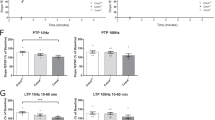
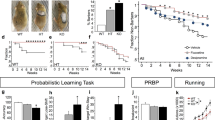
Abstract
Cerebellin-1 (Cbln1) and cerebellin-2 (Cbln2) are secreted glycoproteins that are expressed in distinct subsets of neurons throughout the brain. Cbln1 and Cbln2 simultaneously bind to presynaptic neurexins and postsynaptic GluD1 and GluD2, thereby forming trans-synaptic adhesion complexes. Genetic associations link cerebellins, neurexins and GluD’s to neuropsychiatric disorders involving compulsive behaviors, such as Tourette syndrome, attention-deficit hyperactivity disorder (ADHD), and obsessive-compulsive disorder (OCD). Extensive evidence implicates dysfunction of serotonergic signaling in these neuropsychiatric disorders. Here, we report that constitutive Cbln2 KO mice, but not Cbln1 KO mice, display robust compulsive behaviors, including stereotypic pattern running, marble burying, explosive jumping, and excessive nest building, and exhibit decreased brain serotonin levels. Strikingly, treatment of Cbln2 KO mice with the serotonin precursor 5-hydroxytryptophan or the serotonin reuptake-inhibitor fluoxetine alleviated compulsive behaviors. Conditional deletion of Cbln2 both from dorsal raphe neurons and from presynaptic neurons synapsing onto dorsal raphe neurons reproduced the compulsive behaviors of Cbln2 KO mice. Finally, injection of recombinant Cbln2 protein into the dorsal raphe of Cbln2 KO mice largely reversed their compulsive behaviors. Taken together, our results show that Cbln2 controls compulsive behaviors by regulating serotonergic circuits in the dorsal raphe.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Iacono WG, Malone SM, McGue M. Behavioral disinhibition and the development of early-onset addiction: common and specific influences. Annu Rev Clin Psychol. 2008;4:325–48.
Angoa-Perez M, Kane MJ, Briggs DI, Sykes CE, Shah MM, Francescutti DM, et al. Genetic depletion of brain 5HT reveals a common molecular pathway mediating compulsivity and impulsivity. J Neurochem. 2012;121:974–84.
Freeman RD, Fast DK, Burd L, Kerbeshian J, Roberton MM, Sandor P. An international perspective on Tourette syndrome: selected findings from 3500 individuals in 22 countries. Dev Med Child Neurol. 2000;42:436–47.
Burd L, Li Q, Kerbeshian J, Klug MG, Freeman RD. Tourette syndrome and comorbid pervasive developmental disorders. J Child Neurol. 2009;24:170–5.
Clarke RA, Lee S, Eapen V. Pathogenetic model for Tourette syndrome delineates overlap with related neurodevelopment disorders including autism. Transl Psychiatry. 2012;2:e158.
State MW, Greally JM, Cuker A, Bowers PN, Henegariu O, Morgan TM, et al. Epigenetic abnormalities associated with a chromosome 18(q21-q22) inversion and a Gilles de la Tourette syndrome phenotype. Proc Nat Acad Sci. 2003;100:4684–9.
State MW. The genetics of Tourette disorder. Curr Opin Genet Dev. 2011;21:302–9.
Robbins TW, Vaghi MM, Banca P. Obsessive-compulsive disorder: puzzles and prospects. Neuron 2019;102:27–47. Apr 3
Stein DJ, Costa DLC, Lochner C, Miguel EC, Reddy YCJ, Shavitt RG, et al. Obsessive-compulsive disorder. Nat Rev Dis Prim. 2019;5:52. Aug1
Szechtman H, Harvey BH, Woody EZ, Hoffman KL. The psychopharmacology of obsessive-compulsive disorder: a preclinical roadmap. Pharm Rev. 2020;72:80–151. Jan
Hirai H, Pang Z, Bao D, Miyazaki T, Li L, Miura E, et al. Cbln1 is essential for synaptic integrity and plasticity in the cerebellum. Nat Neurosci. 2005;8:1534–41.
Uemura T, Lee SJ, Yasumura M, Takeuchi T, Yoshida T, Ra M, et al. Trans-synaptic interaction of GluRδ2 and Neurexin through Cbln1 mediates synapse formation in the cerebellum. Cell. 2010;141:1068–79.
