Abstract
Background:
To characterize the ontogeny of plasma albumin and total proteins, due to the lack of a comprehensive pediatric database. Secondly, to establish the magnitude and duration of maturational changes in binding of highly-bound drugs/chemicals.
Methods:
Anonymized plasma samples from 296 donors were pooled in 6 age brackets from birth to adolescence. Total protein and albumin levels were measured in each age group, as was the age-dependency of plasma binding of diazepam (DZP), cyclosporine (CYC), and deltamethrin (DLM), a pyrethroid insecticide.
Results:
Plasma levels of albumin and total proteins steadily increased for the first 1–3 y of life. Unbound DZP and CYC fractions were elevated three- to fourfold in neonates, but decreased to adult levels after 1 and 3 y, respectively. Unbound DLM levels exceeded those in adults for just 1 mo.
Conclusion:
Neonates and infants under 1–3 y may be at risk from increased amounts of free drug, when given standard doses of some highly-bound drugs. Pyrethroid insecticides might be anticipated to pose increased risk for 1 mo.
Similar content being viewed by others
Main
The plasma protein binding of highly-bound drugs can have significant clinical implications (1,2,3). Binding limits the amount of compound that can exit the bloodstream and reach peripheral sites of action, storage, and elimination. Pharmacological activity and toxicity are generally assumed to be correlated with, and determined by unbound drug concentrations in plasma. An equilibrium exists between the concentration of unbound drug in the plasma and the site(s) of action, when drug distribution is governed by passive diffusion. Although therapeutic ranges for drug monitoring are frequently reported in terms of total drug concentration in blood or plasma, the unbound drug concentration provides a better index for monitoring highly-bound compounds. This is particularly true for patients for whom there are disease- or age-related alterations in plasma protein content or composition (4,5,6) Adverse effects of diazepam in newborns, for example, have been attributed to significant elevations in serum concentrations of unbound parent compound and N-desmethyldiazepam, its major active metabolite (7).
There are number of factors that influence plasma binding of drugs and other chemicals, some of which can be age-related. These include the drug and its concentration, the presence of binding competitors, the type and quantity of plasma protein, and the proteins’ affinity for the drug. Nau et al. (7) observed a twofold increase in the free fraction of diazepam in the serum of neonates that accompanied marked increases in free fatty acids and bilirubin, competitors for albumin-binding sites. Albumin levels progressively rose during a 6-d postgestation monitoring period, but remained significantly lower than in adults. Albumin is the major plasma protein, accounting for some 55% of total proteins. Many neutral and some basic drugs bind primarily to albumin by hydrophobic and ionic bonding, respectively (8). α, β, and γ globulins are the second major class, comprising ~38% of proteins. Globulins transport a variety of endogenous substances. The globulin α1-acid glycoprotein (AAG), accounts for only 2% of plasma proteins, but binds many basic, or cationic drugs (8). Lipoproteins play key roles in transporting endogenous lipids, as well as neutral and basic lipophilic drugs (9).
Pediatric patients, especially neonates, have been reported to have lower protein binding of a number of commonly prescribed drugs. A high degree of correlation has been seen between the early postnatal rise in plasma albumin levels and the fraction of drugs bound (10,11,12). The majority of investigations have been conducted with mixed cord blood immediately after parturition. Unfortunately, there are few and incomplete data on the ontogeny of plasma proteins and binding from infancy through adolescence. The size of study populations of infants and children is usually quite small, due to the difficulty of securing healthy subjects to serve as blood donors. Age brackets are typically too broad and the ages too diverse to accurately characterize the sequence of maturational changes ( Table 1 ). In one such study of albumin levels and plasma protein binding of disopyramide, the wide age-brackets included neonates (<1 mo), infants (1 mo to 2 y), children (2–16 y), and adults (13). The adults were healthy, but the young were all patients. Nau et al. (7) assessed 8–13 subjects, for whom five diazepam binding parameters were monitored the first 6 d of life. This study period was too short for albumin and bilirubin levels or drug binding to approach adult values. One of the most comprehensive studies of the ontogeny of albumin was conducted by Kanakoudi et al. (14). They measured serum levels of albumin, AAG and eight other proteins soon after birth and at the end of 1, 3, and 6 mo in relatively large numbers of subjects. Albumin and AAG concentrations remained lower at 6 mo than in adults. The resulting data were quite useful, but did not define increases during the first weeks of life nor subsequent changes in infancy or childhood. Lerman et al. (15) did characterize the ontogeny of AAG in groups of 10–20 subjects from birth to adolescence. These researchers found an inverse relationship between the serum concentrations of AAG and free fraction of lidocaine from birth to 1 y of age.
