Abstract
Pentoxifylline (PTXF) is a methylxanthine derivative which modifies leukocyte function and inhibits tumor necrosis factor (TNF)-α release. As TNF-α is considered a proximal mediator in the cascade leading to septic shock, we evaluated the ability of PTXF to attenuate the cardiovascular manifestations of sepsis secondary to an infusion of group B β-hemolytic streptococci (GBS). Fifteen anesthetized, mechanically ventilated piglets(weight, 2815 ± 552 g) were randomly assigned to a treatment group which received a continuous infusion of PTXF (5 mg/kg/h) beginning 30 min after GBS (7.5 × 108 colony-forming units/kg/min) administration was started or to a control group which received GBS plus saline as placebo. Comparison of the hemodynamic measurements and arterial blood gases over the first 120 min of bacterial infusion for treatment and control groups revealed the following statistically significant differences (120-min values presented): cardiac output was significantly higher in the PTXF group (0.159± 0.035 versus 0.09 ± 0.026 L/kg/min; p < 0.05) as was stroke volume (0.54 ± 0.11 versus 0.27 ± 0.126 mL/kg/beat; p < 0.01). Pulmonary and systemic vascular resistances remained lower in the PTXF-treated animals (167 ± 45versus 233 ± 69 mm Hg/L/kg/min; p < 0.03) and(427 ± 162 versus 828 ± 426 mm Hg/L/kg/min;p < 0.03, respectively). Median survival time was significantly longer in the PTXF group (180 versus 120 min; p < 0.05). In an additional group of animals, PTXF administration before GBS infusion revealed no attenuation in the rise of TNF-α, accompanying sepsis. These data demonstrate that treatment with PTXF may ameliorate some of the deleterious hemodynamic manifestations of GBS sepsis and result in improved survival in a young animal model without significantly modifying plasma TNF-α levels.
Similar content being viewed by others
Main
GBS sepsis is the major cause of early onset bacterial infection in neonates, occurring soon after delivery and manifested by rapid onset of fulminant shock often accompanied by persistent pulmonary hypertension(1). Initiation of this sequence occurs when bacteria and their products stimulate inflammatory cells which release various mediators and cytokines such as TNF-α and IL-1. In fact, TNF-α is purported to be responsible for the lethal effects of endotoxin and other bacterial products(2, 3). Gibson et al.(3) demonstrated an increase in TNF-α plasma levels in neonatal GBS sepsis after 90 min of bacterial infusion, suggesting that therapeutic strategies directed against TNF-α, a cytokine considered to be the proximal mediator of tissue injury and shock associated with sepsis, are appealing and may be useful in the management of septic shock. Fiedler et al.(4) have demonstrated that administration of a TNF-α MAb to primates 30 min after lipopolysaccharide administration offered protection from shock and provided reduced mortality. Recently, Abraham et al.(5) showed that a MAb to TNF-α could also reduce mortality in humans with septic syndrome.
PTXF, a methylxanthine derivative, may be of potential benefit as a therapeutic agent in sepsis because it decreases the production and effects of TNF-α and IL-1(3, 6–8). Furthermore, PTXF attenuates TNF-α-induced endothelial cell injury in the presence of polymorphonuclear cells(9) and has been shown to inhibit the rise in serum TNF-α levels induced by low dose endotoxin infusion in humans(10). In addition, properties of PTXF such as reduction in blood viscosity(11) and subsequent decrease in vascular resistance and improvement in microvascular flow may be also beneficial in neonatal sepsis.
Previous studies in neonatal animals(3, 12) have shown that PTXF administration before an infusion of GBS resulted in decreased TNF-α plasma levels and attenuation of the cardiovascular manifestation of sepsis such as decreased cardiac output and arterial blood pressure and increased pulmonary vascular resistance. These changes were associated with prolonged survival in neonatal animals receiving PTXF(3, 12).
There is no information evaluating the effects of PTXF on the hemodynamic changes when administered after the onset of GBS sepsis. We hypothesized that PTXF, if infused after a bacterial challenge was initiated, would ameliorate the cardiovascular alterations and improved survival in a lethal model of neonatal GBS sepsis.