Matsuda K, Yuzaki M. Cbln family proteins promote synapse formation by regulating distinct neurexin signaling pathways in various brain regions. Eur J Neurosci. 2011;33:1447–61.
Otsuka S, Konno K, Abe M, Motohashi J, Kohda K, Sakimura K, et al. Roles of Cbln1 in non-motor functions of mice. J Neurosci. 2016;36:11801–16.
Seigneur E, Südhof TC. Genetic ablation of all cerebellins reveals synapse organizer functions in multiple regions throughout the brain. J Neurosci. 2018;38:4774–90.
Yuzaki M. The C1q complement family of synaptic organizers: not just complementary. Curr Opin Neurobiol. 2017;45:9–15.
Südhof TC. Synaptic neurexin complexes: a molecular code for the logic of neural circuits. Cell 2017;171:745–69.
Ushkaryov YA, Petrenko AG, Geppert M, Südhof TC. Neurexins: synaptic cell surface proteins related to the α-latrotoxin receptor and laminin. Science. 1992;257:50–56.
Ushkaryov YA, Südhof TC. Neurexin III α: extensive alternative splicing generates membrane-bound and soluble forms. Proc Natl Acad Sci. 1993;90:6410–4.
Ushkaryov YA, Hata Y, Ichtchenko K, Moomaw C, Afendis S, Slaughter CA, et al. Conserved domain structure of b-neurexins.Unusual cleaved signal sequences in receptor-like neuronal cell-surface proteins. J Biol Chem. 1994;269:11987–92.
Tabuchi K, Südhof TC. Structure and evolution of neurexin genes: insight into the mechanism of alternative splicing. Genomics. 2002;79:849–59.
Südhof TC. Neuroligins and neurexins link synaptic function to cognitive disease. Nature. 2008;455:903–11.
Guo SZ, Huang K, Shi YY, Tang W, Zhou J, Feng GY, et al. A case-control association study between the GRID1 gene and schizophrenia in the Chinese Northern Han population. Schizophr Res. 2007;93:385–90.
Venken T, Alaerts M, Souery D, Goossens D, Sluijs S, Navon R, et al. Chromosome 10q harbors a susceptibility locus for bipolar disorder in Ashkenazi Jewish families. Mol Psychiatry. 2008;13:442–50.
Treutlein J, Muhleisen TW, Frank J, Mattheisen M, Herms S, Ludwig KU, et al. Dissection of phenotype reveals possible association between schizophrenia and Glutamate Receptor Delta 1 (GRID1) gene promoter. Schizophr Res. 2009;111:123–30.
Yadav R, Gupta SC, Hillman BG, Bhatt JM, Stairs DJ, Dravid SM. Deletion of glutamate delta-1 receptor in mouse leads to aberrant emotional and social behaviors. PLOS One. 2012;7:e32969.
Arnold P, Askland K, Barlassina C, Bellodi L, Bienvenus OJ, Black D, et al. Revealing the complex genetic architecture of obsessive–compulsive disorder using meta-analysis. Mol Psychiatry. 2018;23:1181–8.
Gainetdinov RR, Wetsel WC, Jones SR, Levin ED, Jaber M, Caron MG. Role of serotonin in the paradoxical calming effect of psychostimulants on hyperactivity. Science. 1999;283:397–401.
Hashimoto H, Shintani N, Tanaka K, Mori W, Hirose M, Matsuda T, et al. Altered psychomotor behaviors in mice lacking pituitary adenylate cyclase-activating polypeptide (PACAP). Proc Natl Acad Sci. 2001;98:13355–60.
Joel D, Doljansky J, Roz N, Rehavi M. Role of the orbital cortex and of the serotonergic system in a rat model of obsessive compulsive disorder. Neuroscience. 2005;130:25–36.
Shintani N, Hashimoto H, Tanaka K, Kawagishi N, Kawaguchi C, Hatanaka M, et al. Serotonergic inhibition of intense jumping behavior in mice lacking PACAP (Adcyap1−/−). Ann NY Acad Sci. 2006;1070:545–9.
