Abstract
Upon reperfusion of ischemic tissues, reactive oxygen metabolites are generated and are responsible for much of the organ damage. Experimental studies have revealed two main sources of these metabolites: 1) the oxidation of hypoxanthine to xanthine and on to uric acid by the oxidase form of xanthine oxidoreductase and 2) neutrophils accumulating in ischemic and reperfused tissue. Blocking either source will reduce reperfusion damage in a number of experimental situations. Although xanthine oxidoreductase activity may be unmeasurably low in organs other than liver and intestine, it may be involved in reperfusion injury elsewhere because of its localization in capillary endothelial cells. Time course considerations suggest that substrate accumulation and NADH inhibition of dehydrogenase activity may be more important in the pathogenesis than conversion of xanthine dehydrogenase into the oxidase form. Neutrophil accumulation may be partly due to oxidants in the first place, suggesting a link between the two sources of reactive oxygen metabolites. In the clinical context, many of the sequelae of perinatal asphyxia may be accounted for by reperfusion damage to organs such as brain, kidney, heart, liver, and lungs. During asphyxia, substrates of xanthine oxidase accumulate, upon resuscitation the cosubstrate oxygen is introduced, and evidence for oxidant production and effects has been obtained. In the pathogenesis of brain damage after asphyxia, both microvascular injury and parenchymal cell damage are important. Oxygen metabolites are involved in the former, but in the latter process their role is less clear because ischemia-reperfusion triggers not only oxidant production but many other phenomena, including gene activation, ATP depletion, glutamate accumulation, and increase of intracellular calcium. A severe insult results in cell necrosis, but more moderate asphyxia may cause delayed neuronal death through apoptosis. The time course of the changes in high energy phosphates as well as of selective neuronal death suggest that in the first hours of life there is a“therapeutic window,” with future possibilities for prevention of permanent damage.
Similar content being viewed by others
Main
Severe and prolonged ischemia or hypoxia of any organ will result in cell death and tissue damage. Thus, restoration of blood flow or oxygen delivery is necessary for organ survival, but it is, on the other hand, critical because damage may be amplified during this period. For example, if the superior mesenteric artery of a cat is clamped for 3 h and then reperfused for 1 h, mucosal damage is more severe than after 4 h of continuous ischemia(1). Reperfusion with deoxygenated blood causes no further damage beyond that observed after 3 h of ischemia(1), suggesting that oxygen and its reactive metabolites are implicated in the pathogenesis. A number of other studies have provided evidence for the basic concept and for the role of oxygen metabolites(2, 3).
Perinatal asphyxia, with its attendant bradycardia and hypotension, is an ischemic condition, which is followed by reperfusion upon resuscitation. After asphyxia, dysfunction of several organs is common, and permanent damage, particularly of the brain, is possible. In this review, we will consider the potential sources of reactive oxygen metabolites in reperfusion injury and their relevance to postasphyxial organ damage, with emphasis on the brain. We will also review the evidence on circulatory changes associated with experimental and clinical asphyxia and the mechanisms of hypoxic-ischemic cell damage, especially the role of oxidants.
GENERATION OF REACTIVE OXYGEN METABOLITES UPON REPERFUSION
Of the main reactive metabolites of oxygen relevant to human pathology, O2[horizontal line over dot] and H2O2 can be formed through several metabolic pathways [reviewed in Kinnula(4)]. They in turn can produce the most reactive of the oxygen metabolites, OH˙, through the Haber-Weiss or Fenton reactions catalyzed by Fe2+(5). In the context of reperfusion injury, the two important sources of reactive oxygen metabolites are XOR(3, 6) and inflammatory cells(7), but other pathways may be significant, depending on the species and cell type(8–11).
XOR. XOR catalyzes the last two steps of purine catabolism in man, oxidation of hypoxanthine to xanthine and on to uric acid(12, 13). Under physiologic conditions, the enzyme exists mainly as a dehydrogenase (XORD), uses NAD+ as the electron acceptor, and generates no harmful metabolites. In ischemic tissue XORD can be converted into an oxidase (XORO), which utilizes molecular oxygen as the electron acceptor and produces O2[horizontal line over dot] and H2O2(14). Also during ischemia, ATP is catabolized and hypoxanthine accumulates(3, 15–17). During reperfusion the cosubstrate O2 is reintroduced, and the stage is set for production of reactive oxygen metabolites (Fig. 1). The other product of XOR, uric acid, is a free radical scavenger and may function as an extracellular antioxidant in man(18). Whether it has any significance in reperfusion injury is unclear.
Evidence for the role of XOR in reperfusion injury is mainly indirect, based on prevention of organ damage in animal models with XOR inhibitors(allopurinol or oxipurinol). Hundreds of such studies have been reported, relevant to reperfusion damage of the heart [reviewed in Godin and Ko(19)], intestine [reviewed in Granger(6)], kidney(20), liver [reviewed in Lemasters and Thurman(21)], lung(22), skeletal muscle(23), and brain [reviewed in Palmer(24)]. However, amelioration of reperfusion injury by allopurinol has not been consistent, and mechanisms unrelated to XOR inhibition may be at play, e.g. increased salvage of purine bases to maintain the ATP pool(25) or direct scavenging of free radicals by allopurinol(26). Therefore, the argument for the role of XOR is strengthened by another approach, namely pretreatment of animals with tungsten, which inactivates XOR by replacing molybdenum in the active site of the enzyme(27). Such animals are protected against reperfusion damage to the heart(28), intestine(29), kidney(30), and lung(22).
Using both direct and spin-trapping techniques of electron paramagnetic resonance spectroscopy, a burst of O2[horizontal line over dot] generation has been shown in the first minutes after reperfusion of ischemic rat heart(31) and reoxygenation of anoxic bovine(32) and human(33) endothelial cells. In each case, allopurinol or oxipurinol prevented the production of superoxide at concentrations that are too low to scavenge free radicals.
In normal adults, XOR activity is found mainly in liver and intestine, with lower levels in kidney and undetectable activities in other organs(12, 34, 35). Our findings of immunoreactive XOR in fetal and neonatal organs(36) are in accordance with the adult data. XOR activity decreases in human intestine but increases in liver during fetal development(37). Immunolocalization studies have shown that in bovine organs XOR is mainly present in capillary endothelial cells(38), and similar findings have been reported for human tissues(39, 40). Activity measurements in cells isolated from rat(41) and bovine(42) brain capillaries as well as from cultured microvascular endothelium(43) have supported this localization. Furthermore, endothelial XOR activity can be induced by γ-interferon(44) and by hypoxia(45–47). Thus, even if in whole tissue homogenates enzyme activity is low, XOR may be relevant, because capillary damage and increased permeability are important in the pathogenesis of reperfusion injury.
