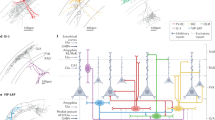
Abstract
The hippocampus contains a diverse array of inhibitory interneurons that gate information flow through local cortico-hippocampal circuits to regulate memory storage. Although most studies of interneurons have focused on their role in fast synaptic inhibition mediated by GABA release, different classes of interneurons express unique sets of neuropeptides, many of which have been shown to exert powerful effects on neuronal function and memory when applied pharmacologically. However, relatively little is known about whether and how release of endogenous neuropeptides from inhibitory cells contributes to their behavioral role in regulating memory formation. Here we report that vasoactive intestinal peptide (VIP)-expressing interneurons participate in social memory storage by enhancing information transfer from hippocampal CA3 pyramidal neurons to CA2 pyramidal neurons. Notably, this action depends on release of the neuropeptide enkephalin from VIP neurons, causing long-term depression of feedforward inhibition onto CA2 pyramidal cells. Moreover, VIP neuron activity in the CA2 region is increased selectively during exploration of a novel conspecific. Our findings, thus, enhance our appreciation of how GABAergic neurons can regulate synaptic plasticity and mnemonic behavior by demonstrating that such actions can be mediated by release of a specific neuropeptide, rather than through classic fast inhibitory transmission.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
22 November 2021
Incorrect ORCID ID was removed for co-author Tobias Bock.
References
Basu J, Zaremba JD, Cheung SK, Hitti FL, Zemelman BV, Losonczy A, et al. Gating of hippocampal activity, plasticity, and memory by entorhinal cortex long-range inhibition. Science. 2016;351:aaa5694–aaa5694.
Letzkus JJ, Wolff SBE, Meyer EMM, Tovote P, Courtin J, Herry C, et al. A disinhibitory microcircuit for associative fear learning in the auditory cortex. Nature. 2011;480:331–5.
Letzkus JJ, Wolff SBE, Lüthi A. Disinhibition, a circuit mechanism for associative learning and memory. Neuron. 2015;88:264–76.
Turi GF, Li W-K, Chavlis S, Pandi I, O’Hare J, Priestley JB, et al. Vasoactive intestinal polypeptide-expressing interneurons in the hippocampus support goal-oriented spatial learning. Neuron. 2019;101:1150–65.e8.
Krabbe S, Paradiso E, D’Aquin S, Bitterman Y, Courtin J, Xu C, et al. Adaptive disinhibitory gating by VIP interneurons permits associative learning. Nat Neurosci. 2019;22:1834–43.
Lee AT, Cunniff MM, See JZ, Wilke SA, Luongo FJ, Ellwood IT, et al. VIP interneurons contribute to avoidance behavior by regulating information flow across hippocampal-prefrontal networks. Neuron. 2019;102:1223–34.e4.
Williams LE, Holtmaat A. Higher-order thalamocortical inputs gate synaptic long-term potentiation via disinhibition. Neuron. 2019;101:91–02.e4.
DeFelipe J, López-Cruz PL, Benavides-Piccione R, Bielza C, Larrañaga P, Anderson S, et al. New insights into the classification and nomenclature of cortical GABAergic interneurons. Nat Rev Neurosci. 2013;14:202–16.
Ögren SO, Kuteeva E, Elvander-Tottie E, Hökfelt T. Neuropeptides in learning and memory processes with focus on galanin. Eur J Pharmacol. 2010;626:9–17.
Marder E. Neuromodulation of neuronal circuits: back to the future. Neuron. 2012;76:1–11.
Borbély É, Scheich B, Helyes Z. Neuropeptides in learning and memory. Neuropeptides. 2013;47:439–50.
Gøtzsche CR, Woldbye DPD. The role of NPY in learning and memory. Neuropeptides. 2016;55:79–89.
Dávid C, Schleicher A, Zuschratter W, Staiger JF. The innervation of parvalbumin-containing interneurons by VIP-immunopositive interneurons in the primary somatosensory cortex of the adult rat. Eur J Neurosci. 2007;25:2329–40.
Kepecs A, Fishell G. Interneuron cell types are fit to function. Nature. 2014;505:318–26.
Acsády L, Görcs TJ, Freund TF. Different populations of vasoactive intestinal polypeptide-immunoreactive interneurons are specialized to control pyramidal cells or interneurons in the hippocampus. Neuroscience. 1996;73:317–34.