Li X, Morrow D, Witkin JM. Decreases in nestlet shedding of mice by serotonin uptake inhibitors: comparison with marble burying. Life Sci. 2006;78:1933–9.
Amilhon B, Lepicard E, Renoir T, Mongeau R, Popa D, Poirel O, et al. VLGUT3 (vesicular glutamate transporter type 3) contribution to the regulation of serotonergic transmission and anxiety. J Neurosci. 2010;30:2198–210.
Greene-Schloesser DM, Ven der Zee EA, Sheppard DK, Catillo MR, Gregg KA, Burrow T, et al. Predictive validity of a non-induced mouse model of compulsive-like behavior. Behav Brain Res. 2011;221:55–62.
Sachs BD, Rodriguiz RM, Siesser WB, Kenan A, Royer EL, Jacobsen JPR, et al. The effects of brain serotonin deficiency on behavioral disinhibition and anxiety-like behavior following mild early life stress. Int J Neuropsychopharm. 2013;16:2081–94.
Wolmarans DW, Brand L, Stein DJ, Harvey BH. Reappraisal of spontaneous stereotypy in the deer mouse as an animal model of obsessive–compulsive disorder (OCD): response to escitalopram treatment and basal serotonin transporter (SERT) density. Behav Brain Res. 2013;256:545–53.
Wolmarans DW, Stein DJ, Harvey BH. Excessive nest building is a unique behavioral phenotype in the deer mouse model of obsessive-compulsive disorder. J Pharm. 2016;30:867–74.
Seigneur E, Südhof TC. Cerebellins are differentially expressed in selective subsets of neurons throughout the brain. J Comp Neurol. 2017;525:3286–311.
Young KA, Berry ML, Mahaffey CL, Saionz JR, Hawes NL, Chang B, et al. Fierce: a new mouse deletion of Nr2e1; violent behaviour and ocular abnormalities are background-dependent. Behav Brain Res. 2002;132:145–58.
Deacon RMJ. Assessing nest building in mice. Nat Protoc. 2006;1:1117–9.
Willner P. The validity of animal models of depression. Psychopharm. 1984;83:1–16.
Kapur S, Remington G. Serotonin–dopamine interaction and its relevance to schizophrenia. Am J Psychiatry. 1996;153:466–76.
Rothman RB, Baumann MH. Balance between dopamine and serotonin release modulates behavioral effects of amphetamine-type drugs. Ann NY Acad Sci. 2006;1074:245–60.
Descarries L, Watkins KC, Garcia S, Beaudet A. The serotonin neurons in the nucleus raphe dorsalis of adult rat: a light and electron microscope radioautographic study. J Comp Neurol. 1982;207:239–54.
Dorocic IP, Furth D, Xuan Y, Johansson Y, Pozzi L, Silberberg G, et al. A whole-brain atlas of inputs to serotonergic neurons of the dorsal and median raphe nuclei. Neuron. 2014;83:663–78.
Zhuang X, Masson J, Gingrich J, Rayport S, Hen R. Targeted gene expression in dopamine and serotonin neurons of the mouse brain. J Neurosci Methods. 2005;143:27–32.
Teissier A, Soiza-Reilly M, Gaspar P. Refining the role of 5-HT in postnatal development of brain circuits. Front Cell Neurosci. 2017;11:139. May 23
Lee HS, Kim MA, Valentino RJ, Waterhouse BD. Glutamatergic afferent projections to the dorsal raphe nucleus of the rat. Brain Res. 2003;963:57–71.
Weissbourd B, Ren J, DeLoach K, Guenthner CJ, Miyamichi K, Luo L. Presynaptic partners of dorsal raphe serotonergic and GABAergic neurons. Neuron. 2014;83:645–62.
Geddes SD, Assadzada S, Lemelin D, Sokolovski A, Bergeron R, Haj-Dahmane S, et al. Target-specific modulation of the descending prefrontal cortex inputs to the dorsal raphe nucleus by cannabinoids. Proc Nat Acad Sci. 2016;113:5429–34.
Chen A, Hubbert KD, Foroudi PF, Lu VF, Janusonis S. Serotonin 5-HT4 receptors modulate the development of glutamatergic input to the dorsal raphe nucleus. Neurosci Lett. 2017;640:111–6.
