Abstract
Fibroepithelial lesions of the breast, comprising the fibroadenoma and phyllodes tumour, are a unique group of neoplasms that share histological characteristics but possess different clinical behaviour. The fibroadenoma is the commonest benign breast tumour in women, while the phyllodes tumour is rare and may be associated with recurrences, grade progression and even metastasis. The diagnosis of fibroadenoma is usually straightforward, with recognised histological variants such as the cellular, complex, juvenile and myxoid forms. The phyllodes tumour comprises benign, borderline and malignant varieties, graded using a constellation of histological parameters based on stromal characteristics of hypercellularity, atypia, mitoses, overgrowth and the nature of tumour borders. While phyllodes tumour grade correlates with clinical behaviour, interobserver variability in assessing multiple parameters that are potentially of different biological weightage leads to significant challenges in accurate grade determination and consequently therapy. Differential diagnostic considerations along the spectrum of fibroepithelial tumours can be problematic in routine practice. Recent discoveries of the molecular underpinnings of these tumours may have diagnostic, prognostic and therapeutic implications.
Similar content being viewed by others
Introduction
Breast fibroepithelial lesions are biphasic neoplasms composed of both epithelial and stromal components, comprising the common fibroadenoma and the less frequently occurring phyllodes tumour [1].
While the diagnosis of fibroadenoma is made relatively often, especially in core biopsies, the phyllodes tumour is a less commonly encountered pathological conclusion, with particular challenges in grading as well as distinction from histological mimics. These differential diagnoses include the cellular fibroadenoma at the benign end of the phyllodes tumour spectrum, to metaplastic spindle-cell carcinoma and primary breast sarcoma at the borderline and malignant extreme.
In this review, the pathology of the fibroadenoma and phyllodes tumour is revisited, with emphasis on diagnostic and management implications. Molecular information that has emerged in recent years will also be highlighted especially in relation to diagnosis and prognosis.
Fibroadenoma
The fibroadenoma is the commonest benign tumour of the breast, occurring most frequently in women of reproductive age group. Clinically symptomatic patients present with round to ovoid painless breast lumps, which are smooth and rubbery in consistency, often slipping away during palpation, hence referred to as ‘breast mice’. Asymptomatic fibroadenomas are also often discovered in older women during mammographic screening, observed radiologically as masses or calcifications. Core biopsy or fine needle aspiration cytology confirmation of fibroadenoma allows avoidance of surgery, unless symptoms and/or rapid growth warrant removal.
Grossly, the fibroadenoma shows rounded to lobulated, variably encapsulated borders, with fibrous to myxoid cut surfaces. Microscopically, it is a biphasic tumour with circumscribed and pushing contours, composed of an admixture of epithelial and stromal elements, with loss of lobular architecture due to the expansion of stroma between epithelial elements. The pericanalicular growth pattern refers to stromal growth around patent tubules, while the intracanalicular appearance comprises stroma pushing against epithelium creating arc-like epithelial shapes (Fig. 1). Both patterns are often seen in the same lesion, which are without clinical significance, apart from recent recognition that the MED12 mutation is more frequently found in the intracanalicular fibroadenoma [2, 3].
A variety of histological changes can be seen in the fibroadenoma. Infarction may occur in pregnant patients and post-instrumentation (Fig. 2). The fibroadenoma stroma is usually of low cellularity, with myxoid, fibroblastic or hyalinised appearances. Stromal multinucleated cells [4,5,6], calcifications, ossification and pseudoangiomatous stromal hyperplasia (PASH) may be present. The epithelium can display usual ductal hyperplasia (UDH), reported to occur in 32.3% of cases (excluding mild hyperplasia) [7], apocrine metaplasia, sclerosing adenosis, atypical ductal hyperplasia (ADH), atypical lobular hyperplasia (ALH), ductal carcinoma in situ, lobular carcinoma in situ and even invasive carcinoma [8] (Fig. 3), although the cancer rate in fibroadenoma is exceedingly rare, from 0.002 to 0.125% [9]. Carter et al. found a 0.81% prevalence of ALH or ADH in fibroadenomas, which when confined within the fibroadenoma, does not translate to a clinically meaningful increased risk of subsequent breast cancer development [10].
Fibroadenoma variants
Fibroadenoma variants include cellular, complex, juvenile and myxoid forms. The cellular variant shows increased density of stromal cells within the architecture of a typical fibroadenoma, without significant stromal atypia, excess stromal mitotic activity or accentuated intracanalicularity (Fig. 4). The main differential diagnosis of the cellular fibroadenoma, especially on core biopsy, is the phyllodes tumour, which is distinguished by the presence of well-formed stromal fronds. In a long-term follow-up study conducted on a series of cellular fibroepithelial lesions that included 35 cellular fibroadenomas, none of which were widely excised, it was concluded that the recurrence rate of these tumours was low, without any phyllodes tumours diagnosed among the recurrences [11]. Genomically, cellular fibroadenomas possessed similar rates of mutations in the most commonly mutated genes MED12, KMT2D and RARA (49%, 13% and 13%) as conventional fibroadenomas (44%, 15% and 8%), indirectly supporting their classification with conventional fibroadenomas [12]. In contrast, the mutation spectrum of benign phyllodes tumours with which they resemble disclosed 62%, 14% and 17% abnormalities in the same set of genes, with a significant difference in the MED12 mutation rate. In addition, TERT promoter mutations were significantly higher in benign phyllodes tumours (32%) than in cellular (4%) and conventional (6%) fibroadenomas [12].
The complex fibroadenoma comprises 14.1–40.4% of all fibroadenomas [7, 13, 14]. It shows any of the following histological features: sclerosing adenosis, papillary apocrine metaplasia, cysts ≥3 mm in size and epithelial calcifications [15] (Fig. 5). Studies indicate a mildly increased risk of about 3× that of the general population of subsequent breast cancer development [13, 15], though it is uncertain if this higher probability is independent of epithelial proliferative changes within the fibroadenoma. This slight increase in risk associated with the complex fibroadenoma does not portend a clinically actionable management impact, further supported by a report that suggests that the complex fibroadenoma is not an independent risk marker for breast cancer [13].
The juvenile fibroadenoma is often described in children and adolescents, but may be diagnosed in women of any age. It can grow to a large size, with lesions exceeding 5 cm in size regarded as giant fibroadenomas [1]. The stroma tends to be cellular, displaying an interlacing fascicular arrangement of fibroblasts and myofibroblasts with a pericanalicular pattern (Fig. 6). The epithelium usually demonstrates moderate to florid UDH, the latter sometimes causing confusion with ADH. The epithelial proliferation may disclose gynaecomastoid features with fine filigree-like, narrow micropapillary epithelial protrusions. Four patterns, referred to as ‘atypical epithelial proliferations’ in juvenile fibroadenomas, were described decades ago: ductal-laciform, ductal-solid, cystic-papillary and lobular-terminal ductal; a conservative interpretation was recommended [16], and one would caution against overdiagnosing malignancy in these cases.