METHODS
Fifteen Yorkshire piglets aged 7-10 d were anesthetized with pentobarbital(30 mg/kg, intraperitoneally). A tracheostomy was performed, and a 4.5-mm endotracheal tube was placed. Femoral arteries and veins were cannulated and used for measurement of Psa, blood sampling, and infusion of bacteria, fluids, and drugs. The left external jugular vein was cannulated, and the catheter was advanced into the right atrium for measurement of Pra and injection of ice-cold saline which was used in the measurement of CO. A 5 Fr Swan-Ganz thermodilution catheter was introduced into the right external jugular vein, advanced under fluoroscopy into the left pulmonary artery, and used to measure Ppa and Pwp. Heparinized normal saline was infused continuously through the pulmonary artery catheter, and 6 mL/kg/h 5% dextrose solution was infused through a peripheral vein. Vascular pressures were measured with pressure transducers (model P23-1D; Gould Instruments, Cleveland, OH) and recorded on a multichannel recorder (model 7 polygraph, Grass Instrument, Quincy, MA).
The animals were ventilated with a time-cycled, pressure-limited, infant ventilator (Bourns BP 200, Riverside, CA). Arterial blood gases in the normal range were obtained by setting peak inflation pressure at 11 ± 1 cm H2O, positive end-expiratory pressure at 2 cm H2O, and the respiratory rate at 40 breaths/min. Animals were ventilated with room air, and settings were not altered during the study period. Paralysis was induced with pancuronium bromide using an initial dose of 0.2 mg/kg i.v. followed by a continuous infusion of 0.4 mg/kg/h. Rectal temperature was continuously monitored with a thermistor probe (Yellow Springs Instrument Co., Yellow Springs, OH), and skin temperature was maintained at 38.5 °C by a servo-controlled radiant warmer.
GBS, type Ia/c, isolated from an infected neonate cared for in the neonatal intensive care unit at Jackson Memorial Hospital, were cultured in Todd-Hewitt broth for 18 h at 37 °C. The organisms were collected by centrifugation, washed twice in pyrogen-free saline, and resuspended in sterile Ringer's lactate solution with 5% dextrose at a concentration determined by OD measurements to be equivalent to 8.7 × 109 colony forming units/mL. The bacterial cell suspension was free of endotoxin as determined by a Limulus amebocyte lysate test (Associate of Cape Cod, MA) which had a sensitivity of >0.03 endotoxin unit/mL. The bacterial suspension was then infused through a femoral vein at a rate calculated to deliver approximately 7.5 × 107 colony-forming units/kg/min. This infusion was started after baseline measurements and continued until the animals expired.
Randomization was performed by an individual not involved in the experiment, and the investigators were unaware of group assignment. Animals were randomly assigned to a treatment group (n = 8)([horizontal bar over]X ± SD; weight, 2778 ± 452 g; age, 9 ± 1 d) which received PTXF as a continuous infusion of 5 mg/kg/h throughout the study period. Seven piglets (weight, 2925 ± 690 g; age, 8 ± 1 d) were assigned to the control group and received 10 mL of normal saline over the same time period, followed by a continuous infusion of normal saline at the same rate as the continuous infusion of PTXF.
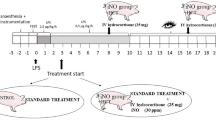
After a 60-min stabilization period, hemodynamic measurements (Psa, Ppa, Ppw, CO, and Pra) and arterial blood gases were obtained before any intervention (baseline). The bacterial suspension was then infused, and 15 min after the onset of pulmonary hypertension, all measurements and arterial blood gases were repeated. Thirty minutes after the onset of pulmonary hypertension the treatment group received a continuous infusion of PTXF (5 mg/kg/h), and the control group received normal saline as a placebo following the same protocol. Hemodynamic measurements and blood gases were obtained 15 min after the beginning of PTXF or placebo infusion and subsequently at 30, 60, and every 30 min until the animals' death. Pulmonary vascular and systemic vascular resistances and stroke volume were calculated.
