Abstract
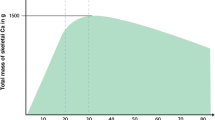
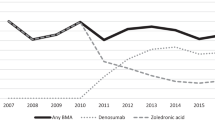
The objective of this study was to clarify the role of bisphosphonates in the treatment of osteoporosis in patients with prostate adenocarcinoma under androgen deprivation therapy (ADT). The Medline, EMBASE, Cancerlit and the American Society of Clinical Oncology abstract databases were searched for published randomized, placebo-controlled trials evaluating the usage of bisphosphonates in patients with prostate cancer (PC) under ADT. The outcomes assessed were fracture, osteoporosis, incidence of adverse events and changes in bone mineral density (BMD) during treatment. A total of 15 articles (2634 participants) were included in the meta-analysis. Treatment with bisphosphonates showed a substantial effect in preventing fractures (risk ratio (RR), 0.80; P=0.005) and osteoporosis (RR, 0.39; P <0.00001). Zoledronic acid showed the best number needed to treat (NTT), compared with placebo, in relation to fractures and osteoporosis (NNT=14.9 and NNT=2.68, respectively). The between-group difference (bisphosphonates vs placebo) in the lumbar spine and femoral neck BMD were 5.18±3.38% and 2.35±1.16%, respectively. This benefit of bone loss prevention could be reached without major side effects (cardiovascular or gastrointestinal events). Bisphosphonates are effective in preventing bone loss in patients with PC who are under ADT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Frydenberg M, Stricker PD, Kaye KW . Prostate cancer diagnosis and treatment. Lancet 1997; 349: 1681–1687.
Bubendorf L, Schopfer A, Wagner U, Sauter G, Moch H, Willi N et al. Metastatic patterns of prostate cancer: an autopsy study of 1589 patients. Hum Pathol 2000; 31: 578–583.
Carlin BI, Andriole GL . The natural history, skeletal complications, and management of bone metastases in patients with prostate carcinoma. Cancer 2000; 88: 2989–2994.
Eaton CL, Colby L, Coleman RE . Pathophysiology of bone metastases from prostate cancer and the role of bisphosphonates treatment. Cancer Treat Rev 2003; 29: 189–198.
Saad F, Schulman CC . Role of bisphosphonates in prostate cancer. Eur Urol 2004; 45: 26–34.
Brufsky AM . Cancer treatment-induced bone loss: patophysiology and clinical perspectives. Oncol 2008; 13: 187–195.
Diamond TH, Higano CS, Smith MR, Guise TA, Singer FR . Osteoporosis in men with prostate carcinoma receiving androgen-deprivation therapy: recommendations for diagnosis and therapies. Cancer 2004; 100: 892–899.
Assessment of Fracture Risk and its Application to Screening for Postmenopausal Osteoporosis WHO Technical Report Series 843 World Health Organization: Geneva.
Oefelein MG, Ricchiuti V, Conrad W, Resnick MI . Skeletal fractures negatively correlate with overall survival in men with prostate cancer. J Urol 2002; 168: 1005–1007.
Saad F, Higano CS, Sartor O, Colombel M, Murray R, Mason MD et al. The role of bisphosphonates in the treatment of prostate cancer: recommendations from an expert panel. Clin Genitourin Cancer 2006; 4: 257–262.
Reid IR . Bisphosphonates: new indications and methods of administration. Curr Opin Rheumatol 2003; 15: 458–463.
Diamond TH, Winters J, Smith A, De Souza P, Kersley JH, Lynch WJ et al. The antiosteoporotic efficacy of intravenous pamidronate in men with prostate carcinoma receiving combined androgen blockade: a double blind, randomized, placebo-controlled crossover study. Cancer 2001; 92: 1444–1450.
Bhoopalam N, Campbell SC, Moritz T, Broderick WR, Iyer P, Arcenas AG et al. Intravenous zoledronic acid to prevent osteoporosis in a veteran population with multiple risk factors for bone loss on androgen deprivation therapy. J Urol 2009; 182: 2257–2264.
Dearnaley DP, Sydes MR, Mason MD, Stott M, Powell CS, Robinson AC et al. A double-blind, placebo-controlled, randomized trial of oral sodium clodronate for metastatic prostate cancer (MRC PR05 Trial). J Natl Cancer Inst 2003; 17: 1300–1311.
Greenspan SL, Nelson JB, Trump DL, Resnick NM . Effect of once-weekly oral alendronate on bone loss in men receiving androgen deprivation therapy for prostate cancer: a randomized trial. Ann Intern Med 2007; 146: 416–424.
Greenspan SL, Nelson JB, Trump DL, Wagner JM, Miller ME, Perera S et al. Skeletal health after continuation, withdrawal, or delay of alendronate in men with prostate cancer undergoing androgen-deprivation therapy. J Clin Oncol 2008; 26: 4426–4434.
Magno C, Anastasi G, Morabito N, Gaudio A, Maisano D, Franchina F et al. Preventing bone loss during androgen deprivation therapy for prostate cancer: early experience with neridronate. Eur Urol 2005; 47: 575–580.