Tyan L, Chamberland S, Magnin E, Camire O, Francavilla R, David LS, et al. Dendritic inhibition provided by interneuron-specific cells controls the firing rate and timing of the hippocampal feedback inhibitory circuitry. J Neurosci. 2014;34:4534–47.
Chamberland S, Topolnik L. Inhibitory control of hippocampal inhibitory neurons. Front Neurosci. 2012;6:165.
Pi H-J, Hangya B, Kvitsiani D, Sanders JI, Huang ZJ, Kepecs A. Cortical interneurons that specialize in disinhibitory control. Nature. 2013;503:521–4.
Kennett JE, Poletini MO, Freeman ME. Vasoactive intestinal polypeptide modulates the estradiol-induced prolactin surge by entraining oxytocin neuronal activity. Brain Res. 2008;1196:65–73.
Blasco-Ibáñez JM, Martínez-Guijarro FJ, Freund TF. Enkephalin-containing interneurons are specialized to innervate other interneurons in the hippocampal CA1 region of the rat and guinea-pig. Eur J Neurosci. 1998;10:1784–95.
Botcher NA, Falck JE, Thomson AM, Mercer A. Distribution of interneurons in the CA2 region of the rat hippocampus. Front Neuroanat. 2014;8:104.
Piskorowski RA, Chevaleyre V. Delta-opioid receptors mediate unique plasticity onto parvalbumin-expressing interneurons in area CA2 of the hippocampus. J Neurosci. 2013;33:14567–78.
Leroy F, Brann DH, Meira T, Siegelbaum SA. Input-timing-dependent Plasticity in the Hippocampal CA2 region and its potential role in social memory. Neuron. 2017;95:1089–102.e5.
Nasrallah K, Piskorowski RA, Chevaleyre V. Inhibitory plasticity permits the recruitment of CA2 pyramidal neurons by CA3. Eneuro. 2015. https://doi.org/10.1523/ENEURO.0049-15.2015.
Benes FM, Kwok EW, Vincent SL, Todtenkopf MS. A reduction of nonpyramidal cells in sector CA2 of schizophrenics and manic depressives. Biol Psychiatry. 1998;44:88–97.
Knable MB, Barci BM, Webster MJ, Meador-Woodruff J, Torrey EF. Molecular abnormalities of the hippocampus in severe psychiatric illness: postmortem findings from the Stanley Neuropathology Consortium. Mol Psychiatry. 2004;9:609–20.
Piskorowski RA, Nasrallah K, Diamantopoulou A, Mukai J, Hassan SI, Siegelbaum SA, et al. Age-dependent specific changes in area CA2 of the hippocampus and social memory deficit in a mouse model of the 22q11.2 deletion syndrome. Neuron. 2016;89:163–76.
Donegan ML, Stefanini F, Meira T, Gordon JA, Fusi S, Siegelbaum SA. Coding of social novelty in the hippocampal CA2 region and its disruption and rescue in a 22q11.2 microdeletion mouse model. Nat Neurosci. 2020;23:1365–75.
Cheng AH, Fung SW, Cheng, YM H-. Limitations of the Avp-IRES2-Cre (JAX #023530) and Vip-IRES-Cre (JAX #010908) models for chronobiological investigations. J Biol Rhythms. 2019;34:634–44.
Mattis J, Tye KM, Ferenczi EA, Ramakrishnan C, O’Shea DJ, Prakash R, et al. Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins. Nat Methods. 2012;9:159–72.
Dudman JT, Tsay D, Siegelbaum SA. A role for synaptic inputs at distal dendrites: instructive signals for hippocampal long-term plasticity. Neuron. 2007;56:866–79.
Chevaleyre V, Siegelbaum SA. Strong CA2 pyramidal neuron synapses define a powerful disynaptic cortico-hippocampal loop. Neuron. 2010;66:560–72.
Basu J, Srinivas KV, Cheung SK, Taniguchi H, Huang ZJ, Siegelbaum SA. A cortico-hippocampal learning rule shapes inhibitory microcircuit activity to enhance hippocampal information flow. Neuron. 2013;79:1208–21.
Bérubé P, Poulin J-F, Laforest S, Drolet G. Enkephalin knockdown in the basolateral amygdala reproduces vulnerable anxiety-like responses to chronic unpredictable stress. Neuropsychopharmacology. 2014;39:1159–68.
