Abstract
Thrombocytopenia is common in sick preterm babies. Despite this, platelet production in thrombocytopenic preterm babies has rarely been assessed. To address this problem we have developed miniaturized assays to study circulating megakaryocyte (MK) progenitors [burst-forming unit (BFU)-MK and colony-forming unit (CFU)-MK], total cultured MK precursors and mature MK, by culturing mononuclear cells purified from 0.5-1 mL of preterm peripheral blood. MK lineage colonies and cells are identified by an anti-IIb/IIIa antibody (CD61). We prospectively studied circulating BFU-MK/CFU-MK, total cultured MK precursors and mature MK in 63 preterm babies (gestational age 24-34 wk). Twenty-six developed early thrombocytopenia (platelets <150× 109/L by 48 h), whereas the remaining 37 babies maintained normal platelet counts. Twenty-one of the 26 thrombocytopenic babies were born to mothers with pregnancy-induced hypertension or were growth retarded. At birth, thrombocytopenic babies had severely reduced numbers of all MK precursors compared with nonthrombocytopenic babies: BFU-MK 82 ± 50versus 663 ± 174 colonies/mL, mean ± SEM; CFU-MK 596± 196 versus 3267 ± 530 colonies/mL; total MK precursors 97 ± 30 versus 301 ± 49 × 103 cells/mL and mature MK 8 ± 2 versus 37 ± 8 × 103cells/mL, respectively. Thrombocytopenia resolved by d 10 in all babies accompanied or preceded by a recovery to normal numbers of circulating MK progenitors. Eighteen (69%) of the thrombocytopenic babies were also neutropenic (neutrophils <2 × 109/L); in these babies neutrophil progenitor cells (CFU-granulocyte/monocyte) were also severely reduced compared with the nonthrombocytopenic babies (539 ± 280versus 1937 ± 348 colonies/mL, mean ± SEM). This indicates that the principal cause of the thrombocytopenia and neutropenia is reduced platelet and neutrophil production occurring as a consequence of reduced numbers of MK and CFU-granuloctype/monocyte progenitors, respectively. Taken together these data suggest the hematologic abnormalities characteristic of newborns born to mothers with pregnancy-induced hypertension or with intrauterine growth retardation are a consequence of dysregulation of fetal hemopoiesis occurring proximal to committed MK and neutrophil progenitors, most likely at the level of the primitive multipotent hemopoietic stem cell.
Similar content being viewed by others
Main
Platelet counts in the healthy human fetus reach adult levels (150-450× 109/L) by the second trimester of pregnancy(1, 2). However, thrombocytopenia is common in the sick newborn baby, occurring in 22-35% of all babies admitted to neonatal intensive care units(3, 4), and in up to 72% of the sick preterm population(4). This high incidence of neonatal thrombocytopenia in preterm babies, coupled with its association with the more severe grades of intraventricular hemorrhage(5, 6), make it one of the most important hematologic problems encountered in the newborn.
Many maternal, fetal, and neonatal conditions are associated with neonatal thrombocytopenia. In some of these conditions the mechanism of thrombocytopenia is well recognized; transplacental passage of maternal auto(7) alloantibodies(8); neonatal giant hemangioma(9); neonatal marrow replacement(10) dysplastic syndromes(11). In others, including birth asphyxia, neonatal bacterial/viral infection and necrotizing enterocolitis, the main mechanism of thrombocytopenia has traditionally been felt to be platelet consumption resulting from disseminated intravascular coagulation(12). More recent evidence suggests that immune-mediated platelet consumption plays a significant role in at least some of these conditions(3, 13, 14). Nevertheless, there remains a large number of conditions, all with well documented associations with thrombocytopenia in the newborn, including PIH(15), IUGR(16), maternal diabetes(17), and red cell alloimmunization(18), in which the mechanism of the thrombocytopenia is unknown.
In the two largest published studies, the underlying mechanisms responsible for neonatal thrombocytopenia remained unknown in 20-60% of cases(3, 4). However, Castle et al.(3) demonstrated the interesting finding that thrombocytopenia occurs mainly in the first few days of life and often follows a regular pattern (seemingly regardless of cause) with a platelet nadir around d 4 and recovery of platelet numbers by the end of the 1st wk. This remarkably regular pattern of early thrombocytopenia, coupled with the high percentage of cases of unknown cause, suggests that an as yet unreported underlying mechanism may be responsible for a large number of episodes of neonatal thrombocytopenia.
Most currently available methods for investigating neonatal thrombocytopenia look for evidence of platelet consumption. There exists to date no large study of platelet production in thrombocytopenic newborns. [In their studies Castle et al.(3) and Mehtaet al.(4) indirectly assessed platelet production by looking at postmortem bone marrow MK numbers in 6 (3%) and 14(11%), respectively, of their thrombocytopenic babies. However, their findings differ markedly, with Castle et al. reporting normal numbers of MK in all six thrombocytopenic babies examined, whereas Mehta et al. report reduced numbers of MK in 12 of their 14 babies.]