The myxoid fibroadenoma is typified by loose hypocellular stroma containing watery myxoid ground substance (Fig. 7), with some cases being part of the Carney’s complex [17], an autosomal dominant disorder characterised by myxomas in different sites, spotty pigmentation and endocrine overactivity. Genomically, the myxoid fibroadenoma differs from the conventional fibroadenoma by its lack of MED12 mutations [18].
Fibroadenomas in the paediatric population
Fibroadenomas in the paediatric population have not been extensively investigated pathologically. A radiological study concluded that fibroadenomas comprised 91% of all histologically evaluated solid breast masses among patients under 19 years of age [19]. Two major morphological studies reviewed microscopic findings with a note of increased stromal cellularity, which may be marked, especially in juvenile fibroadenomas [20], and mitoses that numbered up to 6 mitoses per 10 high-power fields in usual fibroadenomas [21]. Stromal fronds could also be identified [20] although they tended to be less well developed (Fig. 8), and were not associated with recurrences. Stromal nuclear atypia, up to moderate degree, was discovered [20]. It was acknowledged that traditional histological parameters and thresholds used for adult fibroepithelial tumours, when applied to lesions in young patients, could pose challenges. The juvenile fibroadenoma was the commonest form of fibroadenoma in the paediatric age group, with additional observations of slight stromal expansion and intratumoural heterogeneity without stromal atypia [21].
Both studies concurred that fibroadenomas in the young followed a benign course, without predisposition to phyllodes tumour or cancer development. Genomically, TERT promoter mutations which are found more frequently in phyllodes tumours, were not detected in paediatric fibroadenomas [22, 23]. A conservative and cautious approach is therefore recommended in the diagnosis and treatment of paediatric fibroepithelial tumours.
Core biopsy diagnosis
Core biopsy is a standard preoperative diagnostic procedure for breast lesions discovered clinicoradiologically. Fibroadenomas are commonly diagnosed on core biopsy material. A question that is sometimes raised is whether a conclusion of fibroadenoma on core biopsy is accurate and reliable, and whether there should be concern for undersampling of a phyllodes tumour. In a multicentre study incorporating routine diagnoses of fibroadenomas on core biopsies with follow-up, it was found that subsequent discovery of phyllodes tumour is extremely rare, with only 16 (0.38%) out of a total of 4163 cases [24]. These phyllodes tumours were categorised as benign in 14 cases and borderline in 2 cases. It was concluded that the main reason contributing to the core needle biopsy-excision discrepancies was phyllodes tumour heterogeneity with fibroadenoma-like areas, with such foci being discovered in 35.9% of phyllodes tumours in one study [25]. Unfortunately, there were no specific pathological features that were prospectively predictive of phyllodes tumour at excision, but that suspicious imaging features at the time of core needle biopsy or on follow-up should prompt consideration for surgical excision. In addition, all core biopsy diagnoses should be reviewed in the context of the triple approach with clinical and radiological input. Clinically symptomatic and large lesions, and rapid tumour growth are triggers for excision, obviating sampling issues. The key message from the study was that the diagnosis of fibroadenoma on core needle biopsy is reliable and safe in the setting of the triple approach.
Core biopsies of cellular fibroepithelial lesions represent a challenging area, with multiple studies having been conducted to determine factors that could predict phyllodes tumours on excision. Table 1 summarises the data from various publications, with a constellation of histological features including mitotic activity (suggested as 2 or more per 10 high-power fields), marked stromal hypercellularity, stromal overgrowth (variably defined in different studies), adipose infiltration, ill-defined lesional borders, heterogeneity, subepithelial condensation, stromal nuclear pleomorphism, tissue fragmentation, as well as older age group, that were correlated with a phyllodes tumour outcome [26,27,28,29,30,31]. Immunohistochemistry for proliferation markers Ki-67 and topoisomerase 2α was also described as being informative, though this has not been applied diagnostically as thresholds in individual cases are not yet determined [26, 29].
With availability of molecular pathology, authors have attempted to use transcriptomic and genomic tools to assist in distinguishing fibroadenomas from phyllodes tumours on core biopsies. In the former, a 5-gene transcript was accurate in discriminating the two lesions in 92.6% of cases using a reverse-transcription polymerase chain assay [32] whereas in the latter, targeted sequencing of a 16-gene panel allowed development of a risk-scoring system that stratified core biopsies of fibroepithelial lesions into low and high risks of being a phyllodes tumour [33]. Application of digital pathology was unable to enhance diagnostic discrimination [34].
Phyllodes tumour
The phyllodes tumour of the breast is a biphasic neoplasm with an exaggerated, prominent intracanalicular growth pattern with leaf-like stromal fronds covered by benign bilayered epithelium (luminal and myo-epithelium) [1]. It affects females in their 5th decade or older, though it can occur in younger women, especially of Asian and Hispanic ethnicity. While the tumour tends to present symptomatically as a large mass which may exceed 10 cm and distort the breast, the average tumour size is 4–5 cm, with smaller tumours being detected radiologically.
Macroscopically, phyllodes tumours show circumscribed bosselated contours with a variety of appearances—whitish and whorled, grey and fleshy, soft and mucoid, with areas of necrosis and haemorrhage (Fig. 9). Cystic changes may also occur.
Microscopically, the key histological findings are broad patulous fronds of at least mildly cellular stroma covered by benign bilayered epithelium, forming protuberant projections into clefted compressed spaces. Once the diagnosis of phyllodes tumour is made, grading into benign, borderline and malignant categories is accomplished based on an assessment of a constellation of histological criteria—degree of stromal hypercellularity, stromal atypia, stromal mitoses, stromal overgrowth and the nature of the tumour borders (pushing or permeative) [1]. Like in the fibroadenoma, the epithelial component may display usual and atypical hyperplasia (both ductal and lobular), in situ and invasive carcinoma [35], with PASH more often encountered in the phyllodes tumour than in the fibroadenoma (personal observation). Among phyllodes tumours, PASH has also been noticed more frequently in the benign grade [36].
Grading of phyllodes tumours
Phyllodes tumours are graded into benign, borderline and malignant forms. Grade is important due to its correlation with clinical behaviour, in particular with local recurrences and metastases. Table 2 shows the histological grading parameters within the different grades.
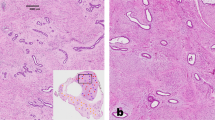
The benign phyllodes tumour displays mild stromal hypercellularity with nil to mild stromal atypia, with scant mitoses numbering up to 4 per 10 high-power fields, without stromal overgrowth [1]. Borders are pushing and smooth contoured (Fig. 10). Peri-epithelial stromal condensation and elongated narrow epithelium-lined clefts may be seen (Fig. 11). The latter may be a clue to a phyllodes tumour diagnosis, with more characteristic phyllodal architecture found elsewhere in the tumour upon thorough search. At the other end of the grading spectrum is the malignant phyllodes tumour, which shows marked and diffuse stromal hypercellularity, marked stromal atypia, brisk mitotic activity exceeding 9 per 10 high-power fields, stromal overgrowth defined as one low-power field (×40 magnification, at ×4 objective with ×10 eyepiece) containing stroma only without any epithelial elements, as well as permeative borders (Fig. 12). Presence of malignant heterologous elements, except well differentiated liposarcoma, warrants a malignant grade even in the absence of other histological parameters indicating malignancy [1]. Straddling between the benign and malignant phyllodes tumours is the borderline grade, which shows moderate stromal hypercllularity, mild to moderate stromal atypia, 5–9 mitoses per 10 high-power fields and focally permeative borders. Stromal overgrowth can be present focally. No malignant heterologous elements are seen (Fig. 13).
a Peri-epithelial or subepithelial stromal condensation is seen as an aggregation of stromal cells hugging the epithelium, which at low magnification may be discerned as a ‘shadow’ around the epithelial component. b Elongated clefts lined by benign epithelium may be a clue to more diagnostic phyllodal areas.