The conversion of XORD to XORO is initially reversible and occurs through sulfhydryl group oxidation, but then becomes irreversible, presumably through proteolysis(13, 14, 48). The hypothesis that XOR is the prime source of reactive oxygen metabolites after reperfusion was originally based on the notion that in ischemic organs the conversion of XORD to XORO occurs within minutes(2). In subsequent studies, the time scale has turned out to be hours rather than minutes both in ischemic rat liver and intestine(49, 50). In the liver it has also been shown that the rates of conversion depend on the nutritional status(51) and differ greatly between hepatocytes, Kupffer cells, and endothelial cells(52). In any case, the rate of conversion appears too slow to account for the burst of radical production and the rapid onset of postischemic damage. However, a minimum of 10-15% of total XOR activity is always in the O-form even in well oxygenated tissues. This is sufficient for rapid generation of oxidants, if the accumulated hypoxanthine and xanthine are metabolized through the oxidase and not the dehydrogenase reaction. This is very likely to happen in ischemic cells, which have elevated levels of NADH, a strong inhibitor of XORD but not of XORO(53).
Inflammatory cells. Ischemia and reperfusion induce an inflammatory response in many tissues, including the brain(54). The expression of a number of cytokines is elevated, including IL-1, IL-6, transforming growth factor-β, and fibroblast growth factor. Some of these changes are rapid, e.g. the mRNA for IL-1β is increased within 15 min of forebrain ischemia in the rat(55). The primary cellular origin of the cytokine response appears to be activated microglia(56–58). The pathogenic role of IL-1 is suggested by the beneficial effects of IL-1 antibody(59), as well as IL-1 receptor antagonist administration(60) or gene transfer(61) on postischemic brain damage. On the other hand, TNF appears to be neuroprotective, because focal ischemic damage was exacerbated in mice lacking TNF receptors(62).
Cytokines presumably mediate the influx of inflammatory cells into reperfused tissue (Fig. 2). Through increased expression of P- and E-selectins and ICAM-1 on endothelial cells(63, 64), and of integrins on leukocytes(65), neutrophils adhere to the endothelium, extravasate through the vessel wall, and accumulate in the interstitium(6, 24, 66, 67). The neutrophils are activated by the inflammatory mediators (cytokines, platelet-activating factor) released during ischemia, and they produce primarily O2[horizontal line over dot], secondarily other oxygen metabolites, and hypochlorous acid. That neutrophils are involved in reperfusion damage is suggested by indirect evidence, based on attenuation by granulocyte depletion of cardiac(68) and intestinal(69) injury. Similar protection is obtained by blocking neutrophil adhesion to endothelium with antibodies to either neutrophil(69–71) or endothelial cell(72) adhesion molecules. Transgenic mice deficient in ICAM-1 have smaller infarct volumes as well as better survival and neurologic function than do wild type animals after transient cerebral ischemia and reperfusion(73).
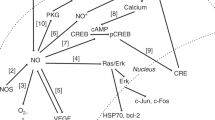
Reperfusion injury to the brain. Ischemia and reperfusion induce both rapid and delayed changes in gene expression. Reactive oxygen metabolites are generated in endothelial and parenchymal cells. They damage cells and promote the expression of adhesion molecules on endothelial cells. Granulocytes accumulate, adhere to and injure endothelial cells, and invade into the parenchyma. Free radicals together with glutamate-induced excitotocity and cytokines from activated microglia cause neuronal damage (see text). Abbreviations: NGF, nerve growth factor; BDNF, brain-derived neurotrophic factor; TGF, transforming growth factor;PAF, platelet-activating factor; ONOO-, peroxynitrite.
Neutrophil accumulation in ischemic tissues can be prevented by allopurinol(74). Thus it has been hypothesized that XORO-derived reactive oxygen metabolites are important for the recruitment of neutrophils(6). A potential mechanism for this is up-regulation of adhesion molecules on endothelial cells(75, 76).
CEREBRAL ISCHEMIA-REPERFUSION IN PERINATAL ASPHYXIA
Animal studies have characterized the hemodynamic changes triggered by asphyxia, i.e. bradycardia, hypotension, and redistribution of blood flow, and documented the changes in cerebral circulation after resuscitation. Also the mechanisms of brain damage after asphyxia, i.e. hypoxic-ischemic encephalopathy, have been studied in experimental animals as well as cultured cells of neural origin. Many of the animal models are unsatisfactory, either because they employ adult rather than immature animals(77), or because they involve ligation of vessels, which results in pathophysiology equivalent to stroke and different from that after global ischemia(78).
In animal models, resuscitation after mild to moderate ischemia is accompanied by an initial period of hyperemia, but a severe insult is followed by hypoperfusion (no reflow phenomenon)(24, 79, 80). The latter has been ascribed to endothelial injury and swelling(81, 82), to granulocyte plugging of microvessels(73, 83, 84), or to intravascular clotting(85). The role of granulocytes is supported by the finding of markedly higher cerebral blood flow after reperfusion in transgenic mice deficient in ICAM-1(86). The microvascular injury is associated with disruption of the blood-brain barrier, secondary brain edema, and possibly secondary neuronal loss(24).
When cerebral blood flow is compromised, brain cells are deprived of oxygen and glucose, resulting in a decrease in high energy phosphate (ATP) levels. Ionic gradients across the cell membrane cannot be maintained, partly due to ATP depletion and loss of Na+/K+-ATPase activity. Influx of Na+ and Ca2+ into the cell is accompanied by water, resulting in cytotoxic edema(78, 87). These changes may become irreversible and lead to cell necrosis and brain infarction as a result of ischemia alone. Reperfusion may temporarily correct the energy failure, but it may trigger a chain of events leading to delayed neuronal death(78, 88) or secondary damage due to brain swelling.
In a newborn piglet model of hypoxic-ischemic encephalopathy, temporary occlusion of the carotid arteries and hypoxic ventilation resulted in“primary” energy failure, documented in MRS as a rapid decrease in the ratios of PCr to Pi and of nucleoside triphosphates to the exchangeable phosphate pool. These changes were equally rapidly normalized upon resuscitation(89). Within several hours, the piglets then developed “secondary” energy failure, as shown by a progressive decrease in high energy phosphate levels. The outcome, in terms of brain damage or death, was correlated with the severity of the primary energy failure. The secondary failure was not associated with a decrease in intracellular pH nor recurring hypoxia or ischemia. Its delayed onset and gradual progression suggest a process triggered by the primary insult, possibly related to excitotoxicity, reactive oxygen metabolites, or cytokine release(89).