In the normal fetus and neonate, platelet production is maintained by the progressive commitment of undifferentiated hemopoietic stem cells to lineage-restricted MK progenitors with further differentiation producing megakaryoblasts, mature MK, and finally platelets. The main site of fetal hemopoiesis, including megakaryocytopoiesis(19), shifts with increasing gestational age from the yolk sac to the liver with the bone marrow only becoming the major hemopoietic organ in the third trimester. Bone marrow examination is rarely undertaken in neonatal clinical practice, but large numbers of multipotent and committed hemopoietic progenitors circulate in the peripheral blood of term(20), and to an even greater extent preterm, babies(21–23), where they are thought to represent a migratory population of progenitors moving from these early gestational sites of hemopoiesis to populate the bone marrow. In the fetus and neonate it is not yet known if the numbers of circulating MK progenitor and precursor cells reflect megakaryocytopoiesis(and therefore platelet production) in general. However, as assaying circulating MK progenitor and precursor cells, especially in preterm babies, theoretically gives access to a large proportion of total MK-producing cells, it seems likely that this approach (at least to some extent) will reflect fetal/neonatal platelet production. To this end we have recently developed miniaturized assays which allow us to assess by in vitro culture of mononuclear cells purified from 0.5-1 mL of neonatal peripheral blood(23): 1) the circulating numbers of the earliest committed MK progenitors-BFU-MK and CFU-MK; 2) the total number of committed MK precursors (BFU/MK, CFU-MK, and megakaryoblasts) obtained in liquid culture from neonatal mononuclear cells; and 3) the ability of these cells to differentiate in vitro into mature MK. The small amount of peripheral blood required to perform these assays enables us to perform longitudinal studies on babies born with, or developing, thrombocytopenia in the early neonatal period.
The aim of this study was to assess megakaryocytopoiesis, as an index of platelet production, in preterm babies with early neonatal thrombocytopenia and in gestational age-matched nonthrombocytopenic preterm controls by assaying circulating BFU-MK, CFU-MK, and total cultured MK precursors, and by assessing the in vitro capacity of these cells to differentiate to produce mature MK.
METHODS
Patients
Subjects were drawn from admissions to the Neonatal Unit at the Hammersmith Hospital. All blood specimens were obtained at times of routine sampling for diagnostic purposes. Culture studies for circulating BFU-MK, CFU-MK, total cultured MK precursors, and mature MK were performed prospectively on all preterm (gestational age ≤34 wk) admissions from whom an initial specimen of peripheral blood was obtained within 24 h of birth. When specimens yielded sufficient mononuclear cells, circulating CFU-GM and BFU-e progenitors were also assayed. Follow-up studies were performed as clinical sampling allowed. The study was approved by the Research Ethics Committee of Hammersmith and Queen Charlotte's Special Health Authority.
Progenitor Assays
All assays were carried out using LDADM cells as previously described(23).
Agar culture for BFU-MK and CFU-MK. LDADM cells were suspended at 4 × 105mL in 0.3% Bacto-Agar (Difco Laboratories, Detroit, MI) with IMDM (ICN Biomedicals Ltd, Thame, Oxfordshire) supplemented (to a final concentration) with 20% pooled human AB plasma, 1% BSA (Life Technologies, Inc., Paisley, Scotland), 200 μg/mL iron saturated transferrin, 2 × 10-5 M α-monothioglycerol (Sigma Chemical Co., St. Louis, MO), 10μL/mL minimal essential medium nonessential amino acids (100×), and 10 μL/mL minimal essential medium vitamins solution (100×) (Life Technologies, Inc.). As a MK progenitor cell growth stimulator, 100 U/mL human IL-3 was used. Aliquots (50 μL) of this suspension were placed into the wells of flat-bottomed 96-well tissue culture plates (Nunc, Roskilde, Denmark). When the cell suspension set, a further 50 μL of feeder layer(IMDM supplemented as above) were placed on top of each cell suspension pellet. Plates were then incubated at 37 °C in a humidified atmosphere of 5% CO2 in air for 21 d. After, cell pellets were mobilized by adding 2% formalin to each well for 60 s. Cell pellets were then removed from the tissue culture plate, soaked overnight in distilled water, and air-dried on glass microscope slides. Slides were either processed immediately, or stored at -20°C before MK progenitor colony identification.
