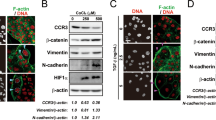
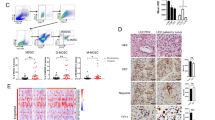
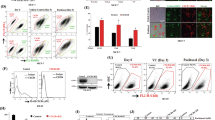
Abstract
In the tumor microenvironment, chemokine system has a critical role in tumorigenesis and metastasis. The acquisition of stem-like properties by cancer cells is involved in metastasis and drug resistance, which are pivotal problems that result in poor outcomes in patients with lung cancer. Patients with advanced lung cancer present high plasma levels of transforming growth factor-β1 (TGFβ1), which correlate with poor prognostic features. Therefore, TGFβ1 may be important in the tumor microenvironment, where chemokines are widely expressed. However, the role of chemokines in TGFβ1-induced tumor progression still remains unclear. In our study, TGFβ1 upregulated CXC chemokine receptor expression, migration, invasion, epithelial–mesenchymal transition (EMT) and cancer stem cell (CSC) formation in lung adenocarcinoma. We found that CXCR7 was the most upregulated chemokine receptor induced by TGFβ1. CXCR7 knockdown resulted in reduction of migration, invasion and EMT induced by TGFβ1, whereas CXCR4 knockdown did not reverse TGFβ1-promoted EMT. CXCR7 silencing significantly decreased cancer sphere-forming capacity, stem-like properties, chemoresistance and TGFβ1-induced CSC tumor initiation in vivo. In clinical samples, high TGFβ1 and CXCR7 expression was significantly associated with the late stages of lung adenocarcinoma. Moreover, TGFβ1 and CXCR7 coexpression was positively correlated with the CSC marker, CD44, which is associated with lymph node metastasis. Besides, patients with high expression of both CXCR7 and TGFβ1 presented a significantly worse survival rate. These results suggest that the TGFβ1-CXCR7 axis may be a prognostic marker and may provide novel targets for combinational therapies to be used in the treatment of advanced lung cancer in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D . Global cancer statistics. CA Cancer J Clin 2011; 61: 69–90.
Bareschino MA, Schettino C, Rossi A, Maione P, Sacco PC, Zeppa R et al. Treatment of advanced non small cell lung cancer. J Thorac Dis 2011; 3: 122–133.
Hesegawa Y, Takanashi S, Kanehira Y, Tsushima T, Imai T, Okumura K . Transforming growth factor-beta1 level correlates with angiogenesis, tumor progression and prognosis in patients with non-small cell lung carcinoma. Cancer 2001; 91: 964–971.
Hou YL, Chen H, Dong ZH, Xue CJ, Wu YF, Luo HX et al. Clinical significance of serum transforming growth factor-beta1 in lung cancer. Cancer Epidemiol 2013; 37: 750–753.
Massague J . TGFbeta in cancer. Cell 2008; 134: 215–230.
Principe DR, Doll JA, Bauer J, Jung B, Munshi HG, Bartholin L et al. TGF-beta: duality of function between tumor prevention and carcinogenesis. J Natl Cancer Inst 2014; 106: djt369.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. The epithelial–mesenchymal transition generates cells with properties of stem cells. Cell 2008; 133: 704–715.
Singh A, Settleman J . EMT cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene 2010; 29: 4741–4751.
Lobo NA, Shimono Y, Qian D, Clarke MF . The biology of cancer stem cells. Annu Rev Cell Dev Biol 2007; 23: 675–699.
Pirozzi G, Tirino V, Camerlingo R, Franco R, La Rocca A, Liguori E et al. Epithelial to mesenchymal transition by TGFbeta-1 induction increases stemness characteristics in primary non small cell lung cancer cell line. PLoS One 2011; 6: e21548.
Sakaki-Yumoto M, Katsuno Y, Derynck R . TGF-beta family signaling in stem cells. Biochim Biophys Acta 2013; 1830: 2280–2296.
Kuperwasser C, Chavarria T, Wu M, Magrane G, Gray JW, Carey L et al. Reconstruction of functionally normal and malignant human breast tissues in mice. Proc Natl Acad Sci USA 2004; 101: 4966–4971.