Michaelson MD, Kaufman DS, Lee H, McGovern FJ, Kantoff PW, Fallon MA et al. Randomized controlled trial of annual zoledronic acid to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer. J Clin Oncol 2007; 25: 1038–1042.
Rodrigues P, Hering FO, Bruna P, Meller A, Afonso Y . Comparative study of the protective effect of different intravenous bisphosphonates on the decrease in bone mineral density in patients submitted to radical prostatectomy undergoing androgen deprivation therapy. A prospective open-label controlled study. Int J Urol 2007; 14: 317–320.
Ryan CW, Huo D, Bylow K, Demers LM, Stadler WM, Henderson TO et al. Suppression of bone density loss and bone turnover in patients with hormone-sensitive prostate cancer and receiving zoledronic acid. BJU Int 2007; 100: 70–75.
Ryan CW, Huo D, Demers LM, Beer TM, Lacerna LV . Zoledronic acid initiated during the first year of androgen deprivation therapy increases bone mineral density in patients with prostate cancer. J Urol 2006; 176: 972–978.
Saad F, Gleason DM, Murray R, Tchekmedyian S, Venner P, Lacombe L et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst 2002; 94: 1458–1468.
Saad F, Gleason DM, Murray R, Tchekmedyian S, Venner P, Lacombe L et al. Long-term efficacy of zoledronic acid for the prevention of skeletal complications in patients with metastatic hormone-refractory prostate cancer. J Natl Cancer Inst 2004; 96: 879–882.
Small EJ, Smith MR, Seaman JJ, Petrone S, Kowalski MO . Combined analysis of two multicenter, randomized, placebo-controlled studies of pamidronate disodium for the palliation of bone pain in men with metastatic prostate cancer. J Clin Oncol 2003; 21: 4277–4284.
Smith MR, Eastham J, Gleason DM, Shasha D, Tchekmedyian S, Zinner N . Randomized controlled trial of zoledronic acid to prevent bone loss in men receiving androgen deprivation therapy for nonmetastatic prostate cancer. J Urol 2003; 169: 2008–2012.
Smith MR, McGovern FJ, Zietman AL, Fallon MA, Hayden DL, Schoenfeld DA et al. Pamidronate to prevent bone loss during androgen-deprivation therapy for prostate cancer. N Engl J Med 2001; 345: 948–955.
Nase JB, Suzuki JB . Osteonecrosis of the jaw and oral bisphosphonate treatment. JADA 2006; 137: 1115–1119.
Cartos VM, Zhu S, Zavras AI . Bisphosphonate use and the risk of adverse jaw outcomes. JADA 2008; 139: 23–30.
Yuen KK, Shelley M, Sze WM, Wilt T, Mason MD . Bisphosphonates for advanced prostate cancer. Cochrane Database Systematic Review 2006; 4, Article number CD006250.
Berry S, Waldron T, Winquist E, Lukka H . The use of bisphosphonates in men with hormone-refractory prostate cancer: a systematic review of randomized trials. Can J Urol 2006; 13: 3180–3188.
Saad F, Adachi JD, Brown JP, Canning LA, Gelmon KA, Josse RG et al. Cancer treatment-induced bone loss in breast and prostate cancer. J Clin Oncol 2008; 26: 5465–5476.
Eastham JA . Bone health in men receiving androgen deprivation therapy for prostate cancer. J Urol 2007; 177: 17–24.
Higano CS . Androgen-deprivation-therapy-induced fractures in men with nonmetastatic prostate cancer: what do we really know? Nat Clin Pract Urol 2008; 5: 24–34.
Polascik TJ . Bone health in prostate cancer patients receiving androgen-deprivation therapy: the role of bisphosphonates. Prostate Cancer Prostatic Dis 2008; 11: 13–19.
Smith MR, Fallon MA, Lee H, Finkelstein JS . Raloxifene to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer: a randomized controlled trial. J Clin Endocrinol Metab 2004; 89: 3841–3846.
Smith MR, Malkowicz SB, Chu F, Forrest J, Price D, Sieber P et al. Toremifene increases bone mineral density in men receiving androgen deprivation therapy for prostate cancer: interim analysis of a multicenter phase 3 clinical study. J Urol 2008; 179: 152–155.
Smith MR, Egerdie B, Hernández Toriz N, Feldman R, Tammela TL, Saad F et al. Denosumab in men receiving androgen-deprivation therapy for prostate cancer. N Engl J Med 2009; 361: 745–755.
Smith MR, Saad F, Egerdie B, Szwedowski M, Tammela TL, Ke C et al. Effects of denosumab on bone mineral density in men receiving androgen deprivation therapy for prostate cancer. J Urol 2009; 182: 2670–2675.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Prostate Cancer and Prostatic Diseases website
Rights and permissions
About this article
Cite this article
Serpa Neto, A., Tobias-Machado, M., Esteves, M. et al. Bisphosphonate therapy in patients under androgen deprivation therapy for prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis 15, 36–44 (2012). https://doi.org/10.1038/pcan.2011.4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2011.4