Liquid culture for total cultured MK precursors and mature MK. LDADM cells were suspended at 106/mL in tissue culture tubes (Nunc) in IMDM supplemented (to final concentrations as above). Human IL-3 (100 units/mL) was used as a MK progenitor growth stimulator, and cultures were incubated at 37 °C in a humidified atmosphere of 5% CO2 in air. Cultures were fed weekly with fresh medium until exhausted (usually 3-4 wk). Each week total cell numbers were counted, and a standard number of cells (5× 104) were removed and cytospun onto a glass microscope slide. Slides were then air-dried, and either processed immediately or stored at -20°C before identification of MK precursors and mature MK.
Identification of MK Lineage Colonies and Cells
MK lineage colonies and cells (cultured in both systems) were identified by the alkaline phosphatase APAAP technique, using a MAb (CD61, Dako, Denmark) directed against the platelet glycoprotein IIb/IIIa complex. Before fixing slides carrying dried agar pellets were first covered with PBS (pH 7.4) containing agarase (Sigma Chemical Co.), 10 units/mL for 60 min at 37 °C. These were then washed in Tris-buffered saline (pH 7.6) and fixed. Slides were fixed with cold (4 °C) methanol-acetone for 90 s for liquid culture slides, and 5 min for agar culture slides. Slides were then flooded with the primary antibody (CD61) at a 1:100 dilution, and incubated for 30 min (liquid culture) and 60 min (agar culture) in a humidified atmosphere at 37 °C. Next, slides were incubated with rabbit anti-mouse Ig and then with APAAP complex (Dako, Denmark) both at 1:100 dilutions, for the same periods. Incubations with rabbit anti-mouse Ig and APAAP complex were then repeated for 10 min (liquid culture) and 30 min (agar culture). Finally, slides were incubated with substrate solution containing levamisole (Sigma Chemical Co.) for 30 min, and then counterstained with hematoxylin solution Gill No. 1(Sigma Chemical Co.). BFU-MK and CFU-MK were identified by the criterion of Briddell et al.(24), with results expressed as the mean of colony numbers from at least three agar pellets. Peak numbers of cultured MK precursors (small positively staining mononuclear cells-predominantly megakaryoblasts) and mature MK (large positively staining cells containing three or more nuclei) were calculated: peak liquid culture cell count ×% of MK precursors and mature MK labeled on matching cytospin. (APAAP staining of LDADM cells before liquid culture demonstrated no mature MK in any subject.) The assays were controlled by staining without primary antibody and by staining with unrelated antibodies. BFU-e and CFU-GM were assayed in methylcellulose by a modification of the technique of Westwoodet al.(25).
Statistics
All statistical comparisons between groups were carried out by the Mann Whitney U test unless otherwise stated.
RESULTS
Circulating MK progenitor and precursor cell studies were carried out in 63 preterm babies at birth. Early thrombocytopenia (present at birth or developing within 48 h) occurred in 26 of these babies. All of the remaining 37 babies maintained platelet counts >150 × 109/L throughout the 1st wk of life. Subject characteristics, platelet count nadir, and the main antenatal and neonatal complications are shown in Table 1. Early thrombocytopenia was significantly associated with maternal PIH, IUGR, and neonatal mortality, but was not associated with maternal infection(clinical chorioamnionitis). Indeed, maternal infection occurred significantly less frequently in babies with early thrombocytopenia compared with those who maintained normal platelet counts.
Circulating MK Progenitor Cell Studies
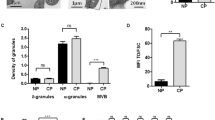
At birth. BFU-MK, CFU-MK, total cultured MK precursors, and mature MK were all significantly reduced in the group of babies with early neonatal thrombocytopenia compared with those babies who maintained normal platelet counts: BFU-MK 82 ± 50 versus 663 ± 174 colonies/mL, mean ± SEM; CFU-MK 596 ± 196 versus 3267± 530 colonies/mL; total MK precursors 97 ± 30 versus 301 ± 49 × 103 cells/mL and mature MK 8 ± 2versus 37 ± 8 × 103cells/mL, respectively(Fig. 1 andTable 2). Furthermore, in both thrombocytopenic and nonthrombocytopenic babies total circulating MK progenitor cell numbers (BFU-MK plus CFU-MK) at birth were closely correlated with the platelet nadir in the 1st wk of life (Fig. 2).
(A) Circulating BFU-MK and CFU-MK in 26 thrombocytopenic (⁃) and 37 nonthrombocytopenic (○) babies at birth. Results are expressed as MK colonies/mL of peripheral blood. (B) Circulating MK precursors (MKp) and mature MK in 26 thrombocytopenic(•) and 37 nonthrombocytopenic (○) babies at birth. Results are expressed as cells/mL (× 1000) of peripheral blood.