The current study was undertaken as part of an effort to determine whether neurological effects of pyrethroids are age-dependent. Children and adults are frequently exposed to low levels of pyrethroids, the most widely-utilized insecticides in the United States and the European Union (16,17). As our pilot experiments during analytical method development indicated that pyrethroids such as deltamethrin (DLM) are highly bound to plasma proteins and lipoproteins (18), we were concerned that lower binding in neonates, infants, or young children may contribute to increased deposition in the central nervous system (CNS). Children 1–2 y old are of concern to the United States EPA due to their potential for higher DLM ingestion and skin exposure. Although most pyrethroids are relatively nontoxic to humans and other mammals, high doses of some congeners can be acutely neurotoxic. Clinical signs are primarily attributed to the binding of the parent (unmetabolized) compound to voltage-gated sodium channels in axonal membranes (19). Thus, an important study objective was to learn whether levels of unbound DLM are elevated during immaturity and for how long this condition persists.
An excellent source of pediatric plasma samples was identified, allowing a comprehensive characterization of the ontogeny of the plasma albumin and total proteins, as well as the binding of DLM, diazepam and cyclosporine. All three compounds are very lipophilic and extensively bound, but bind to different plasma components. Published Log P values for diazepam, DLM, and cyclosporine are 3.12, 4.6, and 6.92. Diazepam binds almost exclusively to albumin, notably to the protein’s so-called benzodiazepine site (20). DLM binds hydrophobically to both albumin and lipoprotein. Albumin and lipoprotein binding account for 60 and 30% of the insecticide in adult human plasma (16). Cyclosporine the most highly lipophilic compound, associates largely with lipoproteins by partitioning into their nonpolar lipid core (9,21). In view of incomplete ontogeny data in the literature, it was deemed worthwhile to establish with greater certainty when plasma binding of each of the three compounds attained adult levels. These findings may be relevant, or applicable to other pharmaceutical agents with similar binding characteristics.
Results
Ontogeny of Plasma Albumin and Total Proteins
Levels of albumin and total proteins progressively increased in the plasma during maturation Table 2 . Apparent increases in albumin concentrations from one age group to the next were modest, such that no one group was significantly different from the next. Values for donors 3 y of age and younger were significantly lower than for adults. In contrast to albumin, total plasma protein levels rose substantially during the second, third, and fourth weeks. The contribution of albumin to total proteins diminished from ~65 to 55% during this period and remained constant through adolescence. The more pronounced rise in total plasma proteins indicates that proteins in addition to albumin are synthesized in increasing amounts during the first year of life.
Plasma Drug Binding During Maturation
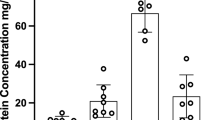
The extent of plasma binding of diazepam ( Figure 1 , panel a) and cyclosporine ( Figure 1 , panel b) exhibited similar changes during maturation. The unbound fraction of each drug was highest in neonates (birth to 1 wk). The percentage of diazepam that was unbound was approximately threefold higher in neonatal (birth to 1 wk) than in adult plasma. Binding of diazepam decreased significantly in successive age groups during the first year of life. Differences from adults were no longer seen in infants and children older than 1 y. The percentage of unbound cyclosporine was approximately fourfold greater in neonates than adults. Although the unbound fraction of the drug progressively decreased, 1- to 3-y olds still exhibited significantly lower binding than children, adolescents, and adults. The extent of plasma binding of both diazepam and cyclosporine remained quite consistent in these three oldest age groups.