To ascertain the effect of PTXF on the plasma TNF-α levels before and after sepsis, we measured TNF-α levels in an additional 14 piglets, aged 7-10 d, which were randomly assigned to a control group (n = 7; weight, 3171 ± 515 g) which received normal saline as placebo or a treatment group (n = 7; weight, 3687 ± 577 g) which received PTXF as a 20 mg/kg bolus over 20 min, followed by a continuous infusion of 5 mg/kg/h throughout the study. Thirty minutes after the start of PTXF or placebo, GBS infusion was initiated. TNF-α levels were measured at baseline, before the bacterial infusion (BL 2), and at 60 and 120 min after the bacterial infusion was begun.
Handling and care of animals were in accordance with the guidelines of the National Institutes of Health. This study protocol was approved by the Animal Care Committee of the University of Miami School of Medicine.
Dependent variables in this study included the following: Ppa, Psa, Pwp, stroke volume, CO, pH, the partial pressure of arterial O2 and CO2, base deficit, PVR, and SVR. Significant mortality in the control group prevented us from analyzing data after 120 min. Data are expressed as mean ± SD. Repeated measures analysis of covariance was used to compare the pattern of response to treatment against the control response, both over time and independent of time. The first post-GBS measurement was used as a covariate to adjust for any possible differences between groups before PTXF was administered. Significant mortality in the control group prevented us from analyzing data after 120 min. As a part of the repeated measures analysis of variance, all parameters were tested in the following two ways. 1) The overall treatment effect was evaluated to determine whether from 45 to 120 min the PTXF animals were different from control animals in a consistent fashion independent of time and 2) the time-treatment interaction was derived to evaluate if the PTXF and control group differences depended on time. When a time-treatment interaction is present, the overall treatment significance test is no longer relevant. In plots of the data in this study a consistent overall treatment effect is indicated by a vertical bar, whereas a time-treatment interaction is indicated by a horizontal bar(13). The values are Huynh-Feldt corrected for correlations between the same animals at different times. Survival time was compared between groups using the Mann-Whitney U test as all animals were followed until death.
RESULTS
Age and weight were not significantly different between treatment and control animals. Hemodynamic measurements at baseline and 15 min after the bacterial infusion was begun were also comparable.
Cardiac output initially decreased similarly in both groups after GBS administration, but was significantly higher (time-treatment interaction,p < 0.05) over time in the PTXF-treated animals (Fig. 1). The net result was a 30% decline in CO between baseline and 120 min after the bacterial infusion was begun in PTXF-treated animals, compared with a 62% decrease from baseline in control animals at 120 min. Heart rate was not significantly different during the bacterial infusion, however, stroke volume, although decreased after the bacterial infusion was begun, remained higher in the PTXF-treated group compared with control animals throughout the study period (time-treatment interaction, p < 0.01) (Fig. 1).
Systemic blood and right atrial and pulmonary wedge pressures were not significantly different between groups (Table 1). However, there was a trend toward significance (p = 0.078) in the mean Ppa (Table 1).
Although PVR increased in both groups after the bacteria were begun, it remained lower in the PTXF group than in control animals (treatment effect,p < 0.03) (Fig. 2). Systemic vascular resistance was also lower in the PTXF-treated than in the control group(time-treatment interaction effect, p < 0.03) (Fig. 2).
Arterial oxygen tension and partial pressure of arterial CO2 were not significantly different. Although pH and base deficit revealed less acidosis in PTXF-treated animals, the differences were not statistically significant (Table 2). The PTXF-treated animals survived significantly longer than control animals [median (range); 180 (180-300)versus 120 (90-180) min, respectively (p < 0.05)].
Pretreatment with PTXF before initiation of the GBS infusion resulted in the following control versus PTXF plasma TNF-α levels:([horizontal bar over]X ± SD); baseline, 428 ± 111versus 411 ± 171 pg/mL; BL 2 (post-PTXF bolus infusion), 391± 111 versus 347 ± 69 pg/mL; and TNF-α levels 60 and 120 min after the bacterial infusion was begun were 559 ± 216versus 454 ± 129 and 777 ± 473 versus 838± 310 pg/mL, respectively. Although TNF-α plasma levels after GBS were significantly higher (p < 0.05) compared with baseline, the groups were not significantly different over the study period.