The role of reperfusion in the alterations of energy metabolism in the brain after asphyxia is not clear. Both spin-trapping(90, 91) and direct(92) electron paramagnetic resonance measurements have indicated that reactive oxygen metabolites are formed in the first minutes of reperfusion after cerebral ischemia. Some of these metabolites may arise in brain parenchyma, because increased amounts are recovered by microdialysis from the striatum(90). However, the main site of their generation appears to be the endothelial cell (Fig. 2). Reduction of their formation by oxipurinol implicates XORO as one source(91), and a similar effect of indomethacin suggests prostanoid synthesis as another(9). Free radicals are probably the major cause of endothelial damage, increased permeability, and brain edema after asphyxia. This conclusion is supported by the finding of reduced infarct volume and neurologic deficits after reperfusion in transgenic mice overexpressing human SOD(93).
In human infants, the transition from fetal to newborn life is normally associated with an immediate decrease in cerebral blood flow velocity, followed by an increase above the fetal level by the age of 24 h, as indicated by the Doppler flow resistance index(94). In asphyxiated infants, the changes in cerebral blood flow have not been clearly documented, and it is unknown whether a “no reflow phenomenon” exists. This is because of methodologic and ethical constraints in the acute stage immediately after birth, and because global cerebral blood flow measurements do not reveal possible flow changes in critical regions.
In infants developing brain injury after severe asphyxia, normal resistance indices in major arteries were generally found in Doppler studies during the first hours of life(95). Later on, usually on the 2nd d of life, a decrease in the resistance index, i.e. an increase in diastolic flow, was observed(96). Increased global cerebral blood flow, in combination with decreased cross-brain oxygen extraction, was shown on the 1st d after severe asphyxia using the Kety-Schmidt method(97). This is in agreement with the finding of increased cerebral blood volume at an equivalent age using near-infrared spectroscopy(98). A 4-h recording in asphyxiated infants using the latter method showed a decrease in cerebral blood volume between 2 and 12 h of age, which was interpreted as hypoperfusion(99). However, this decrement could represent normalization of hyperperfusion, because in another study, using a xenon-133 technique, hyperemia with abolished autoregulation was observed at the same ages(100). Nevertheless, toward the end of the 1st wk of life, positron emission tomography has revealed hypoperfusion in the parasagittal areas(101), and preliminary studies using single-photon emission tomography suggest similar findings(102). Microvascular abnormalities have not been documented in human infants, but cerebral edema is a well known complication of asphyxia.
In newborn infants studied using MRS after resuscitation from asphyxia at the age of 2-4 h(103), the cerebral energy status was usually normal, as judged by the PCr/Pi ratio or the ratio of ATP to total phosphate. In babies with a good outcome, the PCr/Pi remained in the normal range, whereas in babies subsequently developing brain damage the ratio began to decrease after a delay of 12-24 h(104). A subnormal ATP/Pi ratio predicted fatal outcome(104). The similarity of these findings with those obtained with the piglet model(89) suggests that a primary energy failure occurs in the acute stage also in the human infant, followed by rapid normalization upon resuscitation.
HYPOXIC-ISCHEMIC CELL DAMAGE
Severe asphyxia results in typical neuropathologic changes, which correlate with neurologic abnormalities upon follow-up(105). The localization and extent of the pathology depend not only on the extent and duration of the insult but also on the maturity of the brain. In term infants the parasagittal parietal cortex and hippocampus are the most vulnerable, in preterm infants the periventricular white matter. Early MRI studies of term asphyxiated infants have shown specific patterns of brain damage localized to areas undergoing active myelinization(106, 107). Cell damage may occur both during the ischemic phase and upon reperfusion, probably by different mechanisms.
One mechanism of neuronal death after hypoxia-ischemia appears to be excitotoxicity, triggered mainly by elevation of extracellular glutamate concentrations. This is followed by influx of Ca2+ and Na+ through channels associated with NMDA- and other glutamate-activated receptors(108–110). Subsequent intracellular events are poorly characterized but may include activation of lipases, proteases, endonucleases, and nitric oxide synthase(108). Reactive oxygen metabolites may be directly involved in glutamate release(111) but, more importantly, they may participate in the excitotoxic process itself. O2[horizontal line over dot] production has been demonstrated after exposure of cultured cerebellar granule cells to NMDA(112). SOD was neuroprotective both in an in vivo model of transient ischemic injury(113) and in cultured neural cells subjected to hypoxia and reoxygenation, under depolarizing conditions to allow enzyme entry(114). Overexpression of human SOD ameliorated the neurologic sequelae of transient focal ischemia in transgenic mice(93), but this may partly be accounted for by microvascular effects. However, inasmuch as ischemia alone seems sufficient to induce glutamate neurotoxicity, the role of reperfusion in this context is unclear.
Increase of intracellular Ca2+ levels activates nitric oxide synthase, and inhibitors of this enzyme protect cultured cortical neurons against NMDA-induced damage(115). The neurotoxicity of NO may partly be ascribed to peroxynitrite(116), which is formed in a reaction of NO with O2[horizontal line over dot](117).
Not only the extent but also the mechanism of postischemic neuronal damage may depend on the intensity of the insult. Although exposure of cultured cortical cells to high concentrations of NMDA or peroxynitrite results in necrosis, less intense exposure triggers delayed neuronal death through apoptosis(118, 119). This is characterized by cell shrinkage, membrane blebbing, chromatin condensation, and DNA fragmentation due to endonuclease activation, presumably by elevated Ca2+(120). The typical “ladder” in DNA electrophoresis has been demonstrated in models of focal cerebral ischemia(120–123). In rats, after transient occlusion of the middle cerebral artery followed by reperfusion, the number of apoptotic cells in striatum and cortex was directly proportional to the duration of ischemia(124). In newborn piglets, the severity of the initial ischemic insult, as judged by high energy phosphate depletion in MRS, was correlated with the proportion of apoptotic cells in the cingulate sulcus studied at autopsy after 48 h(125). Glutamate-induced death of cultured cortical neurons was largely prevented by cycloheximide, indicating the dependence of programmed cell death on protein synthesis in this model(126). Partial prevention of apoptosis by inhibition of RNA synthesis with actinomycin D, as well as by inhibition of endonuclease with aurintricarboxylic acid, was shown in cultured sympathetic neurons exposed to hypoxia for 24 h and then reoxygenated for 2 h(127). Increased expression of bcl-2, a known inhibitor of apoptosis and oxidant stress(128), also partially prevented glutamate-induced death of cultured neurons(129, 130) and ameliorated tissue damage after focal cerebral ischemia in rats(131).