Effect of age on plasma binding of diazepam (a) and cyclosporine (b). Bars represent mean % unbound drug (SD). Values for age-groups with the same letter (a, b, and c) are not statistically different.
Plasma Pyrethroid Binding During Maturation
The age-dependency of plasma binding of DLM differed from that of diazepam and cyclosporine ( Figure 2 ). The unbound fraction of the pyrethroid insecticide, like the two drugs, was highest in neonates. This fraction remained elevated in the > 1-wk to 4-wk group, but matched adult values thereafter. It is apparent in Figure 2 that DLM binds to both plasma proteins and lipoproteins. Plasma protein binding of DLM rose significantly during the initial 4 wk, though there was relatively little increase in lipoprotein binding for the first 3 y. Once binding maturity was attained after 4 wk, protein and lipoprotein binding accounted for 55 and 35% of the total amount of DLM in plasma, respectively. Only ~10% remained unbound.
Effect of age on plasma binding of Deltamethrin (DLM). Bars represent mean (SD). Asterisks indicate statistically significant difference from unmarked values for the same parameter in other age groups. No fill = Unbound. Filled solid = Lipoprotein bound. Filled gray = Albumin bound.
Discussion
Our evaluation of plasma samples from 296 pediatric donors yielded the most comprehensive and detailed database, to our knowledge, that is available on the ontogeny of plasma albumin and total proteins, as well as maturation of plasma binding of selected drugs and a common insecticide. Albumin concentrations rose progressively, initially attaining values that were not significantly different from adults in the 3- to 6-y age group. Other pediatric albumin data sets we found were incomplete, lacked sufficient numbers of healthy subjects, or had broad age brackets. A survey by Kanakoudi et al. (14) included a large number of donors who were monitored for just the first 6 mo of life. Albumin levels remained lower in their 6-mo-old infants than in adults, as did prealbumin. Although albumin is the most abundant protein in plasma, its concentrations increased modestly during infancy and early childhood in the current study. Total plasma protein levels increased during the first year of life, as did binding of diazepam, cyclosporine, and DLM. Inverse relationships have been observed by several other researchers between the postnatal rise in albumin levels and the free fraction of drugs that bind to it (10,11,12).
Newborns are particularly sensitive to diazepam’s CNS-depressant effects (22,23,24). High maternal doses during labor can result in elevated neonatal blood levels of free drug, which can produce inadequate respiration, apnea, “floppiness,” hypothermia and poor suckling. There is a limited basis on which to judge the age at which this is no longer of concern. One well-conducted study showed a 40% decrease in the free fraction of diazepam during the first week of life, but did not assess neonates older than 11 d of age (7). We found elevated unbound diazepam levels in plasma from infants younger than 1 y. The free fraction was threefold higher in the birth to 1-wk age group than in adults. The unbound fraction was still twofold higher in the > 4 wk to 1 y olds. There was no difference, however, between adults and children 1–3 y of age or older. The decreased binding at <1 y might be attributable to lower affinity of albumin for diazepam, as well as lower albumin levels. Adult and fetal albumin, however, are reported to have the same binding affinity for diazepam (25). Both albumin and total plasma protein levels increased in concert with the extent of diazepam binding in the present study during the initial year after birth. Assuming an absence of metabolism and elimination, a high unbound fraction of diazepam would result in more drug free to diffuse from the bloodstream into tissues, resulting in enhanced and prolonged CNS effects.