DISCUSSION
The results of the present study demonstrate that treatment with PTXF can ameliorate the early deleterious hemodynamic effects of GBS sepsis in a young animal model. Specifically, our data indicate that treatment with PTXF modifies the cardiovascular compromise associated with sepsis such as diminished cardiac output and increased pulmonary and systemic vascular resistances. The result of those beneficial effects is an increase in survival in this highly lethal model of neonatal sepsis.
Previously, we have shown that PTXF ameliorated the decrease in Psa, CO, and PVR, and improved survival in the same model of GBS sepsis when animals were pretreated with PTXF(9). Gibson et al.(3) also demonstrated that PTXF treatment before initiation of a continuous infusion of GBS resulted in mild improvement in Ppa, PVR, and partial pressure of arterial O2. These investigators additionally noted that animals receiving GBS plus PTXF had significantly greater systemic hypotension after 4 h when compared with those receiving GBS alone. The methods and results of the present study differ from the aforementioned works, in that Gibson et al.(3) used a bolus of 20 mg/kg and a continuous infusion of 20 mg/kg/h, whereas del Moral et al.(12) in their pretreatment model used a 20 mg/kg bolus plus a continuous infusion of 5 mg/kg/h. The hypotension associated with PTXF and GBS in the study by Gibson et al. was most likely secondary to the high dose of PTXF used and was absent in the work performed by del Moral et al.(12). The latter study demonstrated that a continuous infusion, using a lower dose of PTXF, could result in a higher Psa than that of control animals despite the use of a more lethal GBS model. In the present work we chose to use a continuous infusion of 5 mg/kg/h without using a loading dose. This decision was based in part on a desire to more closely mimic doses used in adult human clinical trials(14). In addition, previous work showed that a bolus resulted in a 50% increase in serum levels of PTXF above the steady state, and we anticipated that this might result in deleterious effects on Psa in an already compromised septic host. Furthermore, the model used is a highly lethal one in contrast to that used by Gibson et al. and in that respect is similar to the rapid progression and severe hemodynamic instability seen in the severely ill septic neonate. The nature of our model and dose of PTXF used consequently allowed detection of an overall beneficial effect of PTXF on CO, SV, PVR, and length of survival.
In the present study, CO decreased approximately 30% in the PTXF-treated animals versus 62% in control animals. This may have been partially related to increased SVR secondary to improvement in myocardial contractility. PTXF has been shown to increase contractility in isolated dog atria(15), possibly as a result of increased production of cAMP secondary to PTXF-induced adrenergic stimulation(6). PTXF also has been shown to decrease production of TNF-α(3), a cytokine, which has been associated with depression of CO during sepsis(16, 17). This, however, does not appear to be the case in the present study. In addition, properties of PTXF such as reduction in blood viscosity and vascular resistance may largely increase CO by decreasing afterload rather than by affecting myocardial contractility.
Administration of PTXF in this study was associated with a reduction in PVR, which may be secondary to PTXF-induced increase in erythrocyte deformability with resulting improvement in microvascular blood flow and decreased resistance(18). Hakim and Petrella(19) have shown that PTXF has a marked vasodilatory effect in hypoxic pulmonary hypertension, which may be independent of its rheologic properties(20). Whether this effect is direct or via production or liberation of mediators is not clear. PTXF administration has also been reported to result in enhanced vascular wall production of prostacyclin, which may cause systemic and pulmonary vasodilatation(11). This point is supported by the work of Gibson et al.(21), who noted that the systemic hypotensive effects of high dose PTXF may be ameliorated by cyclooxygenase blockade. However, according to other works using this model, the changes in PVR and Ppa occur before significant increases in prostaglandin I2 and in the presence of elevated thromboxane A2, suggesting that another mechanism may be operative(3, 22).
Another significant finding in this work, not noted in similar models, was the improvement in length of survival in PTXF-treated animals. This finding has been reported in adult animal models of peritonitis and endotoxic shock(23, 24) and previous work in newborn animals(12). However, because the therapeutic range of PTXF is narrow, caution is advised. Our previous dose-response relationships suggest that high dose PTXF is associated with hypotension and early deterioration compared with controls(12).