Bhowmick NA, Chytil A, Plieth D, Gorska AE, Dumont N, Shappell S et al. TGF-beta signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science 2004; 303: 848–851.
Bierie B, Moses HL . Tumour microenvironment: TGFbeta: the molecular Jekyll and Hyde of cancer. Nat Rev Cancer 2006; 6: 506–520.
Lazennec G, Richmond A . Chemokines and chemokine receptors: new insights into cancer-related inflammation. Trends Mol Med 2010; 16: 133–144.
Balkwill FR . The chemokine system and cancer. J Pathol 2012; 226: 148–157.
Vandercappellen J, Van Damme J, Struyf S . The role of CXC chemokines and their receptors in cancer. Cancer Lett 2008; 267: 226–244.
Bertran E, Caja L, Navarro E, Sancho P, Mainez J, Murillo MM et al. Role of CXCR4/SDF-1 alpha in the migratory phenotype of hepatoma cells that have undergone epithelial–mesenchymal transition in response to the transforming growth factor-beta. Cell Signal 2009; 21: 1595–1606.
Bartolomé R, Gálvez B, Longo N, Baleux F, Van Muijen G, Sánchez-Mateos P et al. Stromal cell-derived factor-1alpha promotes melanoma cell invasion across basement membranes involving stimulation of membrane-type 1 matrix metalloproteinase and Rho GTPase activities. Cancer Res 2004; 64: 2534–2543.
Zhao XP, Huang YY, Huang Y, Lei P, Peng JL, Wu S et al. Transforming growth factor-beta1 upregulates the expression of CXC chemokine receptor 4 (CXCR4) in human breast cancer MCF-7 cells. Acta Pharmacol Sin 2010; 31: 347–354.
Zoller M . CD44: can a cancer-initiating cell profit from an abundantly expressed molecule? Nat Rev Cancer 2011; 11: 254–267.
Meulmeester E, Ten Dijke P . The dynamic roles of TGF-beta in cancer. J Pathol 2011; 223: 205–218.
Siegel PM, Massague J . Cytostatic and apoptotic actions of TGF-beta in homeostasis and cancer. Nat Rev Cancer 2003; 3: 807–821.
Jiang X, Liu R, You J, Zhou Q, Zhang H . Defective expression of TGFBR3 gene and its molecular mechanisms in non-small cell lung cancer cell lines. Zhongguo Fei Ai Za Zhi 2010; 13: 451–457.
Xu CC, Wu LM, Sun W, Zhang N, Chen WS, Fu XN . Effects of TGF-β signaling blockade on human A549 lung adenocarcinoma cell lines. Mol Med Rep 2011; 4: 1007–1015.
Nagasawa T, Hirota S, Tachibana K, Takakura N, Nishikawa S, Kitamura Y et al. Defects of B-cell lymphopoiesis and bone-marrow myelopoiesis in mice lacking the CXC chemokine PBSF/SDF-1. Nature 1996; 382: 635–638.
Sun X, Cheng G, Hao M, Zheng J, Zhou X, Zhang J et al. CXCL12 / CXCR4 / CXCR7 chemokine axis and cancer progression. Cancer Metast Rev 2010; 29: 709–722.
Guo Y, Hangoc G, Bian H, Pelus LM, Broxmeyer HE . SDF-1/CXCL12 enhances survival and chemotaxis of murine embryonic stem cells and production of primitive and definitive hematopoietic progenitor cells. Stem Cells 2005; 23: 1324–1332.
Kucia M, Reca R, Miekus K, Wanzeck J, Wojakowski W, Janowska-Wieczorek A et al. Trafficking of normal stem cells and metastasis of cancer stem cells involve similar mechanisms: pivotal role of the SDF-1-CXCR4 axis. Stem Cells 2005; 23: 879–894.
Muller A, Homey B, Soto H, Ge N, Catron D, Buchanan M et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001; 410: 50–56.
Hartmann T, Burger J, Glodek A, Fujii N, Burger M . CXCR4 chemokine receptor and integrin signaling co-operate in mediating adhesion and chemoresistance in small cell lung cancer (SCLC) cells. Oncogene 2005; 24: 4462–4471.