Clinical investigations clearly show that neonates’ and infants’ ability to metabolize diazepam is quite limited. The drug undergoes oxidative hydroxylation and demethylation to bioactive metabolites by cytochrome P450 (CYP) enzymes, and is subsequently glucuronidated and excreted. Treluyer et al. (26) found that urinary excretion of diazepam’s major metabolites was very low in the 1- and 2-d olds. There was a substantial increase in metabolite excretion during the initial week, although metabolism of the drug by hepatic microsomes remained lower than in adults in the 3- and 12-mo age groups. Hepatic CYP2C19 is largely responsible for demethylation of diazepam. This enzyme’s expression and activity approach adult levels within ~6 mo (27). Levels of CYP3A4, the enzyme largely responsible for catalyzing diazepam hydroxylation, rise gradually during the first 6 mo, but remain lower in children up to 2 y of age than in adults (28). Thus, increased amounts of free diazepam in the bloodstream, coupled with low metabolic clearance, can enhance CNS deposition. Pharmacodynamic factors may also contribute to the susceptibility of newborns and infants, as may the immature CNS and blood–brain barrier.
The extent of plasma binding of cyclosporine was also shown to be age-dependent in the present investigation. The unbound fraction of the drug was approximately fourfold higher in neonates than adults, but progressively decreased, reaching adult levels in the 3- to 6-y-old group. Sethi et al. (18) previously confirmed that cyclosporine binds primarily to the lipoprotein fraction of adult human plasma. The lipoprotein levels in plasma samples were not quantified, as others have characterized the ontogeny of major lipoproteins. In one such investigation, serum was taken from infants at 2, 4, 6, 9, and 12 mo (29). Cholesterol levels rose markedly for the first 2 mo and more slowly thereafter. Low-density lipoprotein, high-density lipoprotein, very low-density lipoprotein, and triglyceride levels increased slowly during the first 12 mo of life. Another research group measured cholesterol content of high-density lipoproteins in serum of subjects in six age groups ranging from newborns to adolescents and adults (30). high-density lipoprotein cholesterol increased quite modestly in males from birth to age 6–10 y. Somewhat larger increases were observed in females until they reached 11–15 y. It is likely that the progressive increase in cyclosporine binding we observed during the first 3 y in pooled male and female plasma samples was due in part to rising plasma lipoproteins.
Age-related differences in plasma binding have the greatest effect on systemic disposition of compounds that are highly bound to a single plasma component. Diazepam and cyclosporine are both highly (>95%) bound. As described previously diazepam binds primarily to albumin (20), while cyclosporine is largely associated with the lipoprotein fraction (21). Deficits of albumin or lipoproteins in plasma of neonates and infants thus contribute to the significant elevations seen in the unbound fraction of the corresponding drug in these age groups. Although the total binding of DLM to plasma is high (~90%), binding to each of its major components, lipoproteins (~30%) and proteins (~60%), is low to moderate. One of these plasma components serves to buffer/bind excess DLM resulting from age-dependent deficit in the other component. Accordingly, an age-related increase in unbound DLM is manifest only in the group with the lowest plasma protein and lipoprotein levels.
The finding that the amount of free DLM available for brain uptake and/or metabolic clearance is no higher for those older than 4 wk than for adults rules out plasma binding as a contributor to potential age-related differences in the insecticide’s CNS deposition in infants older than 4 wk. Neonates would be predicted to be at greater risk due to lower protein binding; however neonatal pyrethroid exposures should be negligible. In contrast, infants and children may ingest pyrethroids in house dust and from pets by hand to mouth activity, as well as in foods (31). As pyrethroids are very lipophilic chemicals, it appears likely the age-dependency of the plasma binding of highly lipophilic, persistent environmental pollutants (POPs), such as DDT, phthalates, polychlorinated biphenyls and dioxins, may be similar to DLM. Early investigators reported such chemicals are also transported in the bloodstream by albumin and lipoproteins (32,33). Neonatal exposures to POPs can be substantial, as these compounds cross the placenta and are concentrated in breast milk.