Zhou L, Liu M-Z, Li Q, Deng J, Mu D, Sun YG. Organization of functional long-range circuits controlling the activity of sertonergic neurons in the dorsal raphe nucleus. Cell Rep. 2017;18:3018–32.
Herkenham M, Nauta WJ. Afferent connections of the habenular nuclei in the rat. A horseradish peroxidase study, with a note on the fiber-of-passage problem. J Comp Neurol. 1977;173:123–45.
Herkenham M, Nauta WJ. Efferent connections of the habenular nuclei in the rat. J Comp Neurol. 1979;187:19–47.
Seigneur E, Polepalli J, Südhof TC. Cbln2 and Cbln4 are expressed in distinct medial habenula-interpeduncular projections and contribute to different behavioral outputs. Proc Nat Acad Sci. 2018;115:E10235–E10244.
Wagner F, Stroh T, Veh R. Correlating habenular subnuclei in rat and mouse by using topographic, morphological, and cytochemical criteria. J Comp Neurol. 2014;522:2650–62.
Verge D, Daval G, Patey A, Gozlan H, el Mestikawy S, Hamon M. Presynaptic 5-HT autoreceptors on serotonergic cell bodies and/or dendrites but not terminals are of the 5-HT1A subtype. Eur J Pharm. 1985;113:463–4.
Fremeau RT, Burman J, Qureshi T, Tran CH, Proctor J, Johnson J, et al. The identification of vesicular glutamate transporter 3 suggests novel modes of signaling by glutamate. Proc Nat Acad Sci. 2002;99:14488–93.
Gras C, Herzog E, Bellenchi GC, Bernard V, Ravassard P, Pohl M, et al. A third vesicular glutamate transporter expressed by cholinergic and serotonergic neurons. J Neurosci. 2002;22:5442–51.
Soiza-Reilly M, Commons KG. Quantitative analysis of glutamatergic innervation of the mouse dorsal raphe nucleus using array tomography. J Comp Neurol. 2011;519:3802–14.
Gagnon D, Parent M. Distribution of VGLUT3 in highly collateralized axons from the rat dorsal raphe nucleus as revealed by single-neuron reconstructions. PLOS One. 2014;9:e87709.
Voisin AN, Mnie-Filali O, Giguere N, Fortin GM, Vigneault E, Mestikawy SE, et al. Axonal segregation and role of the vesicular glutamate transporter VGLUT3 in serotonin neurons. Front Neuroanat. 2016;10:39.
Tervo DG, Hwang BY, Viswanathan S, et al. A designer AAV variant permits efficient retrograde access to projection neurons. Neuron 2016;92:372–82.
Ito-Ishida A, Miura E, Emi K, Matsuda K, Iijima T, Kondo T, et al. Cbln1 regulates rapid formation and maintenance of excitatory synapses in mature cerebellar Purkinje cells in vitro and in vivo. J Neurosci. 2008;28:5920–30.
Takeuchi E, Ito-Ishida A, Yuzaki M, Yanagihara D. Improvement of cerebellar ataxic gait by injecting Cbln1 into the cerebellum of cbln1-null mice. Sci Rep. 2018;8:6184.
Yael D, Israelashvili M, Bar-Gad I. Animal models of Tourette syndrome—from proliferation to standardization. Front Neurosci. 2016;10:132.
Hirschtritt ME, Darrow SM, Illmann C, Osiecki L, Grados M, Sandor P, et al. Genetic and phenotypic overlap of specific obsessive-compulsive and attention-deficit/hyperactive subtypes with Tourette syndrome. Pschol Med. 2018;48:279–93.
Powell JM, Plummer NW, Scappini EL, Tucker CJ, Jensen P. DEFiNE: a method for enhancement and quantification of fluorescently labeled axons. Front Neuroanat. 2019;12:117.
Acknowledgements
This study was supported by a grant from the NIMH (MH052804 to TCS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Seigneur, E., Wang, J., Dai, J. et al. Cerebellin-2 regulates a serotonergic dorsal raphe circuit that controls compulsive behaviors. Mol Psychiatry 26, 7509–7521 (2021). https://doi.org/10.1038/s41380-021-01187-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-021-01187-x