Abstract
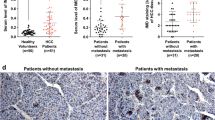
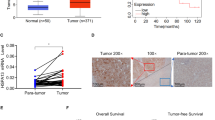
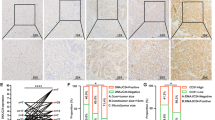
Recent studies indicated that insufficient radiofrequency ablation (RFA) could endow hepatocellular carcinoma (HCC) with higher aggressive potential. Stress-induced phosphoprotein 1 (STIP1), which was found highly expressed in HCC, is a chaperone molecule mediating cell homeostasis under thermal stress. We aimed to explore the role of STIP1 on the metastasis of residual HCC after RFA. Mice model with orthotopic HCC implants or caudal vein injection were employed to assess potential of lung metastasis and/or intrahepatic metastasis (IHM) of HCC cells. Cell culture model was used to determine cell invasion, mesenchymal marker genes expression, and underlying molecular mechanisms. Clinical specimens were collected to analyze the relationship between STIP1 and clinical outcome. We found that insufficient RFA elicited more IHM of HCCLM3 tumors, which could be reduced by silencing STIP1. Knockdown of STIP1 also significantly decreased lung metastatic potential of HCCLM3 cells. In vitro, HCCLM3 and HepG2 displayed a spindle-shaped morphology with upregulation of STIP1 and mesenchymal markers after sublethal heat exposure. Mechanistically, heat exposure induced the formation of STIP1-heat shock protein 90 (HSP90) complex, which could shuttle epithelial transcription repressor Snail1 into nucleus and regulate mesenchymal gene transcription. Blocking the HSP90–STIP1 complex reduced the invasive potential of HCC cells after heat exposure. Using clinical specimen, we found that STIP1 was expressed significantly higher in metastatic tumor tissues and in sera from metastatic HCC patients (p < 0.05). The high expression of STIP1 was significantly linked to shorter recurrence-free survival (p < 0.05). To sum up, our study found that STIP1 is positively associated with the sublethal heat-induced cancer cell metastasis through mediating the mesenchymal gene transcription. Blocking STIP1 activity may suppress HCC cell metastatic potential after RFA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Lau WY, Lai EC. The current role of radiofrequency ablation in the management of hepatocellular carcinoma: a systematic. Ann Surg. 2009;249:20–25.
Lee HY, Rhim H, Lee MW, Kim YS, Choi D, Park MJ, et al. Early diffuse recurrence of hepatocellular carcinoma after percutaneous radiofrequency ablation: analysis of risk factors. Eur Radiol. 2013;23:190–7.
Dong S, Kong J, Kong F, Kong J, Gao J, Ke S, et al. Insufficient radiofrequency ablation promotes epithelial-mesenchymal transition of hepatocellular carcinoma cells through Akt and ERK signaling pathways. J Transl Med. 2013;11:273.
Yoshida S, Kornek M, Ikenaga N, Schmelzle M, Masuzaki R, Csizmadia E, et al. Sublethal heat treatment promotes epithelial-mesenchymal transition and enhances the malignant potential of hepatocellular carcinoma. Hepatology. 2013;58:1667–80.
Thompson SM, Callstrom MR, Jondal DE, Butters KA, Knudsen BE, Anderson JL, et al. Heat stress-induced PI3K/mTORC2-dependent AKT signaling is a central mediator of hepatocellular carcinoma survival to thermal ablation induced heat stress. PLoS ONE. 2016;11:e0162634.
Wang C, Zhang Y, Guo K, Wang N, Jin H, Liu Y, et al. Heat shock proteins in hepatocellular carcinoma: molecular mechanism and therapeutic potential. Int J Cancer. 2016;138:1824–34.
Zavaglia C, Corso R, Rampoldi A, Vinci M, Belli LS, Vangeli M, et al. Is percutaneous radiofrequency thermal ablation of hepatocellular carcinoma a safe procedure? Eur J Gastroenterol Hepatol. 2008;20:196–201.
Takada Y, Kurata M, Ohkohchi N. Rapid and aggressive recurrence accompanied by portal tumor thrombus after radiofrequency ablation for hepatocellular carcinoma. Int J Clin Oncol. 2003;8:332–5.