The binding data in this investigation can be used in the development of pediatric physiologically-based pharmacokinetic (PBPK) models for diazepam, cyclosporine, DLM and other pyrethroid insecticides. PBPK models are used to predict the time-course of a chemical in the blood or specific organs (i.e., the internal dose) affected by the agent (34). These computer-enabled models take into account key anatomical, physiological and biochemical features of laboratory animals and humans, as well as physicochemical properties of the compound of interest. Inclusion of accurate information about the chemical’s absorption, transport in the bloodstream, uptake by different tissues, metabolism, and elimination is essential. Input of complete and accurate data significantly reduces uncertainty in the models’ pharmacokinetic and pharmacodynamic simulations. PBPK models have been widely utilized for three decades to assess the toxicity of chemical agents (35,36). Such models are finding increasing use in drug development and regulation (37,38,39). Very few models, however, have yet been adapted and utilized to forecast the systemic uptake and disposition of therapeutic agents in infants and children. The nature and age-dependency of plasma binding of diazepam, cyclosporine and DLM can now be accounted for in PBPK models, whereas little to no information was previously available.
Methods
Blood and Plasma Donors
Anonymized plasma samples from a total of 296 individuals were obtained in the fall of 2012, from a College of American Pathologists-certified clinical testing laboratory in Children’s Hospital and Health System in Milwaukee, WI. Frozen plasma samples not used for clinical tests were retrieved, divided into appropriate age brackets, thawed, and equivalent volumes pooled by trained laboratory personnel. Unused whole blood samples were also pooled, plasma separated, and combined with thawed plasma samples of the same age bracket. The age brackets and number of samples in each bracket are shown in Table 2 . Specimens, from donors suffering from a condition anticipated to adversely impact albumin or lipoprotein levels, were excluded. The health of the subjects was not otherwise verified. Gender and ethnicity were not inclusion criteria. The pooled aliquots for each age bracket were frozen at −20 °C until analyses could be performed. The collection and use of the anonymous plasma samples were classified as exempted by the Children’s Hospital and Health System Institutional Review Board.
Frozen heparinized, pooled adult human plasma was purchased from Innovative Research (Novi, MI). The name, ethnicity, gender and number of donors were not specified, although the donors were 18–65 y old. Thawed plasma samples were filtered through a 0.45-μm Millipore filter to remove precipitated fibrinogen.
Test Substances/Materials
Radiolabeled 14C-deltamethrin (DLM) (57.9 mCi/mmol) of 99% purity was kindly provided by Bayer CropScience (Stilwell, KS), as was unlabeled DLM of 98.9% purity. N-Methyl-14C-diazepam (55 mCi/mmol) and β-3H-cyclosporine (20 Ci/mmol) were obtained from American Radiolabeled Chemicals (St. Louis, MO). Glycerol formal, acetonitrile (HPLC-grade), hexamethyldisilazane (Reagent-grade), and sodium fluoride (NaF) (purity, 99.0%) were purchased from Sigma Aldrich (St. Louis, MO). Isooctane (purity, 99.0%) and 2-octanol (laboratory grade) were purchased from Fisher Scientific (Pittsburgh, PA).
Albumin and Total Plasma Protein Measurement
Albumin levels in human plasma were measured with a commercially available kit (QuantiChrom BCG Albumin Assay Kit; BioAssay Systems, Hayward, CA) according to the manufacturer’s instructions. Total protein estimations in human plasma were performed using a BCA protein assay kit (Pierce/Thermo Scientific, Rockford, IL) by following the directions of the manufacturer.
DLM Binding Experiment Precautions
Standard plasma binding measurement techniques, such as equilibrium dialysis and ultracentrifugation, are not suitable for highly lipophilic compounds such DLM. DLM and other pyrethroids are practically insoluble in the aqueous media used. Pyrethroids adhere avidly to glass, metal and many polymer components of separation devices. Thus, it was essential to utilize clean glassware silanized with 5% hexamethyldisilazane in toluene, at least 24 h before use in experiments. LoBind plastic pipette tips (Eppendorf, Hamburg, Germany) were used to avoid pyrethroid adherence.
