Abstract
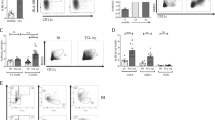
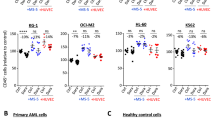
Chronic lymphocytic leukemia (CLL) is a malignant disease of small mature lymphocytes. Signals from the CLL microenvironment promote progression of the disease and induce drug resistance. This phenomenon is largely dependent on direct contact between the malignant B cells and stromal cells. CD84 belongs to the signaling lymphocyte activation molecule family of immunoreceptors, which self-associates, forming an orthogonal homophilic dimer. We therefore hypothesized that CD84 may bridge between CLL cells and their microenvironment, promoting cell survival. Our in vitro results show that CD84 expressed on CLL cells interact with CD84 expressed on cells in their microenvironment, inducing cell survival in both sides. Blocking CD84 in vitro and in vivo disrupt the interaction of CLL cells with their microenvironment, resulting in induced cell death. Thus, our findings suggest novel therapeutic strategies based on the blockade of this CD84-dependent survival pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Rozman C, Montserrat E . Chronic lymphocytic leukemia. N Engl J Med 1995; 333: 1052–1057.
Messmer BT, Messmer D, Allen SL, Kolitz JE, Kudalkar P, Cesar D et al. In vivo measurements document the dynamic cellular kinetics of chronic lymphocytic leukemia B cells. J Clin Invest 2005; 115: 755–764.
Burger JA . No cell is an island unto itself: the stromal microenvironment in chronic lymphocytic leukemia. Leukemia Res 2007; 31: 887–888.
Ramsay AD, Rodriguez-Justo M . Chronic lymphocytic leukaemia—the role of the microenvironment pathogenesis and therapy. Br J Haematol 2013; 162: 15–24.
Lagneaux L, Delforge A, Bron D, De Bruyn C, Stryckmans P . Chronic lymphocytic leukemic B cells but not normal B cells are rescued from apoptosis by contact with normal bone marrow stromal cells. Blood 1998; 91: 2387–2396.
Damiano JS, Cress AE, Hazlehurst LA, Shtil AA, Dalton WS . Cell adhesion mediated drug resistance (CAM-DR): role of integrins and resistance to apoptosis in human myeloma cell lines. Blood 1999; 93: 1658–1667.
Hallek M . Chronic lymphocytic leukemia: 2015 update on diagnosis, risk stratification, and treatment. Am J Hematol 2015; 90: 446–460.
Calpe S, Wang NH, Romero X, Berger SB, Lanyi A, Engel P et al. The SLAM and SAP gene families control innate and adaptive immune responses. Adv Immunol 2008; 97: 177–250.
Martin M, Romero X, de la Fuente MA, Tovar V, Zapater N, Esplugues E et al. CD84 functions as a homophilic adhesion molecule and enhances IFN-gamma secretion: adhesion is mediated by Ig-like domain 1. J Immunol 2001; 167: 3668–3676.
Romero X, Zapater N, Calvo M, Kalko SG, de la Fuente MA, Tovar V et al. CD229 (Ly9) lymphocyte cell surface receptor interacts homophilically through its N-terminal domain and relocalizes to the immunological synapse. J Immunol 2005; 174: 7033–7042.
Yan QR, Malashkevich VN, Fedorov A, Fedorov E, Cao E, Lary JW et al. Structure of CD84 provides insight into SLAM family function. Proc Natl Acad Sci USA 2007; 104: 10583–10588.
Binsky-Ehrenreich I, Marom A, Sobotta MC, Shvidel L, Berrebi A, Hazan-Halevy I et al. CD84 is a survival receptor for CLL cells. Oncogene 2014; 33: 1006–1016.
Nicholas NS, Apollonio B, Ramsay AG . Tumor microenvironment (TME)-driven immune suppression in B cell malignancy. Biochim Biophys Acta 2016; 1863: 471–482.
Bichi R, Shinton SA, Martin ES, Koval A, Calin GA, Cesari R et al. Human chronic lymphocytic leukemia modeled in mouse by targeted TCL1 expression. Proc Natl Acad Sci USA 2002; 99: 6955–6960.