Ischemia and reperfusion induce both rapid and delayed changes in gene expression (Fig. 2). The transcription of several members of the c-fos and c-jun proto-oncogene families is increased in postischemic brain(131), heart(132), and kidney(133). The earliest event may be phosphorylation of the transcription factors c-jun and ATF-2 by stress-activated protein kinases(134), detectable after 5 min of reperfusion of ischemic kidney. This is followed by altered binding of these and other transcription factors, including AP-1, to specific promoters. A similar chain of events occurs in cultured renal epithelial cells, when ATP levels are repleted after chemical anoxia(134, 135). The significance of the resulting pattern of gene activation is poorly understood and does not necessarily result in expression of the corresponding protein. However, one end result appears to be programmed cell death, for which induction of c-jun has been considered necessary(136). Other induced proteins such as vimentin(133) and nerve growth factors(137) may play a role in tissue regeneration and repair after ischemia.
CONCLUDING REMARKS
Out of the complex phenomena that are triggered on the cellular level by perinatal asphyxia, some general principles concerning reperfusion injury are beginning to emerge. Early on, a burst of reactive oxygen metabolites is generated, mainly by the activity of xanthine oxidase in endothelial and, depending on the tissue, in parenchymal cells. These metabolites may directly damage cells, but they also promote the expression of adhesion molecules on endothelial cells. This leads to accumulation of granulocytes in the reperfused tissue, further circulatory disturbances, and amplified cell damage by proteases and oxidants. In the brain, and probably in other tissues as well, reperfusion after severe asphyxia may result in early necrotic cell death. Even if the energy status is initially corrected, a progressive secondary energy failure may develop, leading to neuronal loss through apoptosis.
Animal studies have indicated that after short-term intestinal or renal ischemia, reperfusion is the main mechanism of injury, but direct ischemic organ damage becomes more important as a function of ischemia time(138). Before the dominance of the latter process, there is a “therapeutic window” for potential intervention(138). A similar window of opportunity is suggested by the time course of changes in high energy phosphate levels in piglet(89) and human infant(104) brain after an asphyxial insult. Several potential approaches to intervention can be envisaged based on increasing information on the pathogenic mechanisms of reperfusion injury. Prevention of reactive oxygen metabolite formation, strengthening of cellular antioxidant defenses, amelioration of microvascular injury, blocking of Ca2+ entry into cells, and arrest of the process of apoptosis have all been tested in vitro and in animal models, but still await confirmation and clinical trials in asphyxiated human infants.
Abbreviations
- O2[horizontal line over dot]:
-
superoxide
- OH˙:
-
hydroxyl radical
- XOR:
-
xanthine oxidoreductase
- XORD:
-
xanthine dehydrogenase
- XORO:
-
xanthine oxidase
- TNF:
-
tumor necrosis factor
- ICAM-1:
-
intercellular adhesion molecule-1
- MRS:
-
magnetic resonance spectroscopy
- PCr:
-
phosphocreatine
- Pi:
-
inorganic orthophosphate
- SOD:
-
superoxide dismutase
- NMDA:
-
N- methyl-D-aspartate
References
Parks DA, Granger DN 1986 Contributions of ischemia and reperfusion to mucosal lesion formation. Am J Physiol 250:G749–G753
McCord JM 1985 Oxygen-derived free radicals in postischemic tissue injury. N Engl J Med 312: 159–163
Saugstad OD 1988 Hypoxanthine as an indicator of hypoxia: its role in health and disease through free radical production. Pediatr Res 23: 143–150
Kinnula VL, Crapo JD, Raivio KO 1995 Generation and disposal of reactive oxygen metabolites in the lung. Lab Invest 73: 3–19
Halliwell B, Gutteridge JMC 1992 Free Radicals in Biology and Medicine, 2nd Ed. Clarendon Press, Oxford, UK
Granger DN 1988 Role of xanthine oxidase and granulocytes in ischemia-reperfusion injury. Am J Physiol 255:H1269–H1275
Weiss SJ 1989 Tissue destruction by neutrophils. N Engl J Med 320: 365–376
Kinnula VL, Mirza Z, Crapo JD, Whorton AR 1993 Modulation of hydrogen peroxide release from vascular endothelial cells by oxygen. Am J Respir Cell Mol Biol 9: 603–609
Pourcyrous M, Leffler CW, Bada HS, Korones SB, Busija DW 1993 Brain superoxide anion generation in asphyxiated piglets and the effect of indomethacin at therapeutic dose. Pediatr Res 34: 366–369
Cross AR, Jones OGT 1991 Enzymatic mechanisms of superoxide production. Biochim Biophys Acta 1057: 291–298
Mohazzab-H KM, Kaminski PM, Wolin MS 1994 NADH oxidoreductase is a major source of superoxide anion in bovine coronary artery endothelium. Am J Physiol 266:H2568–H2572
Parks DA, Granger DN 1986 Xanthine oxidase: biochemistry, distribution and physiology. Acta Physiol Scand Suppl 548: 87–99
Nishino T 1994 The conversion of xanthine dehydrogenase to xanthine oxidase and the role of the enzyme in reperfusion injury. J Biochem 116: 1–6
Stirpe F, Della Corte E 1969 The regulation of rat liver xanthine oxidase. Conversion in vitro of the enzyme activity from dehydrogenase (type D) to oxidase (type O). J Biol Chem 244: 3855–3863
Xia Y, Zweier JL 1995 Substrate control of free radical generation from xanthine oxidase in the postischemic heart. J Biol Chem 270: 18797–18803
Hagberg H, Andersson P, Lacarewicz J, Jacobson I, Butcher S, Sandberg M 1987 Extracellular adenosine, inosine, hypoxanthine, and xanthine in relation to tissue nucleotides and purines in rat striatum during transient ischemia. J Neurochem 49: 227–231