Plasma Protein and Lipoprotein Binding of DLM
Binding of DLM to plasma components was quantified by the three-step organic solvent extraction procedure of Sethi et al. (18). Each 80-μl aliquot of plasma was spiked with 10 μl of 14C-DLM in a silanized glass vial to yield a final concentration of 250 nmol/l. Eighty microliters of HSA (40 mg/ml) were similarly spiked with 14C-DLM to produce a 250 nmol/l solution. This concentration was found in the plasma of DLM-dosed rats that exhibit signs of neurotoxicity (40). The spiked plasma solutions were immediately treated with 10 μl of 0.64 M NaF to inhibit serum carboxylesterases, and incubated in an orbital shaker at pH 7.4 for 3 h at 37 °C. The samples were then extracted in turn with 200 μl of isooctane, 2-octanol and acetonitrile, which extracted the unbound, lipoprotein-bound and protein-bound pyrethroid fractions, respectively. DLM concentrations were measured by liquid scintillation counting using a Beckman Coulter LS 6500 instrument.
Plasma Binding of Diazepam and Cyclosporine
Plasma binding of diazepam and cyclosporine were determined by equilibrium dialysis, using a Dispo Equilibrium Dialyzer purchased from Harvard Apparatus (Holliston, MA,). Each 75-μl aliquot of plasma was spiked with 14C-diazepam to yield a 200 ng/ml solution. This was dialyzed against isotonic phosphate buffer at 37 °C for 4 h to establish equilibrium. A 75-μl aliquot of plasma was spiked with 3H-cyclosporine to yield a 1 μg/ml solution. This solution was dialyzed against isotonic phosphate buffer at 37 °C for 18 h to reach equilibrium. No fluid shifts occurred during the incubation. After the specified times, 100-μl aliquots of plasma and buffer were removed simultaneously and mixed with 3 ml of scintillation fluid (MP Biochemicals, Solon, OH). Radioactivity in each sample was quantified in a Beckman LS 6500 scintillation counter. The extent of binding was calculated using the following formula:
% bound fraction = plasma dpm − buffer dpm × 100/plasma dpm
% free fraction = 1 − % bound fraction
Statistical Analysis
Data were analyzed using ANOVA coupled with Tukey’s multiple comparison’s test using Prism 6.4 (GraphPad, San Diego, CA).
Statement of Financial Support
This study was supported in part by the Council for Advancement of Pyrethroid Human Risk Assessment (CAPHRA), 1667 K Street NW, Suite 300, Washington, DC 20006. CAPHRA did not play a role in: the study design; collection, analysis, and interpretation of data; writing of the report; or the decision to submit the manuscript for publication. No honorarium, grant, or other form of payment was received to produce the manuscript.
References
Kearns GL, Abdel-Rahman SM, Alander SW, Blowey DL, Leeder JS, Kauffman RE. Developmental pharmacology–drug disposition, action, and therapy in infants and children. N Engl J Med 2003;349:1157–67.
Berezhkovskiy LM. On the influence of protein binding on pharmacological activity of drugs. J Pharm Sci 2010;99:2153–65.
Schmidt S, Gonzalez D, Derendorf H. Significance of protein binding in pharmacokinetics and pharmacodynamics. J Pharm Sci 2010;99:1107–22.
Greenblatt DJ, Sellers EM, Koch-Weser J. Importance of protein binding for the interpretation of serum or plasma drug concentrations. J Clin Pharmacol 1982;22:259–63.
Trainor GL. The importance of plasma protein binding in drug discovery. Expert Opin Drug Discov 2007;2:51–64.
Dasgupta A. Usefulness of monitoring free (unbound) concentrations of therapeutic drugs in patient management. Clin Chim Acta 2007;377:1–13.
Nau H, Luck W, Kuhnz W. Decreased serum protein binding of diazepam and its major metabolite in the neonate during the first postnatal week relate to increased free fatty acid levels. Br J Clin Pharmacol 1984;17:92–8.
Fournier T, Medjoubi-N N, Porquet D. Alpha-1-acid glycoprotein. Biochim Biophys Acta 2000;1482:157–71.
Wasan KM, Brocks DR, Lee SD, Sachs-Barrable K, Thornton SJ. Impact of lipoproteins on the biological activity and disposition of hydrophobic drugs: implications for drug discovery. Nat Rev Drug Discov 2008;7:84–99.