Li X, Marshall PR, Leighton LJ, Zajaczkowski EL, Wang Z, Madugalle SU, et al. The DNA repair-associated protein Gadd45γ regulates the temporal coding of immediate early gene expression within the prelimbic prefrontal cortex and is required for the consolidation of associative fear memory. J Neurosci. 2019;39:970–83.
Kitamura T, Pignatelli M, Suh J, Kohara K, Yoshiki A, Abe K, et al. Island cells control temporal association memory. Science. 2014;343:896–901.
Oliva A, Fernández-Ruiz A, Leroy F, Siegelbaum SA. Hippocampal CA2 sharp-wave ripples reactivate and promote social memory. Nature. 2020;587:264–9.
Urban DJ, Roth BL. DREADDs (designer receptors exclusively activated by designer drugs): chemogenetic tools with therapeutic utility. Annu Rev Pharm Toxicol. 2015;55:399–417.
Hitti FL, Siegelbaum SA. The hippocampal CA2 region is essential for social memory. Nature. 2014;508:88–92.
Meira T, Leroy F, Buss EW, Oliva A, Park J, Siegelbaum SA. A hippocampal circuit linking dorsal CA2 to ventral CA1 critical for social memory dynamics. Nat Commun. 2018;9:4163.
Stevenson EL, Caldwell HK. Lesions to the CA2 region of the hippocampus impair social memory in mice. Eur J Neurosci. 2014;40:3294–301.
Citri A, Malenka RC. Synaptic plasticity: multiple forms, functions, and mechanisms. Neuropsychopharmacology. 2008;33:18–41.
Andersen P, Morris R, Amaral D, Bliss T, O’ Keefe J. The Hippocampus Book. Oxford University Press; 2006. Published to Oxford Scholarship Online: May 2009.
Wilmes KA, Clopath C. Inhibitory microcircuits for top-down plasticity of sensory representations. Nat Commun. 2019;10:5055.
Díaz J-L, Asai M. Dominant mice show much lower concentrations of methionine-enkephalin in brain tissue than subordinates: cause or effect? Behav Brain Res. 1990;39:275–80.
König M, Zimmer AM, Steiner H, Holmes PV, Crawley JN, Brownstein MJ, et al. Pain responses, anxiety and aggression in mice deficient in pre-proenkephalin. Nature. 1996;383:535–8.
Roques BP. Novel approaches to targeting neuropeptide systems. Trends Pharm Sci. 2000;21:475–83.
Calenco-Choukroun G, Daugé V, Gacel G, Féger J, Roques BP. Opioid δ agonists and endogenous enkephalins induce different emotional reactivity than μ agonists after injection in the rat ventral tegmental area. Psychopharmacology. 1991;103:493–502.
François A, Low SA, Sypek EI, Christensen AJ, Sotoudeh C, Beier KT, et al. A brainstem-spinal cord inhibitory circuit for mechanical pain modulation by GABA and enkephalins. Neuron. 2017;93:822–39.e6.
Nicoll RA, Alger BE, Jahr CE. Enkephalin blocks inhibitory pathways in the vertebrate CNS. Nature. 1980;287:22–5.
Madison DV, Nicoll RA. Enkephalin hyperpolarizes interneurones in the rat hippocampus. J Physiol. 1988;398:123–30.
Nasrallah K, Therreau L, Robert V, Huang AJY, McHugh TJ, Piskorowski RA, et al. Routing hippocampal information flow through parvalbumin interneuron plasticity in area CA2. Cell Rep. 2019;27:86–98.e3.
Domínguez S, Rey CC, Therreau L, Fanton A, Massotte D, Verret L, et al. Maturation of PNN and ErbB4 signaling in area CA2 during adolescence underlies the emergence of PV interneuron plasticity and social memory. Cell Rep. 2019;29:1099–112.e4.
Mankin EA, Diehl GW, Sparks FT, Leutgeb S, Leutgeb JK. Hippocampal CA2 activity patterns change over time to a larger extent than between spatial contexts. Neuron. 2015;85:190–201.
Alexander GM, Farris S, Pirone JR, Zheng C, Colgin LL, Dudek SM. Social and novel contexts modify hippocampal CA2 representations of space. Nat Commun. 2016;7:10300.
Kay K, Sosa M, Chung JE, Karlsson MP, Larkin MC, Frank LM. A hippocampal network for spatial coding during immobility and sleep. Nature. 2016;531:185–90.
