Abstract
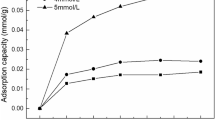
A viscose rayon succinate (VRS) was synthesized using viscose rayon, succinic anhydride and DMSO. The VRS was well characterized by C13 nuclear magnetic resonance, Fourier transform infrared analysis and properties of the VRS up taking trivalent metals from aqueous solution were investigated. Both esterification and carboxyl bonding of the VRS were assigned essentially at 1729 and 1693 cm−1, respectively. And the essential band of bonding between metal and the material was determined at 1605–1639 cm−1. The available adsorption capacity, degree of substitution and pKa of the VRS were 6.2mequiv/g, 2.5 and 3.72, respectively. The adsorption of metal ions on the VRS follows the order of Cr3+>Al3+>Fe3+ with maximum adsorptions capacities 1.48, 1.059 and 1.029 mmol/g. An equilibrium modeling, which is obtained from chemical potential and Boltzmann distribution, was demonstrated trivalent metal adsorption on the VRS in different pHs. Surface potential, Ψ0, an effective ratio of surface equilibrium constants, Keffect, the probability factor P(A) and a degree of protonation, χ represent the physicochemical interactions between carboxyl group and metal ions.
Similar content being viewed by others
Article PDF
References
V. Vuorio, J. A. Manzanares, L. Murtomaki, J. Hirvonen, T. Kankkunen, and K. Kontturi, J. Controlled Release, 91, 439 (2003).
O. A. El Seoud and T. Heinze, Adv. Polym. Sci., 186, 103 (2005).
K. J. Edgar, C. M. Buchanan, J. S. Debenham, P. A. Rundquist, B. D. Seiler, M. C. Shelton, and D. Tindall, Prog. Polym. Sci., 26, 1605 (2001).
W. M. Hosny, Polym. Int., 42, 157 (1997).
T. W. Swaddle, J. Rosenqvist, P. Yu, E. Bylaska, B. L. Phillips, and W. H. Casey, Science, 308, 1450 (2005).
M. A. S. D. Barros, A. S. Zola, P. A. Arroyo, C. R. G. Tavares, and E. F. Sousa-Aguiar, Adsorption, 12, 239 (2006).
C. W. MacDiarmid and R. C. Gardner, J. Biol. Chem., 273, 1727 (1998).
H. Jacqmin, D. Commenges, L. Letenneur, P. Barberger-Gateau, and J.-F. Dartigues, Am. J. Epidemiol., 139, 48 (1994).
S. Lacour, V. Deluchat, J.-C. Bollinger, and S. Bernard, Talanta, 46, 999 (1998).
P. M. Outridge and A. M. Scheuhammer, Rev. Environ. Contam. Toxicol., 130, 31 (1993).
W. N. Harrison, S. M. Bradberry, and J. A. Vale, Clinical Toxicology, 38, 137 (2000).
W. Lin and Y. L. Hsieh, Ind. Eng. Chem. Res., 35, 3817 (1996).
F. Beolchini, F. Pagnanelli, L. Toro, and F. Veglio, Water Res., 40, 144 (2006).
Z. Reddad, C. Gerente, Y. Andres, and P. L. Cloirec, Environ. Sci. Technol., 36, 2067 (2002).
A. Esposito, F. Pagnanelli, and F. Veglio, Chem. Eng. Sci., 57, 307 (2002).
J. Peydecastaing, S. Girardeau, C. Vaca-Garcia, and M. Borredon, Cellulose, 13, 95 (2006).
H. Yuksek, M. Alkan, S. Bahceci, I. Cakmak, Z. Ocak, H. Baykara, O. Aktas, and E. Agyel, J. Mol. Struct., 873 142 (2008).
N. B. Colthup, L. H. Daly, and S. E. Wiberley, “Introduction to Infrared and Raman Spectroscopy,” Academic Press Inc, London, 1964.
R. V. S. Alfaya and Y. Gushikem, J. Colloid Interface Sci., 213, 438 (1999).
A. A. M. A. Nada and M. L. Hassan, J. Appl. Polym. Sci., 102, 1399 (2006).
Y. G. Ko, U. S. Choi, B. G. Ahn, and D. J. Ahn, J. Polym. Sci., Part A: Polym. Chem., 38, 2815 (2000).
D. F. Evans and H. Wennerstrom, “The Colloidal Domain: Where Physics, Chemistry, Biology, and Technology Meet,” 2nd ed., Wiley, New York, 1999 p 127.
A. V. Dobrynin, R. H. Colby, and M. Rubinstein, Macromolecules, 28, 1859 (1995).
R. Schweiss, P. B. Welzel, C. Werner, and W. Knoll, Langmuir, 17, 4304 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khasbaatar, A., Chun, Y. & Choi, U. Trivalent Metal Adsorption Properties on Synthesized Viscose Rayon Succinate. Polym J 40, 302–309 (2008). https://doi.org/10.1295/polymj.PJ2007127
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2007127