Ruth V, Fyhrquist F, Clemons G, Raivio KO 1988 Cord plasma vasopressin, erythropoietin, and hypoxanthine as indices of asphyxia at birth. Pediatr Res 24: 490–494
Ames BN, Cathcart R, Schwiers E, Hochstein P 1981 Uric acid provides an antioxidant defense in humans against oxidant-and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci USA 78: 6858–6862
Godin DV, Ko KM 1991 Allopurinol and ischemia/reperfusion injury: New use for an old drug?. Can J Cardiol 7: 163–169
Paller MS, Hoidal JR, Ferris TF 1984 Oxygen free radicals in ischemic acute renal failure in the rat. J Clin Invest 74: 1156–1164
Lemasters J, Thurman RG 1993 Hypoxia and reperfusion injury to liver. Prog Liver Dis 11: 85–114
Adkins WK, Taylor AE 1990 Role of xanthine oxidase and neutrophils in ischemia-reperfusion injury in rabbit lung. J Appl Physiol 69: 2012–2018
McCutchan HJ, Schwappach JR, Enquist EG, Walden DL, Terada LS, Reiss OK, Leff JA, Repine JE 1990 Xanthine oxidase-derived H2O2 contributes to reperfusion injury of ischemic skeletal muscle. Am J Physiol 258:H1415–H1419
Palmer C 1995 Hypoxic-ischemic encephalopathy. Therapeutic approaches against microvascular injury, and role of neutrophils, PAF, and free radicals. Clin Perinatol 22: 481–517
Lasley RD, Ely SW, Berne RM, Mentzer RM Jr 1988 Allopurinol enhanced adenine nucleotide repletion after myocardial ischemia in the isolated rat heart. J Clin Invest 81: 16–20
Moorhouse PC, Grootveld M, Halliwell B, Quinlan JG, Gutteridge JMC 1987 Allopurinol and oxipurinol are hydroxyl radical scavengers. FEBS Lett 249: 23–28
Johnson JL, Rajagopalan KV, Cohen HV 1974 Molecular basis for the biological function of molybdenum. Effect of tungsten on xanthine oxidase and sulfite oxidase in the rat. J Biol Chem 249: 859–866
Brown JM, Terada LS, Grosso MA, Whitmann GJ, Velasco SE, Patt A, Harken AH, Repine JE 1988 Xanthine oxidase produces hydrogen peroxide which contributes to reperfusion injury of ischemic, isolated, perfused rat hearts. J Clin Invest 81: 1297–1301
Nielsen VG, Tan S, Baird MS, McCammon AT, Parks DA 1996 Gastric intramucosal pH and multiple organ injury: impact of ischemia-reperfusion and xanthine oxidase. Crit Care Med 24: 1339–1344
Linas SL, Whittenburg D, Repine JE 1990 Role of xanthine oxidase in ischemia/reperfusion injury. Am J Physiol 258:F711–F716
Thompson-Gorman SL, Zweier JL 1990 Evaluation of the role of xanthine oxidase in myocardial reperfusion injury. J Biol Chem 265: 6656–6663
Zweier JL, Kuppusamy P, Lutty GA 1988 Measurement of endothelial cell free radical generation: evidence for a central mechanism of free radical injury in postischemic tissues. Proc Natl Acad Sci USA 85: 4046–4050
Zweier JL, Broderick R, Kuppusamy P, Thompson-Gorman S, Lutty GA 1994 Determination of the mechanism of free radical generation in human aortic endothelial cells exposed to anoxia and reoxygenation. J Biol Chem 269: 24156–24162
Krenitsky TA, Tuttle JV, Cattau EL Jr, Wang P 1974 A comparison of the distribution and electron acceptor specificities of xanthine oxidase and aldehyde oxidase. Comp Biochem Physiol 49B: 687–703
Wajner M, Harkness RA 1989 Distribution of xanthine dehydrogenase and oxidase activities in human and rabbit tissues. Biochim Biophys Acta 991: 79–84
Sarnesto A, Linder N, Raivio KO 1996 Organ distribution and molecular forms of human xanthine dehydrogenase/xanthine oxidase protein. Lab Invest 74: 48–56
Vettenranta K, Raivio KO 1990 Xanthine oxidase during human fetal development. Pediatr Res 27: 286–288
Jarasch E-D, Grund C, Bruder G, Heid HW, Keenan TW, Franke WW 1981 Localization of xanthine oxidase in mammary-gland epithelium and capillary endothelium. Cell 25: 67–82
Jarasch E-D, Bruder G, Heid HW 1986 Significance of xanthine oxidase in capillary endothelial cells. Acta Physiol Scand Suppl 548: 39–46
Moriwaki Y, Yamamoto T, Suda M, Nasako Y, Takahashi S, Agbedana OE, Hada T, Higashino K 1993 Purification and immunohistochemical localization of human xanthine oxidase. Biochim Biophys Acta 1164: 327–330
Betz AL 1985 Identification of hypoxanthine transport and xanthine oxidase activity in brain capillaries. J Neurochem 44: 574–579
Terada LS, Willingham IR, Rosandich ME, Leff JA, Kindt GW, Repine JE 1991 Generation of superoxide anion by brain endothelial cell xanthine oxidase. J Cell Physiol 148: 191–196
Hassoun PM, Yu FS, Shedd AL, Zulueta JJ, Thannickal VJ, Lanzillo JJ, Fanburg BL 1994 Regulation of endothelial cell xanthine dehydrogenase/xanthine oxidase gene expression by oxygen tension. Am J Physiol 266:L163–L171
Dupont GP, Huecksteadt TP, Marshall BC, Ryan US, Michael JR, Hoidal JR 1992 Regulation of xanthine dehydrogenase and xanthine oxidase activity and gene expression in cultured rat pulmonary endothelial cells. J Clin Invest 89: 197–202
Partridge CA, Blumenstock FA, Malik AB 1992 Pulmonary microvascular endothelial cells constitutively release xanthine oxidase. Arch Biochem Biophys 294: 184–187
Terada LS, Guidot DM, Leff JA, Willingham IR, Hanley ME, Piermattei D, Repine JE 1992 Hypoxia injures endothelial cells by increasing endogenous xanthine oxidase activity. Proc Natl Acad Sci USA 89: 3362–3366
Hassoun PM, Yu FS, Zulueta JJ, White AC, Lanzillo JJ 1995 Effect of nitric oxide and cell redox status on the regulation of endothelial cell xanthine dehydrogenase. Am J Physiol 268:L809–L817
Waud WR, Rajagopalan KV 1972 The mechanism of conversion of rat liver xanthine dehydrogenase from an NAD+ -dependent form (type D) to an O2- dependent form (type O). Arch Biochem Biophys 172: 365–379
Engerson TD, McKelvey TG, Rhyne DB, Boggio EB, Snyder SJ, Jones HP 1987 Conversion of xanthine dehydrogenase to oxidase in ischemic rat tissues. J Clin Invest 79: 1564–1570