Grandison MK, Boudinot FD. Age-related changes in protein binding of drugs: implications for therapy. Clin Pharmacokinet 2000;38:271–90.
Alcorn J, McNamara PJ. Pharmacokinetics in the newborn. Adv Drug Deliv Rev 2003;55:667–86.
McNamara PJ, Alcorn J. Protein binding predictions in adults. AAPS Pharm Sci 2002;4:1–8.
Holt DW, Hayler AM, Healey GF. Effect of age and plasma concentrations of albumin and alpha 1-acid glycoprotein on protein binding of disopyramide. Br J Clin Pharmacol 1983;16:344–5.
Kanakoudi F, Drossou V, Tzimouli V, et al. Serum concentrations of 10 acute-phase proteins in healthy term and preterm infants from birth to age 6 months. Clin Chem 1995;41:605–8.
Lerman J, Strong HA, LeDez KM, Swartz J, Rieder MJ, Burrows FA. Effects of age on the serum concentration of alpha 1-acid glycoprotein and the binding of lidocaine in pediatric patients. Clin Pharmacol Ther 1989;46:219–25.
Heudorf U, Angerer J, Drexler H. Current internal exposure to pesticides in children and adolescents in Germany: urinary levels of metabolites of pyrethroid and organophosphorus insecticides. Int Arch Occup Environ Health 2004;77:67–72.
Barr DB, Olsson AO, Wong LY, et al. Urinary concentrations of metabolites of pyrethroid insecticides in the general U.S. population: National Health and Nutrition Examination Survey 1999-2002. Environ Health Perspect 2010;118:742–8.
Sethi PK, Muralidhara S, Bruckner JV, White CA. Measurement of plasma protein and lipoprotein binding of pyrethroids. J Pharmacol Toxicol Methods 2014;70:106–11.
Soderlund DM. Molecular mechanisms of pyrethroid insecticide neurotoxicity: recent advances. Arch Toxicol 2012;86:165–81.
Divoll M, Greenblatt DJ. Binding of diazepam and desmethyldiazepam to plasma protein: concentration-dependence and interactions. Psychopharmacology (Berl) 1981;75:380–2.
Sgoutas D, MacMahon W, Love A, Jerkunica I. Interaction of cyclosporin A with human lipoproteins. J Pharm Pharmacol 1986;38:583–8.
Gillberg C. “Floppy infant syndrome” and maternal diazepam. Lancet 1977;2:244.
Warner A. Drug use in the neonate: interrelationships of pharmacokinetics, toxicity, and biochemical maturity. Clin Chem 1986;32:721–7.
Bolatt RJ. Effect of maternal diazepam on the newborn. Br J Anaesth 1978:1:985.
Brodersen R, Honoré B. Drug binding properties of neonatal albumin. Acta Paediatr Scand 1989;78:342–6.
Treluyer JM, Gueret G, Cheron G, Sonnier M, Cresteil T. Developmental expression of CYP2C and CYP2C-dependent activities in the human liver: in-vivo/in-vitro correlation and inducibility. Pharmacogenetics 1997;7:441–52.
Stevens JC, Hines RN, Gu C, et al. Developmental expression of the major human hepatic CYP3A enzymes. J Pharmacol Exp Ther 2003;307:573–82.
Koukouritaki SB, Manro JR, Marsh SA, et al. Developmental expression of human hepatic CYP2C9 and CYP2C19. J Pharmacol Exp Ther 2004;308:965–74.
Kallio MJ, Salmenperä L, Siimes MA, Perheentupa J, Miettinen TA. Exclusive breast-feeding and weaning: effect on serum cholesterol and lipoprotein concentrations in infants during the first year of life. Pediatrics 1992;89(4 Pt 1):663–6.
Asayama K, Miyao A, Kato K. High-density lipoprotein (HDL), HDL2, and HDL3 cholesterol concentrations determined in serum of newborns, infants, children, adolescents, and adults by use of a micromethod for combined precipitation ultracentrifugation. Clin Chem 1990;36:129–31.