Follow-up studies. Thrombocytopenia resolved in 18 of the 21 surviving thrombocytopenic babies by d 7 and in the remaining 3 babies by d 10. Longitudinal studies demonstrated a dramatically different pattern of change in the numbers of circulating BFU-MK and CFU-MK over the 1st wk in the thrombocytopenic babies when compared with the nonthrombocytopenic babies. The number of MK progenitors increased in those with early neonatal thrombocytopenia, but were maintained or fell in nonthrombocytopenic babies(Fig. 3), so that by d 7 there was no longer a significant difference between the numbers of circulating MK progenitors in the two groups (Table 3).
(A) Longitudinal study of circulating BFU-MK in 14 thrombocytopenic (▪) and 18 nonthrombocytopenic (□) babies. Results expressed from studies on each baby on d 1 and 7 and also on d 3 from six thrombocytopenic and seven nonthrombocytopenic babies. Results expressed as BFU-MK colonies/mL of peripheral blood. (B) Longitudinal-study of circulating CFU-MK in 14 thrombocytopenic (▪) and 18 nonthrombocytopenic(□) babies. Results expressed from studies on each baby on d 1 and 7 and also on d 3 from six thrombocytopenic and seven nonthrombocytopenic babies. Results expressed as CFU-MK colonies/mL of peripheral blood.
[Of those assessed only one thrombocytopenic baby did not show this trend of rising circulating MK progenitors over the 1st wk of life (seeFig. 3). This baby's mother had short-lived, but rapidly worsening PIH. The baby was not growth-retarded (birth weight 50th centile) and developed only mild thrombocytopenia (platelets 129 × 109/L by 48 h). However, the baby went on to develop extensive necrotizing enterocolitis associated with severe thrombocytopenia (platelets <30× 109/L) by d 10. Therefore, it is possible that concurrent pathologic processes were responsible for the thrombocytopenia in this baby and that this baby is not representative of the thrombocytopenic group as a whole.]
Circulating CFU-GM and BFU-e Progenitors
At birth 18 babies (69%) with early thrombocytopenia were also neutropenic(neutrophils <2.0 × 109/L). The clinical cause of the neutropenia in these babies was felt to be: PIH ± IUGR in 14, IUGR alone in three, and culture positive sepsis in one. Only three babies (8%) with normal platelet counts were neutropenic, and all three of these babies had culture-positive sepsis at birth. CFU-GM and BFU-e were assayed at birth in all 18 of the babies with thrombocytopenia and neutropenia, in 19 of those with normal platelet and neutrophil counts, and in two of the three babies with neutropenia but normal platelets. CFU-GM were severely reduced in the group with thrombocytopenia and neutropenia when compared with those with normal platelet and neutrophil counts-539 ± 280 versus 1937± 348 colonies/mL (mean ± SEM), respectively(Table 4). In contrast large numbers of CFU-GM (2581 and 3566 colonies/mL) were found in the two babies with neutropenia but normal platelet counts. BFU-e were found in large numbers in all babies and were not significantly different in babies with early thrombocytopenia and neutropenia compared with those with normal platelet and neutrophil counts(Table 4).
DISCUSSION
Thrombocytopenia in the preterm newborn is often associated with a high mortality and morbidity. Despite this, investigation is frequently difficult and in many cases the cause remains elusive. The underlying mechanism must be the consequence either of reduced platelet production, of platelet sequestration (e.g. in the spleen), of increased platelet consumption, or of a combination of these factors. Despite extensive investigation of the mechanisms of increased platelet consumption/sequestration, very little is known about platelet production in thrombocytopenic babies, particularly in preterm infants.
To address this, we prospectively assessed circulating MK progenitors as an index of platelet production in 63 preterm babies, 26 of whom (41%) were thrombocytopenic in the 1st wk of life. We found that at birth the numbers of circulating MK progenitor and precursor cells were severely reduced in the thrombocytopenic babies compared with those babies who did not develop thrombocytopenia. The degree of suppression was similar at all levels of the MK differentiation pathway from the earliest recognizable progenitor cell of the MK lineage (BFU-MK) to mature MK. We suggest, therefore, that the thrombocytopenia seen in such babies is a direct consequence of reduced platelet production secondary to low numbers of primitive MK progenitors. No compensatory increase in MK ploidy or change in MK cytoplasmic volume was seen in the liquid MK cultures of thrombocytopenic babies compared with nonthrombocytopenic babies (data not shown), suggesting that the former were unable to compensate for the lack of progenitor cells by increasing platelet production from more mature MK precursors.
Further evidence of overall reduced platelet production is provided by the contrast between the change in MK progenitors seen during the first week of life in thrombocytopenic and nonthrombocytopenic babies. Whereas MK progenitor numbers in thrombocytopenic babies rose (accompanying or, often, preceding platelet recovery), MK progenitors remained stable or fell in nonthrombocytopenic babies so that at 1 wk of age there was no longer any difference between the two groups. This suggests a “normalization” of megakaryocytopoiesis over the 1st wk of life in the babies with early thrombocytopenia, followed by an increase in platelet production and hence recovery of platelet numbers.