Wintzer ME, Boehringer R, Polygalov D, McHugh TJ. The hippocampal CA2 ensemble is sensitive to contextual change. J Neurosci. 2014;34:3056–66.
Buzsáki G. Hippocampal sharp wave‐ripple: a cognitive biomarker for episodic memory and planning. Hippocampus. 2015;25:1073–188.
Oliva A, Fernández-Ruiz A, Buzsáki G, Berényi A. Role of hippocampal CA2 region in triggering sharp-wave ripples. Neuron. 2016;91:1342–55.
Alexander GM, Brown LY, Farris S, Lustberg D, Pantazis C, Gloss B, et al. CA2 neuronal activity controls hippocampal low gamma and ripple oscillations. Elife. 2018;7:e38052.
Josselyn SA, Tonegawa S. Memory engrams: recalling the past and imagining the future. Science. 2020;367:eaaw4325.
Okuyama T, Kitamura T, Roy DS, Itohara S, Tonegawa S. Ventral CA1 neurons store social memory. Science. 2016;353:1536–41.
Chiang M-C, Huang AJY, Wintzer ME, Ohshima T, McHugh TJ. A role for CA3 in social recognition memory. Behav Brain Res. 2018;354:22–30.
Patel J, Schomburg EW, Berényi A, Fujisawa S, Buzsáki G. Local generation and propagation of ripples along the septotemporal axis of the hippocampus. J Neurosci. 2013;33:17029–41.
Drew LJ, Crabtree GW, Markx S, Stark KL, Chaverneff F, Xu B, et al. The 22q11.2 microdeletion: fifteen years of insights into the genetic and neural complexity of psychiatric disorders. Int J Dev Neurosci. 2011;29:259–81.
Clark SD, Van Snellenberg JX, Lawson JM, Abi-Dargham A. Opioid antagonists are associated with a reduction in the symptoms of schizophrenia: a meta-analysis of controlled trials. Neuropsychopharmacology. 2020;45:1860–9.
Giovannucci A, Friedrich J, Gunn P, Kalfon J, Brown BL, Koay SA, et al. CaImAn an open source tool for scalable calcium imaging data analysis. Elife. 2019;8:e38173.
Mathis A, Mamidanna P, Cury KM, Abe T, Murthy VN, Mathis MW, et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat Neurosci. 2018;21:1281–9.
Favuzzi E, Deogracias R, Marques-Smith A, Maeso P, Jezequel J, Exposito-Alonso D, et al. Distinct molecular programs regulate synapse specificity in cortical inhibitory circuits. Science. 2019;363:413–7.
Chechik G, Sharma V, Shalit U, Bengio S. Large scale online learning of image similarity through ranking. J Mach Learn Res. 2010;11:1109–35.
Acknowledgements
This work was supported by: a 2019 NARSAD young investigator grant to FL from the Brain and Behavior research foundation, funded by the Osterhaus Family; a F32 MH122147-01A1 to CdS; Howard Hughes Medical Institute support to ERK and R01-MH104602 and R01-MH106629 to SAS.
Author information
Authors and Affiliations
Contributions
Conceptualization: FL and SAS; investigation, FL, TB, CAdS, AA, LB, OML, and EB; in vitro intra-cellular recordings: FL and TB; behavioral assays and viral injections: FL and OML; immunohistochemistry and in situ hybridization: FL, CdS, and AA; shRNA design CAdS and AA, Calcium imaging: LMB writing—original draft: FL; writing—review and editing: FL, AA, ERK, and SAS; visualization: FL; supervision: FL; funding acquisition: FL, CAdS, ERK, and SAS.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Leroy, F., de Solis, C.A., Boyle, L.M. et al. Enkephalin release from VIP interneurons in the hippocampal CA2/3a region mediates heterosynaptic plasticity and social memory. Mol Psychiatry 27, 2879–2900 (2022). https://doi.org/10.1038/s41380-021-01124-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-021-01124-y
This article is cited by
-
Heterosynaptic plasticity-induced modulation of synapses
The Journal of Physiological Sciences (2023)
-
The plasticitome of cortical interneurons
Nature Reviews Neuroscience (2023)
-
The role of inhibitory circuits in hippocampal memory processing
Nature Reviews Neuroscience (2022)
-
GABAergic CA1 neurons are more stable following context changes than glutamatergic cells
Scientific Reports (2022)