The exclusion of liposarcoma from the list of malignant heterologous elements that can individually indicate a malignant grade was a consensus decision made by the working group in the WHO 2019 breast tumour classification. It was the collective view that liposarcoma in the breast, and in particular the phyllodes tumour, did not harbour metastatic potential. This assessment was supported by the absence of MDM2 and CDK amplifications in liposarcoma of phyllodes tumours [37,38,39,40], in contrast to their presence in extramammary liposarcoma, though there was one case with focal MDM2 immunopositivity [38]. In addition, genomic studies found that the non-heterologous component of malignant phyllodes tumours displayed more chromosomal aberrations than the liposarcoma element [37]. It is recommended that evaluation of other histological parameters is used to finalise the grade in phyllodes tumours harbouring liposarcoma. The presence of liposarcoma, however, tends to be associated with microscopic features of at least borderline grade (Fig. 14). It is important to recognise that benign adipocytic components can be seen in phyllodes tumours [41, 42] and do not impact grading. Focal fat necrosis may result in adipocytes and histiocytes with reactive nuclear atypia and cytoplasmic vacuolation that should not be interpreted as lipoblasts nor cause consternation for liposarcoma.
a Gross specimen of phyllodes tumour with whitish whorled and yellowish soft mucoid areas. Fronds and clefts are present. b Low magnification shows a hypercellular stromal frond with abnormal cells. c Marked stromal atypia and a mitosis. d Lipoblasts with hyperchromatic scalloped nuclei and vacuolated cytoplasm.
In order to rationalise terminology for cases where the final grade is determined as borderline rather than malignant based on other histological parameters, the liposarcoma foci could be referred to as ‘lipoblast-like areas’ to avoid using the term ‘sarcoma’ for phyllodes tumours that are not diagnosed as malignant. Presence of other malignant heterologous elements like osteosarcoma, chondrosarcoma and rhabdomyosarcoma remains diagnostic of malignant phyllodes tumours. As phyllodes tumours containing heterologous elements are rare, it would be helpful for their continued study to establish the true clinical significance. Koh et al. reported that large phyllodes tumours containing malignant heterologous elements, which included liposarcoma, were predictive of metastatic likelihood [43]. There are also anecdoctal reports of myxoid [44] and pleomorphic [45] liposarcoma in phyllodes tumours, which may be accompanied by aggressive behaviour [38].
Sampling of phyllodes tumours
Phyllodes tumours often display intratumoural heterogeneity. High grade areas may be focal within otherwise low-grade tumours, and stromal overgrowth may mask fibroepithelial architecture. Genomic heterogeneity parallels morphological diversity [46]. Adequate sampling is therefore important. One block per cm of maximum tumour dimension, with sampling of grossly heterogenous areas, is recommended.
Classification and grading challenges
Phyllodes tumour classification and grading are inherently imperfect, due to the need to amalgamate the assessment of multiple histological criteria, for which their relative importance to clinical outcome is not addressed in the current grading scheme. Each histological parameter also has several tiers of stratification and multiple combinational permutations, invariably leading to interobserver variability [47]. In order to circumvent these deficiencies, a study of 605 phyllodes tumours from a single institution was conducted to determine the relative impact of histological grading parameters to recurrence, with development of a formula that takes into account weighting of the criteria [47]. It was found that stromal atypia, mitotic activity, overgrowth and status of surgical margins were key parameters that correlated with recurrent likelihood (AMOS criteria). This Singapore nomogram has been validated in a few studies [25, 48,49,50], and may be accessed through an online calculator (https://mobile.sgh.com.sg/ptrra/), which can be used to counsel individual patients with phyllodes tumours.
Prognosis of phyllodes tumours
While grading of phyllodes tumours has its limitations, the benign, borderline and malignant grade groups are prognostically discriminatory with different recurrence rates. Local recurrence rates are 10–17%, 14–25% and 23–30%, respectively, for benign, borderline and malignant tumours [1], indicating greater recurrences with increasing tumour grades. Grade progression upon recurrence is shown in Fig. 15 with 43.7% of originally benign tumours recurring at higher grades based on a study conducted on 605 cases [47] with a small handful (8.3%) progressing directly to malignancy from an initially benign tumour.
Metastases are reported to occur in 0.1%, 1.6% and 16.7% of benign, borderline and malignant phyllodes tumours, respectively [1]. The anecdotal case reports of metastases of benign tumours need to be critically appraised with regard to grading accuracy and tumour sampling [51]. While occasional borderline tumours are known to metastasise, whether such tumours were appropriately graded also requires review. Metastases are almost exclusively encountered in phyllodes tumours of malignant grade, occurring in up to 2% overall among all phyllodes tumours, with metastatic lesions comprising malignant stroma devoid of epithelium [1, 51, 52]. Predictors of metastases include age >50 years, stromal overgrowth, diffuse marked atypia, necrosis, mitoses ≥10/10 high-power fields [53]; large tumours (>9 cm) with heterologous elements [43]. Continued research into subsets of malignant tumours that are likely to metastasise will help in stratifying therapy.
Differential diagnosis
Diagnostic challenges revolve around the distinction of benign phyllodes tumour from the cellular fibroadenoma, while at the malignant end of the spectrum, separating malignant phyllodes tumour from spindle-cell metaplastic breast carcinoma and sarcoma. Other differential considerations include periductal stromal tumour, fibromatosis and metastases [54].
Benign phyllodes tumour and cellular fibroadenoma
The key histological feature that distinguishes these two entities is the presence of an exaggerated intracanalicular growth pattern, or prominent stromal fronds, in the phyllodes tumour. Both lesions show increased stromal cellularity and overlapping mitotic rates. While mild stromal atypia may be acceptable in the cellular fibroadenoma, greater degrees of atypia should raise consideration for phyllodes tumour. Stromal multinucleated cells may be observed in both lesions, and the general advice is not to overinterpret their presence, even when they appear bizarre as this may be of degenerative nature [5, 55] (Fig. 16). It would be prudent, however, to evaluate the non-multinucleated stromal-cell population in such cases, as lesions with significant stromal atypia including abnormal mitoses have been observed warranting at least a borderline grade (Fig. 17). Immunohistochemistry for Ki-67 and p53 may be useful in grading calibration [56,57,58].
a Gross appearance of the benign tumour with circumscribed borders and a fibrous myxoid cut-surface. b Stroma in between the epithelial component shows low cellularity with scattered enlarged stromal cells. c High magnification of the abnormal stromal cells with multilobated nuclei and nuclear inclusions. Spindled stromal cells present are without atypia or mitoses.
a Gross appearance of the phyllodes tumour with clefts and myxoid fronds. Yellowish areas correspond to infarction. b Low magnification shows phyllodal architecture with stromal fronds. Inset reveals a quadripolar mitosis. c, d Stromal cells among the giant forms display atypia and scattered mitoses.