The reason for the improvement in survival is not entirely clear. PTXF has been shown to decrease TNF-α-related systemic hypotension(7) and TNF-α-induced lung injury(8). However, the results presented in this work suggest that PTXF does not suppress the increase in TNF-α after bacterial infusion when a loading dose of 20 mg/kg plus a continuous infusion of 5 mg/kg/h is used. The dose of PTXF needed to measurably suppress serum TNF-α could result in untoward hemodynamic consequences(3, 12). Consequently, TNF-α was most likely not suppressed in the present study by the continuous infusion of 5 mg/kg/h. We speculate that amelioration of the hemodynamics associated with sepsis in the present work may in part be explained by vasodilatation.
In our desire to avoid PTXF-induced hypotension we may have prejudiced the potential of PTXF to ameliorate sepsis-induced sequelae. The reported protocol may have resulted in less effective PTXF serum levels and consequently only minimally attenuated TNF-α production. However, higher doses used in the pretreatment portion of this study failed to modify TNF-α levels. Furthermore, as mentioned previously, the aim of this study was to mimic the actual therapeutic dosage range now being tested in human clinical trials and to avoid further compromising the septic, potentially hypotensive subject.
In summary, our work demonstrates that PTXF used after GBS infusion was begun was able to ameliorate the decline in CO, stroke volume, and the increase in pulmonary and systemic vascular resistances. It also prolonged the survival in a highly lethal model of group B streptococcal infusion in piglets, suggesting that PTXF treatment may be beneficial in neonatal sepsis. However, caution is advised as the dosage regimen needed to consistently obtain the beneficial effects may result in significant hypotension.
Abbreviations
- CO:
-
cardiac output
- GBS:
-
group B streptococci
- PTXF:
-
pentoxifylline
- PVR:
-
pulmonary vascular resistance
- Ppa:
-
pulmonary artery pressure
- Pwp:
-
pulmonary wedge pressure
- Pra:
-
right atrial pressure
- SVR:
-
systemic vascular resistance
- Psa:
-
systemic arterial blood pressure
- TNF-α:
-
tumor necrosis factor-α
REFERENCES
Meadow W, Rudinsky B 1995 Inflammatory mediators and neonatal sepsis. Clin Perinatol 22: 519–536
Tracey KJ, Lowry SF, Cerami A 1988 Cachectin/TNF in septic shock and septic adult respiratory distress syndrome. Am Rev Respir Dis 138: 1377–1379
Gibson RL, Redding GJ, Henderson WR, Truog WE 1991 Effect of the tumor necrosis factor inhibitor pentoxifylline on hemodynamics and gas exchange. Am Rev Respir Dis 143: 598–604
Fiedler VB, Loof I, Sander E, Voehringer V, Galanos C, Fournel M 1992 monoclonal antibody to tumor necrosis factor-α prevents lethal endotoxin sepsis in adult rhesus monkeys. J Lab Clin Med 120: 574–588
Abraham E, Wunderik R, Silverman H, Perl TM, Nasraway S, Levy H, Bone R, Wenzel R, Balk R, Alfred R, Pennington JS, Wherry JC 1995 Efficacy and safety of monoclonal antibody to human tumor necrosis factorα in patients with sepsis syndrome. JAMA 273: 934–941
Noel P, Nelson S, Bokulic R, Bagby G, Lippton H, Lipcono G, Summer W 1989 Pentoxifylline inhibits lipopolysaccharide-induced serum tumor necrosis factor and mortality. Clin Res 37: 31A
Lilly CM, Sandhu JS, Ishizaka A, Harada H, Yonemaru M, Larrick JW, Shi T, O'Hanley TF, Raffin TA 1989 PTXF prevents tumor necrosis factor-induced lung injury. Am Rev Respir Dis 139: 1361–1368
Sullivan GW, Carper TH, Novick W, Mandell GL 1988 Inhibition of the inflammatory action of interleukin-1 and tumor necrosis factor (alpha) on neutrophil function by pentoxifylline. Infect Immun 56: 1722–1729