McKelvey TG, Hollwarth ME, Granger DN, Engerson TD, Landler U, Jones HP 1988 Mechanisms of conversion of xanthine dehydrogenase to xanthine oxidase in ischemic rat liver and kidney. Am J Physiol 254:G753–G760
Brass CA, Narciso J, Gollan JL 1991 Enhanced activity of the free radical producing enzyme xanthine oxidase in hypoxic rat liver. Regulation and pathophysiologic significance. J Clin Invest 87: 424–431
Wiezorek JS, Brown DH, Kupperman DE, Brass CA 1994 Rapid conversion to high xanthine oxidase activity in viable Kupffer cells during hypoxia. J Clin Invest 94: 2224–2230
Nishino T, Tamura I 1991 The mechanism of conversion of xanthine dehydrogenase to oxidase and the role of the enzyme in reperfusion injury. Adv Exp Med Biol 309A: 327–337
Rothwell NJ, Hopkins SJ 1995 Cytokines and the nervous system. II. Actions and mechanisms of action. Trends Neurol Sci 18: 130–136
Minami M, Kurashi Y, Yabuuchi K, Yamazaki A, Satoh M 1992 Induction of interleukin-1 mRNA in rat brain after transient forebrain ischemia. J Neurochem 58: 390–392
Gehrmann J, Bonnekoh P, Miyazawa T, Oschlies U, Duz E, Hosmann KA, Kreutzberg GW 1992 The microglia reaction in the rat hippocampus following global ischemia: immunoelectron microscopy. Acta Neuropathol 84: 588–595
Morioka T, Kalehua AN, Streit WJ 1992 Progressive expression of immunomolecules on microglial cells in rat dorsal hippocampus following transient forebrain ischemia. Acta Neuropathol 83: 149–157
McRae A, Gilland E, Bona E, Hagberg H 1995 Microglia activation after neonatal hypoxia-ischemia. Dev Brain Res 84: 245–252
Yamasaki Y, Matsuura N, Shozuhara H, Onodera H, Itoyama Y, Kogure K 1995 Interleukin-1 as a pathogenic mediator of ischemic brain damage in rats. Stroke 26: 676–681
Relton JK, Rothwell NJ 1992 Interleukin-1 receptor antagonis inhibits ischaemic and excitotoxic neuronal damage in the rat. Brain Res Bull 29: 243–246
Betz AL, Yang G-Y, Davidson BL 1995 Attenuation of stroke size in rats using an adenoviral vector to induce overexpression of interleukin-1 receptor antagonist in brain. J Cereb Blood Flow Metab 15: 547–551
Bruce AJ, Boling W, Kindy MS, Peschon J, Kraemer PJ, Carpenter MK, Holtsberg FW, Mattson MP 1996 Altered neuronal and microglial responses to excitotoxic and ischemic brain injury in mice lacking TNF receptor. Nat Med 2: 788–794
Okada Y, Copeland BR, Mori E, Tung MM, Thomas WS, del Zoppo GJ 1994 P-selectin and intercellular adhesion molecule-1 expression after focal brain ischemia and reperfusion. Stroke 25: 202–211
Matsuo Y, Onodera H, Shiga Y, Nakamura M, Ninomiya M, Kihara T, Kogure K 1994 Correlation between myeloperoxidase-quantified neutrophil accumulation and ischemic brain injury in the rat. Effects of neutrophil depletion. Stroke 25: 1469–1475
von Andrian UH, Chambers JD, McEvoy LM, Bargatze RF, Arfors KE, Butcher EC 1991 Two-step model of leukocyte-endothelial cell interaction in inflammation: distinct roles for LECAM-1 and the leukocyteβ2 integrins in vivo. Proc Natl Acad Sci USA 88: 7538–7542
Hansen PR 1995 Role of neutrophils in myocardial ischemia and reperfusion. Circulation 91: 1872–1885
Etzioni A 1996 Adhesion molecules-their role in health and disease. Pediatr Res 39: 191–198
Romson JL, Hook BG, Kunkel SL, Abrams GD, Schork MA, Lucchesi BR 1983 Reduction of the extent of ischemic myocardial injury by neutrophil depletion in the dog. Circulation 67: 1016–1023
Hernandez LA, Grisham MB, Twohig B, Arfors KE, Harlan JM, Granger DN 1987 Role of neutrophils in ischemia-reperfusion-induced microvascular injury. Am J Physiol 253:H699–H703
Vedder NB, Winn RK, Rice CL, Chi EY, Arfors KE, Harlan JM 1988 A monoclonal antibody to adherence-promoting leukocyte glycoprotein, CD18, reduces organ injury and improves survival from hemorrhagic shock and resuscitation in rabbits. J Clin Invest 81: 939–944
Ma X-L, Tsao PS, Lefer AM 1991 Antibody to CD-18 exerts endothelial and cardiac protective effects in myocardial ischemia and reperfusion. J Clin Invest 88: 1237–1243
Yoshida N, Granger DN, Anderson DC, Rothlein R, Lane C, Kvietys PR 1992 Anoxia/reoxygenation-induced neutrophil adherence to cultured endothelial cells. Am J Physiol 262:H1891–H1898
Hallenbeck JM, Dutka AJ, Tanishima T, Kochanek PM, Kumaroo KK, Thompson CB, Obrenovich TP, Contreras TJ 1986 Polymorphonuclear leukocyte accumulation in brain regions with low blood flow during the early postischemic period. Stroke 17: 246–253
Grisham MB, Hernandez LA, Granger DN 1986 Xanthine oxidase and neutrophil infiltration in intestinal ischemia. Am J Physiol 251:G567–G574
Patel KD, Zimmerman GA, Prescott SM, McEver RP, McIntyre TM 1991 Oxygen radicals induce human endothelial cells to express GMP-140 and bind neutrophils. J Cell Biol 112: 749–759
McEver RP 1991 Selectins: novel receptors that mediate leukocyte adhesion during inflammation. Thromb Haemost 65: 223–228
Vannucci RC 1993 Experimental models of perinatal hypoxic-ischemic brain damage. Acta Pathol Microb Scand 101( suppl 40): 89–95
Siesjö BK 1992 Pathophysiology and treatment of focal cerebral ischemia. Part I. Pathophysiology. J Neurosurg 77: 169–184
Rosenberg AA, Murdaugh E, White CW 1989 The role of oxygen free radicals in postasphyxia cerebral hypoperfusion in newborn lambs. Pediatr Res 26: 215–219
Karlsson BR, Grögaard B, Gerdin B, Steen PA 1994 The severity of postischemic hypoperfusion increases with duration of cerebral ischemia in rats. Acta Anaesthesiol Scand 38: 248–253
Pluta R, Lossinsky AS, Wisniewski HM, Mossakowski MJ 1994 Early blood-brain barrier changes in the rat following transient complete cerebral ischemia induced by cardiac arrest. Brain Res 633: 41–52
Petito CK, Pulsinelli WA, Jacobson G, Plum F 1982 Edema and vascular permeability in cerebral ischemia: Comparison between ischemic neuronal damage and infarction. J Neuropathol Exp Neurol 41: 423–436