Ruzzenente A, Manzoni GD, Molfetta M, Pachera S, Genco B, Donataccio M, et al. Rapid progression of hepatocellular carcinoma after radiofrequency ablation. World J Gastroenterol. 2004;10:1137–40.
Kasugai H, Osaki Y, Oka H, Kudo M, Seki T, Osaka Liver Cancer Study Group. Severe complications of radiofrequency ablation therapy for hepatocellular carcinoma: an analysis of 3,891 ablations in 2,614 patients. Oncology. 2007;72 Suppl 1:72–75.
Sala M, Llovet JM, Vilana R, Bianchi L, Solé M, Ayuso C, et al. Initial response to percutaneous ablation predicts survival in patients with hepatocellular carcinoma. Hepatology. 2004;40:1352–60.
Home T, Jensen RA, Rao R. Heat shock factor 1 in protein homeostasis and oncogenic signal integration. Cancer Res. 2015;75:907–12.
Longshaw VM, Chapple JP, Balda MS, Cheetham ME, Blatch GL, et al. Nuclear translocation of the Hsp70/Hsp90 organizing protein mSTI1 is regulated by cell cycle kinases. J Cell Sci. 2004;117:701–10.
Daniel S, Bradley G, Longshaw VM, Söti C, Csermely P, Blatch GL, et al. Nuclear translocation of the phosphoprotein Hop (Hsp70/Hsp90 organizing protein) occurs under heat shock, and its proposed nuclear localization signal is involved in Hsp90 binding. Biochim Biophys Acta. 2008;1783:1003–14.
Lam VW, Ng KK, Chok KS, Cheung TT, Yuen J, Tung H, et al. Risk factors and prognostic factors of local recurrence after radiofrequency ablation of hepatocellular carcinoma. J Am Coll Surg. 2008;207:20–29.
Kang TW, Lim HK, Cha DI. Aggressive tumor recurrence after radiofrequency ablation for hepatocellular carcinoma. Clin Mol Hepatol. 2017;23:95–101.
Kim KW, Lee JM, Klotz E, Kim SJ, Kim SH, Kim JY, et al. Safety margin assessment after radiofrequency ablation of the liver using registration of preprocedure and postprocedure CT images. Am J Roentgenol. 2011;196:W565–W572.
Dong S, Kong J, Kong F, Kong J, Gao J, Ji L, et al. Sorafenib suppresses the epithelial-mesenchymal transition of hepatocellular carcinoma cells after insufcient radiofrequency ablation. BMC Cancer. 2015;15:939.
Xu M, Xie XH, Xie XY, Xu ZF, Liu GJ, Zheng YL, et al. Sorafenib suppresses the rapid progress of hepatocellular carcinoma after insufcient radiofrequency ablation therapy: an experiment in vivo. Acta Radiol. 2013;54:199–204.
Chen Z, Xu L, Su T, Ke Z, Peng Z, Zhang N, et al. Autocrine STIP1 signaling promotes tumor growth and is associated with disease outcome in hepatocellular carcinoma. Biochem Biophys Res Commun. 2017;493:365–72.
Schulz R, Streller F, Scheel AH, Rüschoff J, Reinert MC, Dobbelstein M, et al. HER2/ErbB2 activates HSF1 and thereby controls HSP90 clients including MIF in HER2-overexpressing breast cancer. Cell Death Dis. 2014;5:e980.
Smyth T, Paraiso KH, Hearn K, Rodriguez-Lopez AM, Munck JM, Haarberg HE, et al. Inhibition of HSP90 by AT13387 delays the emergence of resistance to BRAF inhibitors and overcomes resistance to dual BRAF and MEK inhibition in melanoma models. Mol Cancer Ther. 2014;13:2793–804.
Savagner P. Leaving the neighborhood: molecular mechanisms involved during epithelial-mesenchymal transition. Bioessays. 2001;23:912–23.
Lei W, Mullen N, McCarthy S, Brann C, Richard P, Cormier J, et al. Heat-shock protein 90 (Hsp90) promotes opioid-induced anti-nociception by an ERK mitogen-activated protein kinase (MAPK) mechanism in mouse brain. J Biol Chem. 2017;292:10414–28.
Kumalo HM, Bhakat S, Soliman ME. Heat-shock protein 90 (Hsp90) as anticancer target for drug discovery: an ample computational perspective. Chem Biol Drug Des. 2015;86:1131–60.