In some cases, it may be very difficult to be absolutely certain if a tumour is a cellular fibroadenoma or benign phyllodes tumour, and in such circumstances, it may be appropriate to use the term ‘benign fibroepithelial neoplasm’ as a reflection of the overlapping histological features [51, 59].
Benign phyllodes tumour and periductal stromal tumour
The periductal stromal tumour differs histologically from phyllodes tumour by the absence of stromal leaf-like fronds [60]. These tumours are closely related [61], in that periductal stromal tumour-like areas are sometimes observed in phyllodes tumours, and some recurrences of periductal stromal tumours are diagnosed as phyllodes tumours [1, 59]. In addition, there is a genomic similarity of these lesions [46], with the latest WHO breast tumour classification regarding the periductal stromal tumour as a subtype of phyllodes tumour [1].
Borderline phyllodes tumour and fibromatosis
Stroma predominant phyllodes tumour, often in the setting of borderline grade, may resemble fibromatosis (Fig. 18). Finding stromal fronds on histology allows the correct diagnosis. Presence of periductal stromal condensation and narrow elongated clefted ducts should raise suspicion of a phyllodes tumour. Immunohistochemistry for CD34 generally shows stromal positivity in phyllodes tumours, with a higher rate in the benign grade [62,63,64], whereas it is negative in fibromatosis [65, 66]. Nuclear beta-catenin, often described as a diagnostic feature for fibromatosis, is also observed in phyllodes tumours, so it cannot be used for discriminating these two lesions [65, 67,68,69]. Fibromatosis-like metaplastic carcinoma (Fig. 19) is positive for epithelial markers on immunohistochemistry—nuclear beta-catenin can also be expressed [67, 70].
a Gross appearance shows a rounded firm nodule within breast tissue. b Low magnification shows intersecting spindle-cell fascicles with an ill-defined border, which may resemble stroma predominant phyllodes tumour. c. High magnification shows bland spindle cells. Inset reveals immunohistochemical nuclear and cytoplasmic reactivity for beta-catenin, though phyllodes tumour stromal cells may also be positive for beta-catenin.
a Irregular greyish-yellow tumour within the breast tissue. b Low magnification shows a fibrosclerotic lesion with ill-defined borders, with a few lymphocytic aggregates at the periphery. c High magnification shows plump-spindled cells within a collagenous background, with minimal atypia. d CK14 immunohistochemistry shows diffuse positivity of spindle cells, indicating epithelial differentiation.
Malignant phyllodes tumour, metaplastic spindle-cell carcinoma and sarcoma
Malignant phyllodes tumours may have large areas of stromal overgrowth that overrun the epithelial compartment, effacing their characteristic stromal fronds, thus resembling spindle-cell metaplastic carcinoma and breast sarcoma histologically. Presence of accompanying in situ or invasive carcinoma, and immunohistochemical positivity for epithelial markers, support the diagnosis of metaplastic carcinoma. While immunostaining positivity is often diffuse, there can be variability observed for different antibodies, which highlights the need for using a panel rather than a single marker. Spindle-cell carcinoma invading into a fibroepithelial tumour may mimic a malignant phyllodes tumour (Fig. 20).
The distinction between sarcoma and malignant phyllodes tumour may not be so critical in view of the overlapping biologic and genomic characteristics [71,72,73]. Primary breast sarcoma is extremely rare, and it is suggested that many of these are likely phyllodes tumours in which the epithelial component was not identified. Metastatic sarcoma is a rare occurrence, but an appropriate clinical history and workup should be able to alert one to the diagnosis. Rare entities like melanoma can be distinguished through a combination of morphological recognition and immunohistochemistry.
Pitfalls of immunohistochemistry
Epithelial markers, comprising a variety of keratins (MNF116, AE1/3, Cam5.2, 34βE12, CK5/6 and CK14), are relied upon to confirm the diagnosis of metaplastic spindle-cell carcinoma. Phyllodes tumours, however, have been discovered to express keratins as well, patchily and focally, especially in the malignant grade [74]. Similarly, p63 and p40 that are expressed in metaplastic carcinoma, are also found in malignant phyllodes tumours [75] (Fig. 21). These have implications on interpretation especially in small biopsy samples, where focal immunohistochemical expression of these markers should not automatically lead to a diagnosis of metaplastic carcinoma.
Surgical margins
The mainstay of treatment of breast phyllodes tumours is complete surgical excision with negative margins [51]. The questions often posed are whether all breast phyllodes tumours have to be widely excised, and if so, what is an optimal surgical margin width. Reports have used 1, 10 and >10 mm as surgical margin distances, and found variable correlation with recurrences [51]. A meta-analysis reported positive surgical margins to be associated with local recurrence in malignant phyllodes tumours, with a trend for increased local recurrence in benign and borderline tumours [76]. An overall local recurrence rate of 20.4% was documented for borderline and malignant phyllodes tumours that were subjected to breast conserving surgery with negative margins, when data of various studies were combined [77]. Hence, a direct association between margin status and local recurrence remains uncertain. There is increasing evidence that benign tumours may not need to be widely excised (Table 3), with low recurrence rates observed after enucleation without negative margins [78,79,80,81,82]. Recurrent and malignant tumours, however, would require complete excision, and most authors would advocate that borderline tumours are widely excised as well. As there is no universal agreement on what constitutes a clear margin, a consensus review suggested that positive margins be regarded as tumour that extends to ink, or <1 mm away [51].
The role of adjuvant therapy is not established. While malignant and most borderline tumours may be offered radiotherapy, administration of systemic chemotherapy is considered on individual cases.
Molecular genetics
In 2014, recurrent MED12 mutations were reported in a series of fibroadenomas, found only within the stromal component [83]. Prior to this discovery, fibroadenomas were regarded as genomically quiescent with only sporadic reports of molecular changes [84]. MED12 mutations were subsequently soon documented in phyllodes tumours (Table 4) [2, 3, 12, 22, 23, 33, 37, 83, 85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103]. MED12 mutations were previously described only in the uterine leiomyoma, a benign tumour that is hormonally linked, extrapolating to a possible hormonal aetiology for the fibroadenoma and phyllodes tumour as well. MED12 is a gene located on the X chromosome, whose functions are shown in Fig. 22 [104, 105].
a The Mediator complex initiates transcription through binding with DNA and recruiting transcription factors and RNA polymerase II (RNA Pol II). A kinase module comprising cyclin-dependent kinase 8 (CDK8), Cyclin C, MED12 and MED13 regulates the Mediator complex by interfering in its association with RNA Pol II to repress transcription. MED12 is essential for assembly of the kinase module, binding Cyclin C-CDK8 to core Mediator and stimulating kinase activity. b Mutation in MED12 leads to transcriptional misregulation as Cyclin C-CDK8 binding and activation are compromised.