Burger JA, Tsukada N, Burger M, Zvaifler NJ, Dell'Aquila M, Kipps TJ . Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood 2000; 96: 2655–2663.
Kurtova AV, Balakrishnan K, Chen R, Ding W, Schnabl S, Quiroga MP et al. Diverse marrow stromal cells protect CLL cells from spontaneous and drug-induced apoptosis: development of a reliable and reproducible system to assess stromal cell adhesion-mediated drug resistance. Blood 2009; 114: 4441–4450.
Burger JA, Ghia P, Rosenwald A, Caligaris-Cappio F . The microenvironment in mature B-cell malignancies: a target for new treatment strategies. Blood 2009; 114: 3367–3375.
Sivina M, Hartmann E, Kipps TJ, Rassenti L, Krupnik D, Lerner S et al. CCL3 (MIP-1alpha) plasma levels and the risk for disease progression in chronic lymphocytic leukemia. Blood 2011; 117: 1662–1669.
Burger JA, Quiroga MP, Hartmann E, Burkle A, Wierda WG, Keating MJ et al. High-level expression of the T-cell chemokines CCL3 and CCL4 by chronic lymphocytic leukemia B cells in nurselike cell cocultures and after BCR stimulation. Blood 2009; 113: 3050–3058.
Zucchetto A, Benedetti D, Tripodo C, Bomben R, Dal Bo M, Marconi D et al. CD38/CD31, the CCL3 and CCL4 chemokines, and CD49d/vascular cell adhesion molecule-1 are interchained by sequential events sustaining chronic lymphocytic leukemia cell survival. Cancer Res 2009; 69: 4001–4009.
Guan E, Wang J, Roderiquez G, Norcross MA . Natural truncation of the chemokine MIP-1 beta /CCL4 affects receptor specificity but not anti-HIV-1 activity. J Biol Chem 2002; 277: 32348–32352.
Binder M, Lechenne B, Ummanni R, Scharf C, Balabanov S, Trusch M et al. Stereotypical chronic lymphocytic leukemia B-cell receptors recognize survival promoting antigens on stromal cells. Plos One 2010; 5: e15992.
Kay NE, Shanafelt TD, Strege AK, Lee YK, Bone ND, Raza A . Bone biopsy derived marrow stromal elements rescue chronic lymphocytic leukemia B-cells from spontaneous and drug induced cell death and facilitates an "angiogenic switch". Leuk Res 2007; 31: 899–906.
Plander M, Ugocsai P, Seegers S, Orso E, Reichle A, Schmitz G et al. Chronic lymphocytic leukemia cells induce anti-apoptotic effects of bone marrow stroma. Ann Hematol 2011; 90: 1381–1390.
Bojarska-Junak A, Hus I, Szczepanek EW, Dmoszynska A, Rolinski J . Peripheral blood and bone marrow TNF and TNF receptors in early and advanced stages of B-CLL in correlation with ZAP-70 protein and CD38 antigen. Leuk Res 2008; 32: 225–233.
Reittie JE, Yong KL, Panayiotidis P, Hoffbrand AV . Interleukin-6 inhibits apoptosis and tumour necrosis factor induced proliferation of B-chronic lymphocytic leukaemia. Leuk Lymphoma 1996; 22: 83–90.
Moreno A, Villar ML, Camara C, Luque R, Cespon C, Gonzalez-Porque P et al. Interleukin-6 dimers produced by endothelial cells inhibit apoptosis of B-chronic lymphocytic leukemia cells. Blood 2001; 97: 242–249.
Binsky I, Lantner F, Grabovsky V, Harpaz N, Shvidel L, Berrebi A et al. TAp63 regulates VLA-4 expression and CLL cell migration to the BM in a CD74 dependent manner. J Immunol 2010; 184: 4761–4769.
Kalchenko V, Shivtiel S, Malina V, Lapid K, Haramati S, Lapidot T et al. Use of lipophilic near-infrared dye in whole-body optical imaging of hematopoietic cell homing. J Biomed Opt 2006; 11: 050507.