Furusato B, Mohamed A, Uhlen M, Rhim JS . CXCR4 and cancer. Pathol Int 2010; 60: 497–505.
Burns JM, Summers BC, Wang Y, Melikian A, Berahovich R, Miao Z et al. A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J Exp Med 2006; 203: 2201–2213.
Balabanian K, Lagane B, Infantino S, Chow KY, Harriague J, Moepps B et al. The chemokine SDF-1/CXCL12 binds to and signals through the orphan receptor RDC1 in T lymphocytes. J Biol Chem 2005; 280: 35760–35766.
Maksym RB, Tarnowski M, Grymula K, Tarnowska J, Wysoczynski M, Liu R et al. The role of stromal-derived factor-1-CXCR7 axis in development and cancer. Eur J Pharmacol 2009; 625: 31–40.
Sanchez-Martin L, Sanchez-Mateos P, Cabanas C . CXCR7 impact on CXCL12 biology and disease. Trends Mol Med 2013; 19: 12–22.
Miao Z, Luker KE, Summers BC, Berahovich R, Bhojani MS, Rehemtulla A et al. CXCR7 (RDC1) promotes breast and lung tumor growth in vivo and is expressed on tumor-associated vasculature. Proc Natl Acad Sci USA 2007; 104: 15735–15740.
Iwakiri S, Mino N, Takahashi T, Sonobe M, Nagai S, Okubo K et al. Higher expression of chemokine receptor CXCR7 is linked to early and metastatic recurrence in pathological stage I nonsmall cell lung cancer. Cancer 2009; 115: 2580–2593.
Su L, Zhang J, Xu H, Wang Y, Chu Y, Liu R et al. Differential expression of CXCR4 is associated with the metastatic potential of human non-small cell lung cancer cells. Clin Cancer Res 2005; 11: 8273–8280.
Suzuki M, Mohamed S, Nakajima T, Kubo R, Tian L, Fujiwara T et al. Aberrant methylation of CXCL12 in non-small cell lung cancer is associated with an unfavorable prognosis. Int J Oncol 2008; 33: 113–119.
Schaeffer D, Somarelli JA, Hanna G, Palmer GM, Garcia-Blanco MA . Cellular migration and invasion uncoupled: increased migration is not an inexorable consequence of epithelial-to-mesenchymal transition. Mol Cell Biol 2014; 34: 3486–3499.
Even-Ram S, Yamada KM . Cell migration in 3D matrix. Curr Opin Cell Biol 2005; 17: 524–532.
Yang WH, Lan HY, Huang CH, Tai SK, Tzeng CH, Kao SY et al. RAC1 activation mediates Twist1-induced cancer cell migration. Nat Cell Biol 2012; 14: 366–374.
Choi YH, Burdick MD, Strieter BA, Mehrad B, Strieter RM . CXCR4 but not CXCR7, discriminates metastatic behavior in non-small cell lung cancer cells. Mol Cancer Res 2014; 12: 38–47.
Acknowledgements
This study was supported by the Ministry of Science and Technology (MOST 103-2320-B-010-039-MY3, 103-2321-B-010-029), the Ministry of Education (Aim for the Top University Plan), the Tri-Service General Hospital and the National Defense Medical Center (TSGHC-C103-062, TSGH-104-507) and the Taipei City Hospital, Taiwan.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Wu, YC., Tang, SJ., Sun, GH. et al. CXCR7 mediates TGFβ1-promoted EMT and tumor-initiating features in lung cancer. Oncogene 35, 2123–2132 (2016). https://doi.org/10.1038/onc.2015.274
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.274
This article is cited by
-
CXC chemokine receptor 4 (CXCR4) blockade in cancer treatment
Journal of Cancer Research and Clinical Oncology (2023)
-
Integrative stemness characteristics associated with prognosis and the immune microenvironment in lung adenocarcinoma
BMC Pulmonary Medicine (2022)
-
Cancer drug resistance induced by EMT: novel therapeutic strategies
Archives of Toxicology (2021)
-
Overexpression of CXCR7 accelerates tumor growth and metastasis of lung cancer cells
Respiratory Research (2020)
-
SOX4 promotes the growth and metastasis of breast cancer
Cancer Cell International (2020)