The studies led to a proposed pathogenesis for phyllodes tumours through the MED12 pathway, whereby mutations in the MED12 gene initiate the development of fibroadenoma, and progressive acquisition of additional gene abnormalities leads to the formation of phyllodes tumours, with cancer driver gene derangements being associated with borderline and malignant tumours. A MED12 wild-type progression pathway is also proposed (Fig. 23). The event triggering the progression from fibroadenoma to phyllodes tumour is exceptionally rare, in light of the frequency of fibroadenomas and the rarity of phyllodes tumours.
Apart from insights into pathogenesis, these genomic discoveries augment grading of phyllodes tumours and have potential clinical relevance by enhancing diagnoses. Phyllodes tumour may be differentiated from fibroadenoma with the presence of TERT promoter mutations [93], which are rarely encountered in fibroadenoma. The separation from other spindle-cell tumours may also be assisted through molecular interrogation, with presence of MED12 mutations leaning towards phyllodes tumour and away from metaplastic carcinoma [106, 107]. Discovery of candidate therapeutic targets in borderline/malignant phyllodes tumours such as PIK3CA activating mutations and EGFR amplifications may open additional treatment avenues. Prognostically, MED12 mutations are correlated with improved disease-free survival [88, 94].
Conclusion
In summary, fibroadenomas and phyllodes are a fascinating group of fibroepithelial tumours that share not only morphological appearances but also genomic changes that underpin their pathogenesis. Continued work to refine the prediction of recurrences, identify which tumours would recur with grade progression, as well as triggers of metastases, is warranted. Distinction from histological mimics requires a combination of morphological and adjunctive studies. The roles of the epithelium and epithelial–stromal interaction in the aetiology and pathogenesis remain relatively unexplored. As phyllodes tumours are uncommon, especially borderline and malignant grades, collective international efforts for combining knowledge will go a long way towards bridging information gaps for effective treatment.
References
WHO Classification of Tumours Editorial Board. Breast tumours. vol 2. 5th ed. France, Lyon: IARC; 2019.
Mishima C, Kagara N, Tanei T, Naoi Y, Shimoda M, Shimomura A, et al. Mutational analysis of MED12 in fibroadenomas and phyllodes tumors of the breast by means of targeted next-generation sequencing. Breast Cancer Res Treat. 2015;152:305–12.
Pfarr N, Kriegsmann M, Sinn P, Klauschen F, Endris V, Herpel E, et al. Distribution of MED12 mutations in fibroadenomas and phyllodes tumors of the breast—implications for tumor biology and pathological diagnosis. Genes Chromosomes Cancer. 2015;54:444–52.
Berean K, Tron VA, Churg A, Clement PB. Mammary fibroadenoma with multinucleated stromal giant cells. Am J Surg Pathol. 1986;10:823–7.
Powell CM, Cranor ML, Rosen PP. Multinucleated stromal giant cells in mammary fibroepithelial neoplasms. A study of 11 patients. Arch Pathol Lab Med. 1994;118:912–6.
Heneghan HM, Martin ST, Casey M, Tobbia I, Benani F, Barry KM. A diagnostic dilemma in breast pathology–benign fibroadenoma with multinucleated stromal giant cells. Diagn Pathol. 2008;3:33.
Kuijper A, Mommers EC, van der Wall E, van Diest PJ. Histopathology of fibroadenoma of the breast. Am J Clin Pathol. 2001;115:736–42.
Krishnamurthy K, Alghamdi S, Gyapong S, Kaplan S, Poppiti RJ. A clinicopathological study of fibroadenomas with epithelial proliferation including lobular carcinoma in-situ, atypical ductal hyperplasia, DCIS and invasive carcinoma. Breast Dis. 2019;38:97–101.
Wu YT, Chen ST, Chen CJ, Kuo YL, Tseng LM, Chen DR, et al. Breast cancer arising within fibroadenoma: collective analysis of case reports in the literature and hints on treatment policy. World J Surg Oncol. 2014;12:335.
Carter BA, Page DL, Schuyler P, Parl FF, Simpson JF, Jensen RA, et al. No elevation in long-term breast carcinoma risk for women with fibroadenomas that contain atypical hyperplasia. Cancer. 2001;92:30–6.
Yasir S, Nassar A, Jimenez RE, Jenkins SM, Hartmann LC, Degnim AC, et al. Cellular fibroepithelial lesions of the breast: a long term follow up study. Ann Diagn Pathol. 2018;35:85–91.
Md Nasir ND, Ng CCY, Rajasegaran V, Wong SF, Liu W, Ng GXP, et al. Genomic characterisation of breast fibroepithelial lesions in an international cohort. J Pathol. 2019;249:447–60.
Nassar A, Visscher DW, Degnim AC, Frank RD, Vierkant RA, Frost M, et al. Complex fibroadenoma and breast cancer risk: a Mayo Clinic Benign Breast Disease Cohort Study. Breast Cancer Res Treat. 2015;153:397–405.
Sklair-Levy M, Sella T, Alweiss T, Craciun I, Libson E, Mally B. Incidence and management of complex fibroadenomas. AJR Am J Roentgenol. 2008;190:214–8.
Dupont WD, Page DL, Parl FF, Vnencak-Jones CL, Plummer WD Jr., Rados MS, et al. Long-term risk of breast cancer in women with fibroadenoma. N Engl J Med. 1994;331:10–5.
Mies C, Rosen PP. Juvenile fibroadenoma with atypical epithelial hyperplasia. Am J Surg Pathol. 1987;11:184–90.
Carney JA, Toorkey BC. Myxoid fibroadenoma and allied conditions (myxomatosis) of the breast. A heritable disorder with special associations including cardiac and cutaneous myxomas. Am J Surg Pathol. 1991;15:713–21.
Lozada JR, Burke KA, Maguire A, Pareja F, Lim RS, Kim J, et al. Myxoid fibroadenomas differ from conventional fibroadenomas: a hypothesis-generating study. Histopathology. 2017;71:626–34.
Sanchez R, Ladino-Torres MF, Bernat JA, Joe A, DiPietro MA. Breast fibroadenomas in the pediatric population: common and uncommon sonographic findings. Pediatr Radio. 2010;40:1681–9.
Tay TK, Chang KT, Thike AA, Tan PH. Paediatric fibroepithelial lesions revisited: pathological insights. J Clin Pathol. 2015;68:633–41.
Ross DS, Giri DD, Akram MM, Catalano JP, Olcese C, Van Zee KJ, et al. Fibroepithelial lesions in the breast of adolescent females: a clinicopathological study of 54 cases. Breast J. 2017;23:182–92.
Tay TKY, Guan P, Loke BN, Nasir NDM, Rajasegaran V, Thike AA, et al. Molecular insights into paediatric breast fibroepithelial tumours. Histopathology. 2018;73:809–18.