Hofbauer JP, Heyder C, Denk U, Kocher T, Holler C, Trapin D et al. Development of CLL in the TCL1 transgenic mouse model is associated with severe skewing of the T-cell compartment homologous to human CLL. Leukemia 2011; 25: 1452–1458.
ten Hacken E, Burger JA . Microenvironment dependency in chronic lymphocytic leukemia: the basis for new targeted therapies. Pharmacol Therapeut 2014; 144: 338–348.
Witowski J, Jorres A . Peritoneal cell culture: fibroblasts. Perit Dial Int 2006; 26: 292–299.
Witowski J, Thiel A, Dechend R, Dunkel K, Fouquet N, Bender TO et al. Synthesis of C-X-C and C-C chemokines by human peritoneal fibroblasts: induction by macrophage-derived cytokines. Am J Pathol 2001; 158: 1441–1450.
Cohen Y, Gutwein O, Garach-Jehoshua O, Bar-Haim A, Kornberg A . Bone marrow expression of CCL3 is not correlated with the extent of lytic bone lesions. Acta Haematol 2014; 132: 129–133.
Binsky I, Haran M, Starlets D, Gore Y, Lantner F, Harpaz N et al. IL-8 secreted in a macrophage migration-inhibitory factor- and CD74-dependent manner regulates B cell chronic lymphocytic leukemia survival. Proc Natl Acad Sci USA 2007; 104: 13408–13413.
Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Dohner H et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 2008; 111: 5446–5456.
Binet JL, Leporrier M, Dighiero G, Charron D, Dathis P, Vaugier G et al. Clinical staging system for chronic lymphocytic-leukemia—prognostic significance. Cancer 1977; 40: 855–864.
Haran M, Chebatco S, Flaishon L, Lantner F, Harpaz N, Valinsky L et al. Grb7 expression and cellular migration in chronic lymphocytic leukemia: a comparative study of early and advanced stage disease. Leukemia 2004; 18: 1948–1950.
Cannons JL, Qi H, Lu KT, Dutta M, Gomez-Rodriguez J, Cheng J et al. Optimal germinal center responses require a multistage T cell: B cell adhesion process involving integrins, SLAM-associated protein, and CD84. Immunity 2010; 32: 253–265.
Ray A, Dittel BN . Isolation of mouse peritoneal cavity cells. J Vis Exp 2010; 35: e1488.
Gordin M, Tesio M, Cohen S, Gore Y, Lantner F, Leng L et al. c-Met and its ligand hepatocyte growth factor/scatter factor regulate mature B cell survival in a pathway induced by CD74. J Immunol 2010; 185: 2020–2031.
Acknowledgements
We thank members of the Shachar laboratory for fruitful discussion and support. IS is the incumbent of the Dr Morton and Ann Kleiman Professorial Chair. This research was supported by the Israel Science Foundation, Israel Cancer association, Israel Cancer Research Fund (ICRF), DKFZ-MOST cooperation in cancer research, Quinquin foundation and Rubenstein charitable foundation.
Author contributions
AM, AFB, MPK, HL, IB-E ans SC- designed the research, performed the reserach and analyzed the data and wrote the paper. AT-K, VK, YK performed the research and analyzed the data. VM, ND, MS, PLS and LS contributed the vital reagent. YC performed the research, contributed the vital reagent. MH and YH contributed the vital reagent and designed the research. IS designed the research, analyzed the data and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Marom, A., Barak, A., Kramer, M. et al. CD84 mediates CLL-microenvironment interactions. Oncogene 36, 628–638 (2017). https://doi.org/10.1038/onc.2016.238
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.238
This article is cited by
-
The survival and function of IL-10-producing regulatory B cells are negatively controlled by SLAMF5
Nature Communications (2021)
-
Leukaemia: a model metastatic disease
Nature Reviews Cancer (2021)
-
Bone marrow dendritic cells support the survival of chronic lymphocytic leukemia cells in a CD84 dependent manner
Oncogene (2020)
-
FUS(1-359) transgenic mice as a model of ALS: pathophysiological and molecular aspects of the proteinopathy
neurogenetics (2018)