del Zoppo GJ, Schmid-Schönbein GW, Mori E, Copeland BR, Chang C-M 1991 Polymorphonuclear leukocytes occlude capillaries following middle cerebral artery occlusion and reperfusion in baboons. Stroke 22: 1276–1283
Zhang R-L, Chopp M, Chen H, Garcia JH 1994 Temporal profile of ischemic tissue damage, neutrophil response, and vascular plugging following permanent and transient (2H) middle cerebral artery occlusion in the rat. J Neurol Sci 125: 3–10
Thomas WS, Mori E, Copeland BR, Yu J-Q, Morrissey JH, del Zoppo GJ 1993 Tissue factor contributes to microvascular defects after focal cerebral ischemia. Stroke 24: 847–854
Connolly ES, Winfree CJ, Springer TA, Naka Y, Liao H, Yan SD, Stern DM, Solomon RA, Gutierrez-Ramos J-C, Pinsky DJ 1996 Cerebral protection in homozygous null ICAM-1 mice after middle cerebral artery occlusion. Role of neutrophil adhesion in the pathogenesis of stroke. J Clin Invest 97: 209–216
Vannucci RC 1990 Experimental biology of cerebral hypoxia-ischemia: relation to perinatal brain damage. Pediatr Res 27: 317–326
Pulsinelli WA, Brierley JB, Plum F 1982 Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 11: 491–498
Lorek A, Takei Y, Cady E, Wyatt J, Penrice J, Edwards A, Peebles D, Wylezinska M, Owen-Reece H, Kirkbridge V, Cooper C, Aldrige R, Roth S, Brown G, Delpy D, Reynolds EOR 1994 Delayed (“secondary”) cerebral energy failure following acute hypoxia-ischemia in the newborn piglet: continuous 48-hour studies by phosphorus magnetic resonance spectroscopy. Pediatr Res 36: 699–706
Zini I, Tomasi A, Grimaldi R, Vannini V, Agnati LF 1992 Detection of free radicals during brain ischemia and reperfusion by spin trapping and microdialysis. Neurosci Lett 138: 279–282
Phillis JW, Sen S 1993 Oxypurinol attenuates hydroxyl radical production during ischemia/reperfusion injury of the rat cerebral cortex: an ESR study. Brain Res 628: 309–312
Hasegawa K, Yoshioka H, Sawada T, Nishikawa H 1993 Direct measurement of free radicals in the neonatal mouse brain subjected to hypoxia: an electron spin resonance spectroscopic study. Brain Res 607: 161–166
Yang G, Chan PH, Chen J, Carlson E, Chen SF, Weinstein P, Epstein CJ, Kamii H 1994 Human copper-zinc superoxide dismutase transgenic mice are highly resistant to reperfusion injury after focal cerebral ischemia. Stroke 25: 165–170
Connors G, Hunse C, Gagnon R, Richardson B, Han V, Rosenberg H 1992 Perinatal assessment of cerebral blood flow velocity wave forms in the human fetus and neonate. Pediatr Res 31: 649–652
Eken P, Toet MC, Groenendaal F, de Vries LS 1995 Predictive value of early neuroimaging, pulsed Doppler and neurophysiology in full term infants with hypoxic-ischemic encephalopathy. Arch Dis Child 73:F75–F80
Archer LNJ, Levene MI, Evans DH 1986 Cerebral artery Doppler ultrasonography for prediction of outcome after perinatal asphyxia. Lancet 2: 1116–1118
Frewen TC, Kissoon N, Kronick J, Fox M, Lee R, Bradwin N, Chance G 1991 Cerebral blood flow, cross-brain oxygen extraction, and fontanelle pressure after hypoxic-ischemic injury in newborn infants. J Pediatr 118: 265–271
Wyatt JS 1993 Near-infrared spectroscopy in asphyxial brain injury. Clin Perinatol 20: 369–378
Van Bel F, Dorrepaal CA, Benders MJNL, Zeeuwe PEM, van de Bor M, Berger HM 1993 Changes in cerebral hemodynamics and oxygenation in the first 24 hours after birth asphyxia. Pediatrics 92: 365–372
Pryds O, Greisen G, Lou H, Friis-Hansen B 1990 Vasoparalysis associated with brain damage in asphyxiated term infants. J Pediatr 117: 119–125
Volpe JJ, Herscovitch P, Perlman JM, Kreusser KL, Raichle ME 1985 Positron emission tomography in the asphyxiated term newborn: parasagittal impairment of cerebral blood flow. Ann Neurol 17: 287–296
Shankaran S, Kottamasu SR, Kuhns L 1993 Brain sonography, computed tomography, and single-photon emission computed tomography in term neonates with perinatal asphyxia. Clin Perinatol 20: 379–394
Wyatt JS, Edwards AD, Azzopardi D, Reynolds EO 1989 Magnetic resonance and near infrared spectroscopy for investigation of perinatal hypoxic-ischaemic brain injury. Arch Dis Child 64: 953–963
Azzopardi D, Wyatt JS, Cady EB, Delpy DT, Baudin J, Stewart AL, Hope PL, Hamilton PA, Reynolds EO 1989 Prognosis of newborn infants with hypoxic-ischemic brain injury assessed by phosphorus magnetic resonance spectroscopy. Pediatr Res 25: 445–451
Volpe, JJ 1995 Neurology of the Newborn, 3rd ed. WB Saunders, Philadelphia, pp 260–369
Rademakers RP, van der Knaap MS, Verbeeten B Jr, Barth PG, Valk J 1995 Central cortico-subcortical involvement: a distinct pattern of brain damage caused by perinatal and postnatal asphyxia in term infants. J Comput Assist Tomogr 19: 256–263
Barkovich AJ, Westmark K, Partridge C, Sola A, Ferriero DM 1995 Perinatal asphyxia: MR findings in the first 10 days. AJNR 16: 427–438
Dugan LL, Choi DW 1994 Excitotoxicity, free radicals, and cell membrane changes. Ann Neurol 35:S17–S21
Lipton SA, Rosenberg PA 1994 Mechanisms of disease: excitatory amino acids as a final common pathway for neurological disorders. N Engl J Med 330: 613–622
Choi DW 1995 Calcium: still center-stage in hypoxic-ischemic neuronal cell death. Trends Neurosci 18: 58–60
Pellegrini-Giampietro DE, Cherici G, Alesiani M, Carla F, Moroni F 1988 Excitatory amino acid release from rat hippocampal slices as a consequence of free-radical formation. J Neurochem 51: 1960–1963
Lafon-Cazal M, Pietri S, Culcasi M, Bockaert J 1993 NMDA-dependent superoxide production and neurotoxicity. Nature 364: 535–537
Uyama O, Matsuyama T, Michishita H, Nakamura H, Sugita M 1992 Protective effects of human recombinant superoxide dismutase on transient ischemic injury of CA1 neurons in gerbils. Stroke 23: 75–81
Rosenbaum DM, Kalberg J, Kessler JA 1994 Superoxide dismutase ameliorates neuronal death from hypoxia in culture. Stroke 25: 857–863