Pareja F, Da Cruz Paula A, Murray MP, Hoang T, Gularte-Merida R, Brown D, et al. Recurrent MED12 exon 2 mutations in benign breast fibroepithelial lesions in adolescents and young adults. J Clin Pathol. 2019;72:258–62.
Jacobs TW, Chen Y-Y, Guinee DG, Eby PR, Thike AA, Vohra P, et al. Phyllodes tumor (PT) subsequent to a diagnosis of fibroadenoma (FA) on breast core needle biopsy (CNB): frequency and characteristics. Mod Pathol. 2014;27:34–90.
Chng TW, Gudi M, Lim SH, Li H, Tan PH. Validation of the Singapore nomogram for outcome prediction in breast phyllodes tumours in a large patient cohort. J Clin Pathol. 2018;71:125–8.
Jacobs TW, Chen YY, Guinee DG Jr., Holden JA, Cha I, Bauermeister DE, et al. Fibroepithelial lesions with cellular stroma on breast core needle biopsy: are there predictors of outcome on surgical excision? Am J Clin Pathol. 2005;124:342–54.
Lee AH, Hodi Z, Ellis IO, Elston CW. Histological features useful in the distinction of phyllodes tumour and fibroadenoma on needle core biopsy of the breast. Histopathology. 2007;51:336–44.
Resetkova E, Khazai L, Albarracin CT, Arribas E. Clinical and radiologic data and core needle biopsy findings should dictate management of cellular fibroepithelial tumors of the breast. Breast J. 2010;16:573–80.
Jara-Lazaro AR, Akhilesh M, Thike AA, Lui PC, Tse GM, Tan PH. Predictors of phyllodes tumours on core biopsy specimens of fibroepithelial neoplasms. Histopathology. 2010;57:220–32.
Yasir S, Gamez R, Jenkins S, Visscher DW, Nassar A. Significant histologic features differentiating cellular fibroadenoma from phyllodes tumor on core needle biopsy specimens. Am J Clin Pathol. 2014;142:362–9.
Jung J, Kang E, Chae SM, Kim H, Park SY, Yun B, et al. Development of a management algorithm for the diagnosis of cellular fibroepithelial lesions from core needle biopsies. Int J Surg Pathol. 2018;26:684–92.
Tan WJ, Cima I, Choudhury Y, Wei X, Lim JC, Thike AA, et al. A five-gene reverse transcription-PCR assay for pre-operative classification of breast fibroepithelial lesions. Breast Cancer Res. 2016;18:31.
Sim Y, Ng GXP, Ng CCY, Rajasegaran V, Wong SF, Liu W, et al. A novel genomic panel as an adjunctive diagnostic tool for the characterization and profiling of breast fibroepithelial lesions. BMC Med Genom. 2019;12:142.
Dessauvagie BF, Lee AHS, Meehan K, Nijhawan A, Tan PH, Thomas J, et al. Interobserver variation in the diagnosis of fibroepithelial lesions of the breast: a multicentre audit by digital pathology. J Clin Pathol. 2018;71:672–9.
Sin EI, Wong CY, Yong WS, Ong KW, Madhukumar P, Tan VK, et al. Breast carcinoma and phyllodes tumour: a case series. J Clin Pathol. 2016;69:364–9.
Tan PH, Jayabaskar T, Chuah KL, Lee HY, Tan Y, Hilmy M, et al. Phyllodes tumors of the breast: the role of pathologic parameters. Am J Clin Pathol. 2005;123:529–40.
Liu SY, Joseph NM, Ravindranathan A, Stohr BA, Greenland NY, Vohra P, et al. Genomic profiling of malignant phyllodes tumors reveals aberrations in FGFR1 and PI-3 kinase/RAS signaling pathways and provides insights into intratumoral heterogeneity. Mod Pathol. 2016;29:1012–27.
Bacchi CE, Wludarski SC, Lamovec J, Ben Dor D, Ober E, Salviato T, et al. Lipophyllodes of the breast. A reappraisal of fat-rich tumors of the breast based on 22 cases integrated by immunohistochemical study, molecular pathology insights, and clinical follow-up. Ann Diagn Pathol. 2016;21:1–6.
Inyang A, Thomas DG, Jorns J. Heterologous liposarcomatous differentiation in malignant phyllodes tumor is histologically similar but immunohistochemically and molecularly distinct from well-differentiated liposarcoma of soft tissue. Breast J. 2016;22:282–6.
Lyle PL, Bridge JA, Simpson JF, Cates JM, Sanders ME. Liposarcomatous differentiation in malignant phyllodes tumours is unassociated with MDM2 or CDK4 amplification. Histopathology. 2016;68:1040–5.
Powell CM, Rosen PP. Adipose differentiation in cystosarcoma phyllodes. A study of 14 cases. Am J Surg Pathol. 1994;18:720–7.
Rowe JJ, Cheah AL, Calhoun BC. Lipomatous tumors of the breast: a contemporary review. Semin Diagn Pathol. 2017;34:453–61.
Koh VCY, Thike AA, Nasir NDM, Yip GWC, Bay BH, Tan PH. Size and heterologous elements predict metastases in malignant phyllodes tumours of the breast. Virchows Arch. 2018;472:615–21.
Kim JM, Jung EJ, Kim JY, Lee HS, An HJ, Park T, et al. A rare case of mixed type liposarcoma of breast arising in malignant phyllodes tumor. Breast J. 2020;26:271–3.
Sancheti SM, Sawaimoon SK, Ahmed R. Pleomorphic liposarcoma arising in a malignant phyllodes tumor of breast: a rare occurrence. J Cancer Res Ther. 2015;11:1032.
Tan BY, Md Nasir ND, Chang HY, Ng CC, Guan P, Nagarajan S, et al. Morphologic and genetic heterogeneity in breast fibroepithelial lesions—a comprehensive mapping study. Mod Pathol. 2020. https://doi.org/10.1038/s41379-020-0533-0.
Tan PH, Thike AA, Tan WJ, Thu MM, Busmanis I, Li H, et al. Predicting clinical behaviour of breast phyllodes tumours: a nomogram based on histological criteria and surgical margins. J Clin Pathol. 2012;65:69–76.
Nishimura R, Tan PH, Thike AA, Tan MH, Taira N, Li HH, et al. Utility of the Singapore nomogram for predicting recurrence-free survival in Japanese women with breast phyllodes tumours. J Clin Pathol. 2014;67:748–50.
Chng TW, Lee JY, Lee CS, Li H, Tan MH, Tan PH. Validation of the Singapore nomogram for outcome prediction in breast phyllodes tumours: an Australian cohort. J Clin Pathol. 2016;69:1124–6.
Cristando C, Li H, Almekinders M, Tan PH, Brogi E, Murray M. Validation of the Singapore nomogram for outcome prediction in a US-based population of women with breast phyllodes tumors (PT). Mod Pathol. 2017;30:27–81.
Tan BY, Acs G, Apple SK, Badve S, Bleiweiss IJ, Brogi E, et al. Phyllodes tumours of the breast: a consensus review. Histopathology. 2016;68:5–21.