Lu C, Barr DB, Pearson M, Bartell S, Bravo R. A longitudinal approach to assessing urban and suburban children’s exposure to pyrethroid pesticides. Environ Health Perspect 2006;114:1419–23.
Borlakoglu JT, Welch VA, Edwards-Webb JD, Dils RR. Transport and cellular uptake of polychlorinated biphenyls (PCBs)–II. Changes in vivo in plasma lipoproteins and proteins of pigeons in response to PCBs, and a proposed model for the transport and cellular uptake of PCBs. Biochem Pharmacol 1990;40:273–81.
Gómez-Catalán J, To-Figueras J, Rodamilans M, Corbella J. Transport of organochlorine residues in the rat and human blood. Arch Environ Contam Toxicol 1991;20:61–6.
Espié P, Tytgat D, Sargentini-Maier ML, Poggesi I, Watelet JB. Physiologically based pharmacokinetics (PBPK). Drug Metab Rev 2009;41:391–407.
Anderson ME. Development of physiologically based pharmacokinetic and physiologically based pharmacokinetic models for application in toxicology risk management. Toxicol Lett 1995; 79: 35–44.
Lipscomb JC, Haddad S, Poet T, Krishnan K. Physiologically-based pharmacokinetic (PBPK) models in toxicity testing and risk assessment. Adv Exp Med Biol 2012;745:76–95.
Rowland M, Peck C, Tucker G. Physiologically-based pharmacokinetics in drug development and regulatory science. Annu Rev Pharmacol Toxicol 2011;51:45–73.
Jones HM, Dickins M, Youdim K, et al. Application of PBPK modelling in drug discovery and development at Pfizer. Xenobiotica 2012;42:94–106.
Shardlow CE, Generaux GT, Patel AH, Tai G, Tran T, Bloomer JC. Impact of physiologically based pharmacokinetic modeling and simulation in drug development. Drug Metab Dispos 2013;41:1994–2003.
Kim KB, Anand SS, Kim HJ, et al. Age, dose, and time-dependency of plasma and tissue distribution of deltamethrin in immature rats. Toxicol Sci 2010;115:354–68.
Sethi PK, White CA, Cummings BS, Hines RN, Muralidhara S, Bruckner, JV. Ontogeny of plasma proteins, albumin and binding of diazepam, cyclosporine and deltamethrin. Ped Res, in press.
Smith JN, Timchalk C, Bartels MJ, Poet TS. In vitro age-dependent enzymatic metabolism of chlorpyrifos and chlorpyrifos-oxon and human hepatic microsomes and chlorpyrifos-oxon in plasma. Drug Metab Dispos 2011;39:1353–62.
Pickoff AS, Kessler KM, Singh S, et al. Age-related differences in the protein binding of quinidine. Dev Pharmacol Ther 1981;3:108–115.
Meistelman C, Benhamou D, Barre J, et al. Effects of age on plasma protein binding of sufentanil. Anesthesiology 1990;72:470–73.
Mazoit JX, Denson DD, Samii K. Pharmacokinetics of bupivacaine following caudal anesthesia in infants. Anesthesiology 1988;68:387–91.
Acknowledgements
The authors would like to thank Leslie Standridge for the careful preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Sethi, P., White, C., Cummings, B. et al. Ontogeny of plasma proteins, albumin and binding of diazepam, cyclosporine, and deltamethrin. Pediatr Res 79, 409–415 (2016). https://doi.org/10.1038/pr.2015.237
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2015.237
This article is cited by
-
Challenges of pediatric pharmacotherapy: A narrative review of pharmacokinetics, pharmacodynamics, and pharmacogenetics
European Journal of Clinical Pharmacology (2024)
-
Overview of Albumin Physiology and its Role in Pediatric Diseases
Current Gastroenterology Reports (2021)
-
Allometric Scaling of Clearance in Paediatric Patients: When Does the Magic of 0.75 Fade?
Clinical Pharmacokinetics (2017)