuge T et al. Expression and gene amplification of actinin-4 in invasive ductal carcinoma of the pancreas. Clin Cancer Res 2008; 14: 5348–5356.
Yamamoto S, Tsuda H, Honda K, Onozato K, Takano M, Tamai S et al. Actinin-4 gene amplification in ovarian cancer: a candidate oncogene associated with poor patient prognosis and tumor chemoresistance. Mod Pathol 2009; 22: 499–507.
Shao H, Wang JH, Pollak MR, Wells A . alpha-actinin-4 is essential for maintaining the spreading, motility and contractility of fibroblasts. PLoS One 2010; 5: e13921.
Te Velthuis AJ, Isogai T, Gerrits L, Bagowski CP . Insights into the molecular evolution of the PDZ/LIM family and identification of a novel conserved protein motif. PLoS One 2007; 2: e189.
Klaavuniemi T, Kelloniemi A, Ylanne J . The ZASP-like motif in actinin-associated LIM protein is required for interaction with the alpha-actinin rod and for targeting to the muscle Z-line. J Biol Chem 2004; 279: 26402–26410.
Etienne-Manneville S . Cdc42—the centre of polarity. J Cell Sci 2004; 117: 1291–1300.
Fritz G, Brachetti C, Bahlmann F, Schmidt M, Kaina B . Rho GTPases in human breast tumours: expression and mutation analyses and correlation with clinical parameters. Br J Cancer 2002; 87: 635–644.
Fritz G, Just I, Kaina B . Rho GTPases are over-expressed in human tumors. Int J Cancer 1999; 81: 682–687.
Johnson E, Seachrist DD, DeLeon-Rodriguez CM, Lozada KL, Miedler J, Abdul-Karim FW et al. HER2/ErbB2-induced breast cancer cell migration and invasion require p120 catenin activation of Rac1 and Cdc42. J Biol Chem 2010; 285: 29491–29501.
Yamaguchi H, Lorenz M, Kempiak S, Sarmiento C, Coniglio S, Symons M et al. Molecular mechanisms of invadopodium formation: the role of the N-WASP-Arp2/3 complex pathway and cofilin. J Cell Biol 2005; 168: 441–452.
Pichot CS, Arvanitis C, Hartig SM, Jensen SA, Bechill J, Marzouk S et al. Cdc42-interacting protein 4 promotes breast cancer cell invasion and formation of invadopodia through activation of N-WASp. Cancer Res 2010; 70: 8347–8356.
Bouzahzah B, Albanese C, Ahmed F, Pixley F, Lisanti MP, Segall JD et al. Rho family GTPases regulate mammary epithelium cell growth and metastasis through distinguishable pathways. Mol Med 2001; 7: 816–830.
Tu Y, Huang Y, Zhang Y, Hua Y, Wu C . A new focal adhesion protein that interacts with integrin-linked kinase and regulates cell adhesion and spreading. J Cell Biol 2001; 153: 585–598.
Tu Y, Wu S, Shi X, Chen K, Wu C . Migfilin and mig-2 link focal adhesions to filamin and the actin cytoskeleton and function in cell shape modulation. Cell 2003; 113: 37–47.
Shi X, Wu C . A suppressive role of mitogen inducible gene-2 in mesenchymal cancer cell invasion. Mol Cancer Res 2008; 6: 715–724.
Zhang Y, Tu Y, Gkretsi V, Wu C . Migfilin interacts with vasodilator-stimulated phosphoprotein (VASP) and regulates VASP localization to cell-matrix adhesions and migration. J Biol Chem 2006; 281: 12397–12407.
Uehara N, Matsuoka Y, Tsubura A . Mesothelin promotes anchorage-independent growth and prevents anoikis via extracellular signal-regulated kinase signaling pathway in human breast cancer cells. Mol Cancer Res 2008; 6: 186–193.
Shi X, Ma YQ, Tu Y, Chen K, Wu S, Fukuda K et al. The MIG-2/integrin interaction strengthens cell-matrix adhesion and modulates cell motility. J Biol Chem 2007; 282: 20455–20466.
Nobes CD, Hall A . Rho GTPases control polarity, protrusion, and adhesion during cell movement. J Cell Biol 1999; 144: 1235–1244.
Acknowledgements
This work was supported by NIH Grant GM65188 to CW.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, Z., Zhan, Y., Tu, Y. et al. PDZ and LIM domain protein 1(PDLIM1)/CLP36 promotes breast cancer cell migration, invasion and metastasis through interaction with α-actinin. Oncogene 34, 1300–1311 (2015). https://doi.org/10.1038/onc.2014.64
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.64