Tan BY, Tan PH. A diagnostic approach to fibroepithelial breast lesions. Surg Pathol Clin. 2018;11:17–42.
Slodkowska E, Nofech-Mozes S, Xu B, Parra-Herran C, Lu FI, Raphael S, et al. Fibroepithelial lesions of the breast: a comprehensive morphological and outcome analysis of a large series. Mod Pathol. 2018;31:1073–84.
Tan PH, Sahin AA. Atlas of differential diagnosis in breast pathology. New York: Springer; 2017.
Tse GM, Law BK, Chan KF, Mas TK. Multinucleated stromal giant cells in mammary phyllodes tumours. Pathology. 2001;33:153–6.
Tan PH, Jayabaskar T, Yip G, Tan Y, Hilmy M, Selvarajan S, et al. p53 and c-kit (CD117) protein expression as prognostic indicators in breast phyllodes tumors: a tissue microarray study. Mod Pathol. 2005;18:1527–34.
Koo CY, Bay BH, Lui PC, Tse GM, Tan PH, Yip GW. Immunohistochemical expression of heparan sulfate correlates with stromal cell proliferation in breast phyllodes tumors. Mod Pathol. 2006;19:1344–50.
Jara-Lazaro AR, Tan PH. Molecular pathogenesis of progression and recurrence in breast phyllodes tumors. Am J Transl Res. 2009;1:23–34.
Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ. WHO classification of tumours of the breast. 4th ed. France, Lyon: IARC; 2012.
Burga AM, Tavassoli FA. Periductal stromal tumor: a rare lesion with low-grade sarcomatous behavior. Am J Surg Pathol. 2003;27:343–8.
Hart WR, Bauer RC, Oberman HA. Cystosarcoma phyllodes. A clinicopathologic study of twenty-six hypercellular periductal stromal tumors of the breast. Am J Clin Pathol. 1978;70:211–6.
Ho SK, Thike AA, Cheok PY, Tse GM, Tan PH. Phyllodes tumours of the breast: the role of CD34, vascular endothelial growth factor and beta-catenin in histological grading and clinical outcome. Histopathology. 2013;63:393–406.
Moore T, Lee AH. Expression of CD34 and bcl-2 in phyllodes tumours, fibroadenomas and spindle cell lesions of the breast. Histopathology. 2001;38:62–7.
Chen CM, Chen CJ, Chang CL, Shyu JS, Hsieh HF, Harn HJ. CD34, CD117, and actin expression in phyllodes tumor of the breast. J Surg Res. 2000;94:84–91.
Lee AH. Recent developments in the histological diagnosis of spindle cell carcinoma, fibromatosis and phyllodes tumour of the breast. Histopathology. 2008;52:45–57.
Dunne B, Lee AH, Pinder SE, Bell JA, Ellis IO. An immunohistochemical study of metaplastic spindle cell carcinoma, phyllodes tumor and fibromatosis of the breast. Hum Pathol. 2003;34:1009–15.
Lacroix-Triki M, Geyer FC, Lambros MB, Savage K, Ellis IO, Lee AH, et al. beta-catenin/Wnt signalling pathway in fibromatosis, metaplastic carcinomas and phyllodes tumours of the breast. Mod Pathol. 2010;23:1438–48.
Sawyer EJ, Hanby AM, Rowan AJ, Gillett CE, Thomas RE, Poulsom R, et al. The Wnt pathway, epithelial-stromal interactions, and malignant progression in phyllodes tumours. J Pathol. 2002;196:437–44.
Sawyer EJ, Hanby AM, Poulsom R, Jeffery R, Gillett CE, Ellis IO, et al. Beta-catenin abnormalities and associated insulin-like growth factor overexpression are important in phyllodes tumours and fibroadenomas of the breast. J Pathol. 2003;200:627–32.
Hayes MJ, Thomas D, Emmons A, Giordano TJ, Kleer CG. Genetic changes of Wnt pathway genes are common events in metaplastic carcinomas of the breast. Clin Cancer Res. 2008;14:4038–44.
Lim SZ, Ong KW, Tan BK, Selvarajan S, Tan PH. Sarcoma of the breast: an update on a rare entity. J Clin Pathol. 2016;69:373–81.
Lim SZ, Ng CCY, Rajasegaran V, Guan P, Selvarajan S, Thike AA, et al. Genomic profile of breast sarcomas: a comparison with malignant phyllodes tumours. Breast Cancer Res Treat. 2019;174:365–73.
Lim SZ, Selvarajan S, Thike AA, Nasir ND, Tan BK, Ong KW, et al. Breast sarcomas and malignant phyllodes tumours: comparison of clinicopathological features, treatment strategies, prognostic factors and outcomes. Breast Cancer Res Treat. 2016;159:229–44.
Chia Y, Thike AA, Cheok PY, Yong-Zheng Chong L, Man-Kit Tse G, Tan PH. Stromal keratin expression in phyllodes tumours of the breast: a comparison with other spindle cell breast lesions. J Clin Pathol. 2012;65:339–47.
Cimino-Mathews A, Sharma R, Illei PB, Vang R, Argani P. A subset of malignant phyllodes tumors express p63 and p40: a diagnostic pitfall in breast core needle biopsies. Am J Surg Pathol. 2014;38:1689–96.
Lu Y, Chen Y, Zhu L, Cartwright P, Song E, Jacobs L, et al. Local recurrence of benign, borderline, and malignant phyllodes tumors of the breast: a systematic review and meta-analysis. Ann Surg Oncol. 2019;26:1263–75.
Barth RJ Jr. Borderline and malignant phyllodes tumors: how often do they locally recur and is there anything we can do about it? Ann Surg Oncol. 2019;26:1973–5.
Tan EY, Tan PH, Yong WS, Wong HB, Ho GH, Yeo AW, et al. Recurrent phyllodes tumours of the breast: pathological features and clinical implications. ANZ J Surg. 2006;76:476–80.
Borhani-Khomani K, Talman ML, Kroman N, Tvedskov TF. Risk of local recurrence of benign and borderline phyllodes tumors: a Danish population-based retrospective study. Ann Surg Oncol. 2016;23:1543–8.
Cowan ML, Argani P, Cimino-Mathews A. Benign and low-grade fibroepithelial neoplasms of the breast have low recurrence rate after positive surgical margins. Mod Pathol. 2016;29:259–65.
Moutte A, Chopin N, Faure C, Beurrier F, Ho Quoc C, Guinaudeau F, et al. Surgical management of benign and borderline phyllodes tumors of the breast. Breast J. 2016;22:547–52.
Moo TA, Alabdulkareem H, Tam A, Fontanet C, Lu Y, Landers A, et al. Association between recurrence and re-excision for close and positive margins versus observation in patients with benign phyllodes tumors. Ann Surg Oncol. 2017;24:3088–92.
Lim WK, Ong CK, Tan J, Thike AA, Ng CC, Rajasegaran V, et al. Exome sequencing identifies highly recurrent MED12 somatic mutations in breast fibroadenoma. Nat Genet. 2014;46:877–80.