Dawson VL, Dawson TM, London ED, Bredt DS, Snyder SH 1991 Nitric oxide mediates glutamate neurotoxicity in primary cortical cultures. Proc Natl Acad Sci USA 88: 6368–6371
Lipton SA, Choi Y-B, Pan Z-H, Lei SZ, Chen H-SV, Sucher NJ, Loscalzo J, Singel DJ, Stamler JS 1993 A redox-based mechanism for the neuroprotective and neurodestructive effects of nitric oxide and related nitroso compounds. Nature 364: 626–632
Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA 1990 Apparent hydroxyl radical production by peroxynitrite: implications for endothelial injury from nitric oxide and superoxide. Proc Natl Acad Sci USA 87: 1620–1624
Bonfoco E, Krainc D, Ankarcrona M, Nicotera P, Lipton SA 1995 Apoptosis and necrosis: two distinct events induced, respectively, by mild and intense insults with N-methyl-D-aspartate or nitric oxide/superoxide in cortical cell cultures. Proc Natl Acad Sci USA 92: 7162–7166
Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, Nicotera P 1995 Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron 15: 961–973
Tominaga T, Kure S, Narisawa K, Yoshimoto T 1993 Endonuclease activation following focal ischemic injury in the rat brain. Brain Res 608-: 21–26
Kure S, Tominaga T, Yoshimoto T, Tada K, Narisawa K 1991 Glutamate triggers internucleosomal DNA cleavage in neuronal cells. Biochem Biophys Res Commun 179: 39–45
MacManus JP, Buchan AM, Hill IE, Rasquinha I, Preston E 1993 Global ischemia can cause DNA fragmentation indicative of apoptosis in rat brain. Neurosci Lett 164: 89–92
Linnik MD, Zahos P, Geschwind MD, Federoff HJ 1995 Expression of bcl-2 from a defective herpes simplex virus-1 vector limits neuronal death in focal cerebral ischemia. Stroke 26: 1670–1675
Li Y, Chopp M, Jiang N, Zhang ZG, Zaloga C 1995 Induction of DNA fragmentation after 10 to 120 minutes of focal cerebral ischemia in rats. Stroke 26: 1252–1258
Mehmet H, Yue X, Squier MV, Lorek A, Cady E, Penrice J, Sarraf C, Wylezinska M, Kirkbridge V, Cooper C, Brown GC, Wyatt JS, Reynolds EOR, Edwards AD 1994 Increased apoptosis in the cingulate sulcus of newborn piglets following transient hypoxia-ischemia is related to the degree of high energy phosphate depletion during the insult. Neurosci Lett 181: 121–125
Ratan RR, Murphy TH, Baraban JM 1994 Oxidative stress induces apoptosis in embryonic cortical neurons. J Neurochem 62: 376–379
Rosenbaum DM, Michaelson M, Batter DK, Doshi P, Kessler JA 1994 Evidence for hypoxia-induced, programmed cell death of cultured neurons. Ann Neurol 36: 864–870
Hockenbery DM, Oltvai ZN, Yin X-M, Milliman CL, Korsmeyer SJ 1993 Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 75: 241–251
Zhong L, Kane DJ, Bredesen DE 1993 BCL-2 blocks glutamate toxicity in neural cell lines. Mol Brain Res 19: 353–355
Behl C, Hovey LI, Krajewski S, Schubert D, Reed J 1993 Bcl-2 prevents killing of neuronal cells by glutamate but not by amyloid beta protein. Biochem Biophys Res Commun 197: 949–956
An G, Lin T-N, Liu J-S, Xue J-J, He Y-Y, Hsu CY 1993 Expression of c-fos and c-jun family genes after focal cerebral ischemia. Ann Neurol 33: 457–464
Brand T, Sharma HS, Fleischmann KE, Duncker DJ, McFalls EO, Verdouw PD, Schaper W 1992 Proto-oncogene expression in porcine myocardium subjected to ischemia and reperfusion. Circ Res 71: 1351–1360
Witzgall R, Brown D, Schwarz C, Bonventre JV 1994 Localization of proliferating cell nuclear antigen, vimentin, c-Fos, and clusterin in the postischemic kidney. Evidence for a heterogenous genetic response among nephron segments, and a large pool of mitotically active and dedifferentiating cells. J Clin Invest 93: 2175–2188
Pombo CM, Bonventre JV, Avruch J, Woodgett JR, Kyriakis JM, Force T 1994 The stress-activated protein kinases are major c-jun amino-terminal kinases activated by ischemia and reperfusion. J Biol Chem 269: 26546–26551
Morooka H, Bonventre JV, Pombo CM, Kyriakis JM, Force T 1995 Ischemia and reperfusion enhance ATF-2 and c-jun binding to cAMP response elements and to an AP-1 binding site from the c-jun promoter. J Biol Chem 270: 30084–30092
Estus S, Zaks WJ, Freeman RS, Gruda M, Bravo R, Johnson EM Jr 1994 Altered gene expression in neurons during programmed cell death: identification of c-jun as necessary for neuronal apoptosis. J Cell Biol 127: 1717–1727
Lindvall O, Ernfors P, Bengzon J, Kokaia Z, Smith M-L, Siesjö BK, Persson H 1992 Differential regulation of mRNAs for nerve growth factor, brain-derived neurotrophic factor, and neurotrophin 3 in the adult rat brain following cerebral ischemia and hypoglycemic coma. Proc Natl Acad Sci USA 89: 648–652
Rangan U, Bulkley GB 1993 Prospects for treatment of free radical-mediated tissue injury. Br Med Bull 49: 700–718
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fellman, V., Raivio, K. Reperfusion Injury as the Mechanism of Brain Damage after Perinatal Asphyxia. Pediatr Res 41, 599–606 (1997). https://doi.org/10.1203/00006450-199705000-00001
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199705000-00001
This article is cited by
-
Elevated blood pressure among children born to women with obstructed labour in Eastern Uganda: a cohort study
Clinical Hypertension (2024)
-
Hydrogen gas can ameliorate seizure burden during therapeutic hypothermia in asphyxiated newborn piglets
Pediatric Research (2024)
-
Renal artery flow alterations in neonates with hypoxic ischemic encephalopathy
Pediatric Nephrology (2024)
-
Occurrence of hyperoxia during iNO treatment for persistent pulmonary hypertension of the newborn: a cohort study
European Journal of Pediatrics (2024)
-
Impact of hydrogen gas inhalation during therapeutic hypothermia on cerebral hemodynamics and oxygenation in the asphyxiated piglet
Scientific Reports (2023)