In our Unit the main factor associated with early neonatal thrombocytopenia was maternal PIH. This was present in 69% of the mothers of those with early thrombocytopenia, reflecting our referral pattern (an obstetric tertiary referral center for this disease). The mechanism of fetal/neonatal (and maternal) thrombocytopenia in pregnancies complicated by PIH is uncertain, but most current evidence has implicated immune-mediated platelet consumption. PIH has been reported to be associated with raised levels of maternal PAIg(26–29) and platelet-associated IgG, IgM, and C3(27–29) have also been found to be raised in babies of mothers with PIH. Indeed, Castle et al.(3) have suggested that the principal mechanism for thrombocytopenia in newborns requiring intensive care is immune-mediated platelet consumption as a consequence of raised levels of neonatal platelet-associated IgG, thus leading to reduced platelet survival time(14). (However, PIH was recorded in only 10% of the mothers of babies with severe thrombocytopenia-platelets <100 × 109/L in their study.)
In contrast to the evidence supporting immune-mediated platelet consumption in both mothers with PIH and their babies and in thrombocytopenic newborns in general: a recent study found normal levels of maternal PAIg in PIH(30); whereas raised levels of PAIg in nonthrombocytopenic mothers with PIH(26, 27); raised levels of PAIg in healthy nonthrombocytopenic pregnant women(27); and raised levels of PAIg in nonthrombocytopenic newborns after both normal pregnancies(3, 27) and pregnancies complicated by PIH(27) are all reported in the same studies. Taken together these data suggest that immune-mediated platelet consumption is unlikely to be either the only, or principle, mechanism of fetal/neonatal thrombocytopenia in babies of mothers with PIH.
In view of these conflicting findings, the need to minimize sample size and inherent methodologic difficulties, PAIg and platelet survival time were not measured in our study babies or their mothers. We, therefore, cannot exclude completely the possibility that some of the reduction in circulating megakaryocyte progenitor and precursor cells found in thrombocytopenic babies could have resulted from immune-mediated consumption of these cells by PAIg formed in association with maternal PIH. However, only 3 (14%) of the 21 study mothers with PIH were thrombocytopenic at delivery. In contrast, the vast majority of babies of mothers with PIH were thrombocytopenic 18/21 (86%) and virtually all the thrombocytopenic babies of mothers with PIH 17/18 (94%) had low numbers of circulating MK progenitor and precursor cells. These data suggest that the reduction in MK progenitor and precursor cells and resulting thrombocytopenia seen in babies of mothers with PIH is unlikely to be the result of transplacental passage of maternal PAIg, as in this situation thrombocytopenia would be expected to be present in a far higher proportion of the mothers with PIH. In addition, we have previously found normal to increased numbers of circulating MK precursors in thrombocytopenic preterm babies with severe infections and necrotizing enterocolitis(31) [both of which are associated with increased levels of platelet associated IgG(13, 14)]. This finding further suggests that PAIg when present in preterm babies do not cause thrombocytopenia principally by destruction/inhibition of MK progenitor and precursor cells.
If the reduction in circulating megakaryocyte progenitor and precursor cells seen in thrombocytopenic babies of mothers with PIH in this study is not immune-mediated, then what is the likely mechanism? Maternal PIH is strongly associated not only with early neonatal thrombocytopenia, but also with early neonatal neutropenia(32). Previous studies have already shown that the neonatal neutropenia common in babies of mothers with PIH is the result of reduced neutrophil production as a consequence of reduced numbers of CFU-GM(32, 33). In addition this impairment of granulopoiesis is transient “normalizing” over the 1st wk of life in common with the increase in MK progenitors seen in babies with early neonatal thrombocytopenia in this study.
Not surprisingly a large proportion (69%) of babies with early thrombocytopenia in this study were concurrently neutropenic at birth. The clinical cause of the neutropenia in these babies was felt to be maternal PIH± IUGR in 14 cases, IUGR alone in three cases, and proven sepsis in only one case. Confirming the previous studies cited above, these babies also had severely reduced numbers of circulating CFU-GM at birth when compared with babies with normal platelet and neutrophil counts. [Interestingly the one thrombocytopenic/neutropenic baby with proven sepsis who suffered neither maternal PIH or IUGR (see above), still had low numbers of both MK progenitors(BFU-MK, 87 colonies/mL; CFU-MK, 438 colonies/mL) and CFU-GM (489 colonies/mL) at birth. This is in direct contrast to the two neutropenic babies with proven sepsis who maintained normal platelet counts and had normal to increased numbers of MK progenitors and CFU-GM (see “Results”). This suggests that the thrombocytopenic/neutropenic baby with proven sepsis had an underlying abnormality of both MK and neutrophil progenitors unrelated to the onset of sepsis, although the cause of this abnormality at this stage is obscure).