Zheng H, Crowley J, Chan JC, Hoffman H, Hatherill JR, Ishizaka A, Raffin TA 1990 Attenuation of TNF-induced endothelial cells cytotoxicity and neutrophil chemiluminescence. Am Rev Respir Dis 142: 1043–1078
Zabel P, Wolter DT, Schonhartin M, Schade UF 1989 Pentoxifylline suppresses endotoxin-induced TNF formation in men. In: On Pentoxifylline and Analogues Effects on Leukocyte Function. Saint Paul-de-Vence, France, p 50 (abstr)
Schroer RH 1985 Antithrombotic potential of pentoxifylline: a hemortheologically active drug. Angiology 36: 387–389
del Moral T, Goldberg R, Suguihara C, Martinez O, Feuer WJ, Bancalari E 1992 Effects of pentoxifylline on the cardiovascular manifestations of group B streptococcal sepsis in the piglet. Pediatr Res 31: 596–600
Fleiss JL 1986 The Design and Analysis of Clinical Experiments. Wiley, New York, p 188
Montravers P, Fagon JY, Gibert C, Blanchet F, Novara A, Chastre J 1993 Pilot study of cardiopulmonary risk from pentoxifylline in adult respiratory distress syndrome. Chest 103: 1017–1022
Watanabe H, Furukana, Schira 1982 Cardiovascular effects of aminophylline and pentoxifylline on intact dogs and isolated dog atria. Jpn Heart J 23: 235–243
Osiovich H, Goldberg R, Adams JA, Offenbacher S, Kuo G, Devia C, Suguihara C, Bancalari E 1989 Cardiovascular effects of TNF (r-TNF) in piglets. Pediatr Res 25: 279A( abstr)
Truog WE, Gibson RL, Henderson WR, Redding GJ 1990 Tumor necrosis factor induced neonatal pulmonary hypertension: effects of dazmegrel pretreatment. Pediatr Res 27: 466–471
Chick T, Scotto P, Icenoglem Sikes W, Doyle P, Riedel C, Wood S, Loeppky JA 1988 Effects on pentoxifylline on pulmonary hemodynamics during acute hypoxia in anesthetized dogs. Am Rev Respir Dis 137: 1099–1103
Hakim TS, Petrella J 1988 Attenuation of pulmonary and systemic vasoconstriction with pentoxifylline and aminophylline. Can J Physiol Pharmacol 66: 396–401
Hakim TS 1988 Reversal of pulmonary hypoxic vasoconstriction with pentoxifylline and aminophylline in isolated lungs. Can J Physiol Pharmacol 66: 146–151
Gibson RL, Truog WE, Henderson WR Jr, Redding GJ 1992 Group B streptococcal sepsis in piglets: effect of combined pentoxifylline and indomethacin pretreatment. Pediatr Res 31: 222–227
Runkle B, Goldberg RN, Streitfeld MM, Clark Mb, Buron E, Setzer ES, Bancalari E 1984 Cardiovascular changes in group B streptococcal sepsis in the piglet: response to indomethacin and the relationship to prostacyclin and thromboxane A2 . Pediatr Res 18: 874–878
Bjornson S, Cave C, Bjornson A 1987 Effects of pentoxifylline on survival and chemotaxis of polymorphonuclear leukocytes in a rat model of bacterial peritonitis. In: Mandell GL, Novick WJ (eds) Pentoxifylline and Leukocyte Function. Hoechst-Roussel Pharmaceuticals, Somerville, NJ, pp 138–154
Schonharting MM, Schade UF 1989 The effect of pentoxifylline in septic shock: new pharmacological aspects of an established drug. J Med 20: 97–105
Acknowledgements
We are grateful to Dr. W. J. Novick and to Hoechst-Roussel Pharmaceuticals, Inc., for generously supplying pentoxifylline.
Author information
Authors and Affiliations
Additional information
Supported by the University of Miami Project: New Born.
Presented in part at the Society for Pediatric Research, Baltimore, May 1992.
Rights and permissions
About this article
Cite this article
Moral, T., Goldberg, R., Urbon, J. et al. Effects of Treatment with Pentoxifylline on the Cardiovascular Manifestations of Group B Streptococcal Sepsis in the Piglet. Pediatr Res 40, 469–474 (1996). https://doi.org/10.1203/00006450-199609000-00017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199609000-00017