Ernst A, Anders H, Kapfhammer H, Orth M, Hennel R, Seidl K, et al. HSP90 inhibition as a means of radiosensitizing resistant, aggressive soft tissue sarcomas. Cancer Lett. 2015;365:211–22.
Sadikot T, Swink M, Eskew JD, Brown D, Zhao H, Kusuma BR, et al. Development of a high-throughput screening cancer cell-based luciferase refolding assay for identifying Hsp90 inhibitors. Assay Drug Dev Technol. 2013;11:478–88.
Chang CH, Drechsel DA, Kitson RR, Siegel D, You Q, Backos DS, et al. 19-Substituted benzoquinone ansamycin heat shock protein-90 inhibitors: biological activity and decreased off-target toxicity. Mol Pharmacol. 2014;85:849–57.
Wang TH, Chao A, Tsai CL, Chang CL, Chen SH, Lee YS, et al. Stress-induced phosphoprotein 1 as a secreted biomarker for human ovarian cancer promotes cancer cell proliferation. Mol Cell Proteom. 2010;9:1873–84.
Walsh N, Clynes M, Crown J, O’Donovan N. Alterations in integrin expression modulates invasion of pancreatic cancer cells. J Exp Clin Cancer Res. 2009;28:140.
Pimienta G, Herbert KM, Regan L. A compound that inhibits the HOP-Hsp90 complex formation and has unique killing effects in breast cancer cell lines. Mol Pharm. 2011;8:2252–61.
Zhang W, Luo E, Gan J, Song X, Bao Z, Zhang H, et al. Long-term survival of hepatocellular carcinoma after percutaneous radiofrequency ablation guided by ultrasound. World J Surg Oncol. 2017;15:122.
Li Y, Wang K, Chen L, Zhu X, Zhou J. Quantification of mRNA levels using real-time polymerase chain reaction (PCR). Methods Mol Biol. 2016;1406:73–79.
Cikos S, Bukovská A, Koppel J. Relative quantification of mRNA: comparison of methods currently used for real-time PCR data analysis. BMC Mol Biol. 2007;8:113.
Acknowledgements
We wish to appreciate Dr. Bing Liao and Dr. Zhengkai Cao for reviewing our immunohistochemical staining data.
Author contributions
Conception and design: SP, MK. Development of methodology: SP, MK. Acquisition of data: SP, TS, JL, YW, ZD. Analysis and interpretation of data: SP, TS, JL, MK. Writing, review and/or revision of the manuscript: SP, QZ, MK. Administrative, technical, or material support: LX, SC, ZP. Study supervision: MK.
Funding
This work was supported by the National Natural Science Foundation of China (81470817, 81771958), the Guangdong Science and Technology Plan (2016A020215189, 2014A020212396), the National Natural Science Foundation of Guangdong (2015A030311039) and the Guangzhou Science and Technology Plan (201704020099).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
These authors contributed equally: Tianhong Su, Junbin Liao, Zihao Dai.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Su, T., Liao, J., Dai, Z. et al. Stress-induced phosphoprotein 1 mediates hepatocellular carcinoma metastasis after insufficient radiofrequency ablation. Oncogene 37, 3514–3527 (2018). https://doi.org/10.1038/s41388-018-0169-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0169-4
This article is cited by
-
Autophagy-amplifying nanoparticles evoke immunogenic cell death combined with anti-PD-1/PD-L1 for residual tumors immunotherapy after RFA
Journal of Nanobiotechnology (2023)
-
Mitochondrial ROS driven by NOX4 upregulation promotes hepatocellular carcinoma cell survival after incomplete radiofrequency ablation by inducing of mitophagy via Nrf2/PINK1
Journal of Translational Medicine (2023)
-
Augmented EPR effect post IRFA to enhance the therapeutic efficacy of arsenic loaded ZIF-8 nanoparticles on residual HCC progression
Journal of Nanobiotechnology (2022)
-
Insufficient ablation induces E3-ligase Nedd4 to promote hepatocellular carcinoma progression by tuning TGF-β signaling
Oncogene (2022)
-
The Hsp70–Hsp90 go-between Hop/Stip1/Sti1 is a proteostatic switch and may be a drug target in cancer and neurodegeneration
Cellular and Molecular Life Sciences (2021)