However, these results show that babies from mothers with PIH ± IUGR or indeed babies with fetal IUGR alone for other reasons, commonly share not only the clinical presentation of early neonatal thrombocytopenia and neutropenia, but also concurrent reductions of circulating MK and neutrophil progenitor cells and their progeny. These progenitor cell abnormalities are present at birth and must, therefore, have their origins in fetal life.
In addition to early thrombocytopenia and neutropenia, many preterm babies born to mothers with PIH or with IUGR are polycythemic at birth(34–36). This specific pattern of hematologic abnormalities (thrombocytopenia, neutropenia, and polycythemia) is a common finding too in infants of diabetic mothers(17). Thrombocytopenia and neutropenia are also common in babies with red cell alloimmunization(37). Thus, these diverse syndromes of fetomaternal abnormalities all display an increased drive to fetal erythropoiesis, as a consequence of raised levels of fetal erythropoietin, thought to be stimulated by fetal tissue hypoxia(38, 39).
It is not known whether this erythropoietin-driven increased fetal erythropoiesis has any baring on the development of the abnormalities of circulating MK and granulocyte progenitors seen in such babies in this study. Indeed, with the data from this study alone it is not possible to conclude with certainty that these abnormalities of peripheral MK and granulocyte progenitor cells reflect true impairments of fetal/neonatal megakaryocytopoiesis and granulopoiesis. However, similar bone marrow hemopoietic and peripheral blood hematologic changes are known to occur in experimental animals exposed to long-term hypoxia(40, 41). Indeed, because platelets, neutrophils, and erythrocytes all derive from a common multipotent hemopoietic progenitor cell-CFU-GEMM-an erythropoietin-driven disruption of normal hemopoiesis at this primitive progenitor cell level, leading to an imbalance of commitment to lineage restricted progenitor cells, is an attractive hypothesis to explain all of the observed neonatal hematologic abnormalities in such babies.
If this single mechanism (excess erythropoietin) is responsible for multiple disruptions of fetal hemopoiesis, then two modes of action are compatible with the results of this study. A state of increased erythropoietin could allow continued commitment of CFU-GEMM to erythropoiesis (maintaining BFU-e numbers), at the expense of the production of committed megakaryocyte and neutrophil progenitor cells, despite developing polycythemia which might be expected to down-regulate erythropoiesis. Alternatively, erythropoietin may have no effect on CFU-GEMM lineage commitment, but in addition to its stimulatory effects on erythroid progenitor cells, exhibit a direct inhibitory effect on committed megakaryocyte and neutrophil progenitor cells.
Furthermore, erythropoietin falls to very low levels within hours of birth, even in babies with considerably raised levels at birth(42). Thus if erythropoietin does exert a mechanistic role in impairment of fetal megakaryocytopoiesis and granulopoiesis this would be consistent with the rapid return to normal levels of circulating MK progenitor and precursor cells seen after birth in the thrombocytopenic babies in this study. Further studies will clearly be required to fully evaluate these theories.
In conclusion, this study is the first to show that the majority of preterm babies with early thrombocytopenia have reduced numbers of circulating MK progenitor and precursor cells at birth. We have also demonstrated an increase in circulating MK progenitor cell numbers in thrombocytopenic babies to“control” levels over the 1st wk of life in tandem with resolution of thrombocytopenia. Although the evidence is indirect, we believe these data strongly suggest that the main underlying mechanism of early thrombocytopenia in such babies is platelet underproduction as a consequence of impaired fetal megakaryocytopoiesis.
In addition, this study demonstrates that thrombocytopenic preterm babies are commonly also neutropenic at birth and that this neutropenia is associated with a concurrent reduction in circulating primitive granulocyte progenitor cells. Taken together these data suggest multiple disruptions of fetal hemopoiesis (possibly with their origins in feto/maternal pregnancy complications resulting in fetal hypoxia) may be responsible for many of the common hematologic problems of preterm babies in the immediate neonatal period.
Although further studies will be required to confirm these findings and resultant theories, they do begin to provide a scientific foundation for the use of hemopoietic growth factors (granuloctye-CSF, GM-CSF, and thrombopoietin) in the treatment of the common hematologic problems of preterm babies.