Loke BN, Md Nasir ND, Thike AA, Lee JYH, Lee CS, Teh BT, et al. Genetics and genomics of breast fibroadenomas. J Clin Pathol. 2018;71:381–7.
Cani AK, Hovelson DH, McDaniel AS, Sadis S, Haller MJ, Yadati V, et al. Next-gen sequencing exposes frequent MED12 mutations and actionable therapeutic targets in phyllodes tumors. Mol Cancer Res. 2015;13:613–9.
Yoshida M, Sekine S, Ogawa R, Yoshida H, Maeshima A, Kanai Y, et al. Frequent MED12 mutations in phyllodes tumours of the breast. Br J Cancer. 2015;112:1703–8.
Nagasawa S, Maeda I, Fukuda T, Wu W, Hayami R, Kojima Y, et al. MED12 exon 2 mutations in phyllodes tumors of the breast. Cancer Med. 2015;4:1117–21.
Ng CC, Tan J, Ong CK, Lim WK, Rajasegaran V, Nasir ND, et al. MED12 is frequently mutated in breast phyllodes tumours: a study of 112 cases. J Clin Pathol. 2015;68:685–91.
Yoshida M, Ogawa R, Yoshida H, Maeshima A, Kanai Y, Kinoshita T, et al. TERT promoter mutations are frequent and show association with MED12 mutations in phyllodes tumors of the breast. Br J Cancer. 2015;113:1244–8.
Tan J, Ong CK, Lim WK, Ng CC, Thike AA, Ng LM, et al. Genomic landscapes of breast fibroepithelial tumors. Nat Genet. 2015;47:1341–5.
Piscuoglio S, Murray M, Fusco N, Marchio C, Loo FL, Martelotto LG, et al. MED12 somatic mutations in fibroadenomas and phyllodes tumours of the breast. Histopathology. 2015;67:719–29.
Lien HC, Huang CS, Yang YW, Jeng YM. Mutational analysis of MED12 exon 2 in a spectrum of fibroepithelial tumours of the breast: implications for pathogenesis and histogenesis. Histopathology. 2016;68:433–41.
Piscuoglio S, Ng CK, Murray M, Burke KA, Edelweiss M, Geyer FC, et al. Massively parallel sequencing of phyllodes tumours of the breast reveals actionable mutations, and TERT promoter hotspot mutations and TERT gene amplification as likely drivers of progression. J Pathol. 2016;238:508–18.
Yoon N, Bae GE, Kang SY, Choi MS, Hwang HW, Kim SW, et al. Frequency of MED12 mutations in phyllodes tumors: Inverse correlation with histologic grade. Genes Chromosom Cancer. 2016;55:495–504.
Tan WJ, Chan JY, Thike AA, Lim JC, Md Nasir ND, Tan JS, et al. MED12 protein expression in breast fibroepithelial lesions: correlation with mutation status and oestrogen rece ptor expression. J Clin Pathol. 2016;69:858–65.
Piscuoglio S, Geyer FC, Burke KA, Murray MP, Ng CK, Mota A, et al. Massively parallel sequencing analysis of synchronous fibroepithelial lesions supports the concept of progression from fibroadenoma to phyllodes tumor. NPJ Breast Cancer. 2016;2:16035.
Lae M, Gardrat S, Rondeau S, Richardot C, Caly M, Chemlali W, et al. MED12 mutations in breast phyllodes tumors: evidence of temporal tumoral heterogeneity and identification of associated critical signaling pathways. Oncotarget. 2016;7:84428–38.
Nozad S, Sheehan CE, Gay LM, Elvin JA, Vergilio JA, Suh J, et al. Comprehensive genomic profiling of malignant phyllodes tumors of the breast. Breast Cancer Res Treat. 2017;162:597–602.
Pareja F, Geyer FC, Kumar R, Selenica P, Piscuoglio S, Ng CKY, et al. Phyllodes tumors with and without fibroadenoma-like areas display distinct genomic features and may evolve through distinct pathways. NPJ Breast Cancer. 2017;3:40.
Garcia-Dios DA, Levi D, Shah V, Gillett C, Simpson MA, Hanby A, et al. MED12, TERT promoter and RBM15 mutations in primary and recurrent phyllodes tumours. Br J Cancer. 2018;118:277–84.
Kim JY, Yu JH, Nam SJ, Kim SW, Lee SK, Park WY, et al. Genetic and clinical characteristics of phyllodes tumors of the breast. Transl Oncol. 2018;11:18–23.
Darooei M, Khan F, Rehan M, Zubeda S, Jeyashanker E, Annapurna S, et al. MED12 somatic mutations encompassing exon 2 associated with benign breast fibroadenomas and not breast carcinoma in Indian women. J Cell Biochem. 2019;120:182–91.
Xie SN, Cai YJ, Ma B, Xu Y, Qian P, Zhou JD, et al. The genomic mutation spectrums of breast fibroadenomas in Chinese population by whole exome sequencing analysis. Cancer Med. 2019;8:2372–9.
Wang H, Shen Q, Ye LH, Ye J. MED12 mutations in human diseases. Protein Cell. 2013;4:643–6.
Turunen M, Spaeth JM, Keskitalo S, Park MJ, Kivioja T, Clark AD, et al. Uterine leiomyoma-linked MED12 mutations disrupt mediator-associated CDK activity. Cell Rep. 2014;7:654–60.
Yeong J, Thike AA, Young Ng CC, Md Nasir ND, Loh K, Teh BT, et al. A genetic mutation panel for differentiating malignant phyllodes tumour from metaplastic breast carcinoma. Pathology. 2017;49:786–9.
Lien HC, Huang CS, Yang YW, Jeng YM. MED12 exon 2 mutation as a highly sensitive and specific marker in distinguishing phyllodes tumours from other spindle neoplasms of the breast. APMIS. 2016;124:356–64.
Acknowledgements
The support by the Singapore General Hospital (SGH) Division of Pathology Breast Research Group, SGH Department of Anatomical Pathology and the International Fibroepithelial Consortium is appreciated. The assistance of Ms Nur Diyana Binte Md Nasir and Ms Valerie Koh with the tables and figures is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest in this review.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tan, P.H. Fibroepithelial lesions revisited: implications for diagnosis and management. Mod Pathol 34 (Suppl 1), 15–37 (2021). https://doi.org/10.1038/s41379-020-0583-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41379-020-0583-3
This article is cited by
-
Third International Consensus Conference on lesions of uncertain malignant potential in the breast (B3 lesions)
Virchows Archiv (2023)
-
Genomic characteristics of two breast malignant phyllodes tumors during pregnancy and lactation identified through whole-exome sequencing
Orphanet Journal of Rare Diseases (2022)
-
The value of whole-tumor histogram and texture analysis based on apparent diffusion coefficient (ADC) maps for the discrimination of breast fibroepithelial lesions: corresponds to clinical management decisions
Japanese Journal of Radiology (2022)
-
Fibroepithelial tumours of the breast—a review
Virchows Archiv (2022)
-
Morphological features of 52 cases of breast phyllodes tumours with local recurrence
Virchows Archiv (2022)