Abbreviations
- MK:
-
megakaryocyte
- BFU:
-
burst-forming unit
- CFU:
-
colony-forming unit
- GM:
-
granulocyte/monocyte
- BFU-e:
-
burst-forming unit erythroid
- PIH:
-
pregnancy-induced hypertension
- IUGR:
-
intrauterine growth retardation
- LDADM:
-
light density adherent cell-depleted peripheral blood mononuclear
- APAAP:
-
alkaline phosphatase anti-alkaline phosphatase
- GEMM:
-
granulocyte/erythroid/monocyte/megakaryocyte
- IMDM:
-
Iscove's modification of Dulbecco's medium
- PAIg:
-
platelet-associated immunoglobulin
References
Forestier F, Daffos F, Catherine N, Renard M, Andreux J-P 1991 Developmental hematopoiesis in normal human fetal blood. Blood 77: 2360–2363
Holmberg L, Gustavii B, Jonsson A 1983 A prenatal study of fetal platelet count and size with application to fetus at risk for Wiskhott Aldrich syndrome. J Pediatr 102: 773–781
Castle V, Andrew M, Kelton J, Giron D, Johnston M, Carter C 1986 Frequency and mechanism of neonatal thrombocytopenia. J Pediatr 108: 749–755
Mehta P, Vasa R, Neumann L, Karpatkin M 1980 Thrombocytopenia in the high risk infant. J Pediatr 97: 791–794
Andrew M, Castle V, Saigal S, Carter C, Kelton J 1987 Clinical impact of neonatal thrombocytopenia. J Pediatr 110: 457–464
Van De Bor M, Briet E, Van Bel F, Ruys JH 1986 Hemostasis and periventricular-intraventricular hemorrhage in the newborn. Am J Dis Child 140: 1131–1134
Cook RL, Miller RC, Katz VL, Cefalo RC 1991 Immune thrombocytopenic purpura in pregnancy: A reappraisal of management. Obstet Gynecol 78: 578–583
Mueller-Eckhardt C, Kiefel V, Grubert A, Kroll H, Weisheit M, Schmidt S, Muller-Eckhardt G, Santoso S 1989 348 cases of suspected neonatal alloimmune thrombocytopenia. Lancet 1: 636–366
Currie BG, Schell D, Bowring AC 1991 Giant hemangioma of the arm associated with cardiac failure and the Kasabach-Merritt syndrome in a neonate. J Pediatr Surg 26: 734–737
Chan WC, Carroll A, Alvarado CS, Phillips S, Gonzalez-Crussi F, Kurczynski E, Pappo A, Emami A, Bowman P, Head DR 1992 Acute megakaryoblastic leukemia in infants with t(1;22)(p13;q13) abnormality. Am J Clin Pathol 98: 214–221
Homans AC, Cohen JL, Mazur EM 1988 Defective megakaryocytopoiesis in the syndrome of thrombocytopenia with absent radii. Br J Haematol 70: 205–210
Gross SJ, Filston HC, Anderson JC 1982 Controlled study of treatment for disseminated intravascular coagulation in the neonate. J Pediatr 100: 445–448
Tate DY, Carlton GT, Johnson D, Sorenson RL, Nesbit M, White J, Thompson T, Krivit W 1981 Immune thrombocytopenia in severe neonatal infections. J Pediatr 98: 449–453
Castle V, Coates G, Kelton J, Andrew M 1987 111 In-oxine platelet survivals in thrombocytopenic infants. Blood 70: 652–656
Burrows RF, Andrew M 1990 Neonatal thrombocytopenia in the hypertensive disorders of pregnancy. Obstet Gynecol 76: 234–238
Van den Hof MC, Nicolaides KH 1990 Platelet count in normal, small, and anemic fetuses. Am J Obstet Gynecol 162: 735–739
Salvesen DR, Brudenell MJ, Nicolaides KH 1992 Fetal polycythemia and thrombocytopenia in pregnancies complicated by maternal diabetes mellitus. Am J Obstet Gynecol 166: 1287–1292
Saade GR, Moise KJ Jr, Copel JA, Belfort MA, Carpenter RJ Jr 1993 Fetal platelet counts correlate with the severity of anemia in red-cell alloimmunisation. Obstet Gynecol 82: 987–991
Graeve JLA, De Alarcon PA 1989 Megakaryocytopoiesis in the human fetus. Arch Dis Child 64: 481–484
Bodger MP 1987 Isolation of hemopoietic progenitor cells from human umbilical cord blood. Exp Hematol 15: 869–876
Christensen RD 1987 Circulating pluripotent hematopoietic progenitor cells in neonates. J Pediatr 110: 622–625
Clapp DW, Baley JE, Gerson ST 1989 Gestational age-dependent changes in circulating hemopoietic stem cells in newborn infants. J Lab Clin Med 113: 422–427
Murray NA, Roberts IAG 1995 Circulating megakaryocytes and their progenitors (BFU-MK and CFU-MK) in term and pre-term neonates. Br J Haematol 89: 41–46
Briddell RA, Brandt JE, Straneva JE, Srour EF, Hoffman R 1989 Characterization of the human burst-forming unit megakaryocyte. Blood 74: 145–151
Westwood NB, Emmerson AJB 1991 Erythropoietic progenitor cells from premature neonates studied using a validated serum deprived method. Cytotechnology 6: 241–246
Burrows RF, Hunter DJ, Andrew M, Kelton JG 1987 A prospective study investigating the mechanism of thrombocytopenia in preeclampsia. Obstet Gynecol 70: 334–338
Samuels P, Main EK, Tomaski A, Mennuti MT, Gabbe SG, Clines DB 1987 Abnormalities in platelet antiglobulin tests in preeclamptic mothers and their neonates. Am J Obstet Gynecol 157: 109–113
Rote NS, Lau RJ, Harrison MR, Branch DW, Scott JR 1987 Platelet-binding immunoglobulins in pregnancy-induced hypertension. I. J Reprod Immunol 10: 261–272
Rote NS, Harrison MR, Scott JR 1987 Platelet-binding immunoglobulins in pregnancy-induced hypertension. II. J Reprod Immunol 10: 273–284
Rinder HM, Bonan JL, Anandan S, Rinder CS, Rodrigues PA, Smith BR 1994 Noninvasive measurement of platelet kinetics in normal and hypertensive pregnancies. J Obstet Gynecol 170: 117–122
Murray NA, Roberts IAG 1994 Circulating megakaryocyte precursors in neonatal thrombocytopenia. Pediatr Res 36: 31A( abstr)
Koenig JM, Christensen RD 1989 Incidence, neutrophil kinetics, and natural history of neonatal neutropenia associated with maternal hypertension. N Engl J Med 321: 557–562
Roberts IAG, Hindawi S, McMullin MF, Murray NA 1992 Absent circulating CFU-GM in neutropenic premature infants born to hypertensive mothers. Exp Hematol 20: 801
Kurlat I, Sola A 1992 Neonatal polycythemia in appropriately grown infants of hypertensive mothers. Acta Paediatr 81: 662–664
Huang S-C, Chang F-M 1994 The adverse effect on fetal hemogram by preeclampsia: marked anisocytosis with normocytic, normochromic erythrocythemia as well as thrombocytopenia. Early Hum Dev 37: 91–98
Meberg A, Jakobsen E, Halvorsen K 1982 Humoral regulation of erythropoiesis and thrombopoiesis in appropriate and small for gestational age infants. Acta Paediatr Scand 71: 769–773
Koenig JM, Christensen RD 1989 Neutropenia and thrombocytopenia in infants with Rh hemolytic disease. J Pediatr 114: 625–631
Teramo KA, Widness JA, Clemons GK, Voutilainen P, McKinlay S, Schwartz R 1987 Amniotic fluid erythropoietin correlates with umbilical plasma erythropoietin in normal and abnormal pregnancy. Obstet Gynecol 69: 710–715
Salvesen DR, Brudenell JM, Snijders RJM, Ireland RM, Nicolaides KH 1993 Fetal plasma erythropoietin in pregnancies complicated by maternal diabetes mellitus. Am J Obstet Gynecol 168: 88–94
Petursson SR, Chervenick PA 1987 Effects of hypoxia on megakaryocytopoiesis and granulopoiesis. Eur J Haematol 39: 267–273
Rolovic Z, Basara N, Biljanovic-Paunovic L, Stojanovic N, Suvajdzic N, Pavlovic-Kentera V 1990 Megakaryocytopoiesis in experimentally induced chronic normobaric hypoxia. Exp Hematol 18: 190–194
Ruth V, Widness J, Clemons G, Raivio K 1990 Postnatal changes in serum immunoreactive erythropoietin in relation to hypoxia before and after birth. J Pediatr 116: 950–954
Author information
Authors and Affiliations
Additional information
Supported by grants from Action Research.
Rights and permissions
About this article
Cite this article
Murray, N., Roberts, I. Circulating Megakaryocytes and Their Progenitors in Early Thrombocytopenia in Preterm Neonates. Pediatr Res 40, 112–119 (1996). https://doi.org/10.1203/00006450-199607000-00020
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199607000-00020
This article is cited by
-
Thrombocytopenia and insufficient thrombopoietin production in human small-for-gestational-age infants
Pediatric Research (2023)
-
Bleeding in neonates with severe thrombocytopenia: a retrospective cohort study
BMC Pediatrics (2022)
-
The effects of maternal hypertension on the early neonatal platelet count
Journal of Perinatology (2022)
-
Circulating Mature Megakaryocytes in an Unusual Case of BCR-ABL1-Positive CML
Indian Journal of Hematology and Blood Transfusion (2018)
-
Neonatal thrombocytopenia—causes and outcomes following platelet transfusions
European Journal of Pediatrics (2018)