Abstract
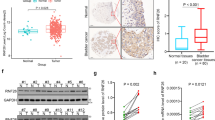
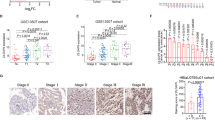
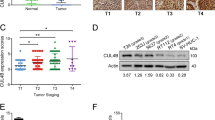
The transcription factor MYB proto-oncogene like 2 (MYBL2) is critical in regulating gene expression and tumorigenesis. However, the biological function of MYBL2 in bladder cancer (BLCA) remains to be elucidated. Here, we first revealed that MYBL2 was elevated in BLCA tissues and significantly correlated with clinicopathological parameters and cancer-specific survival in BLCA patients. Phenotypic assays showed that MYBL2 deficiency suppressed the proliferation and migration of BLCA cells in vitro and in vivo, whereas MYBL2 overexpression contributed to the opposite phenotype. Mechanistically, MYBL2 could bind to the promoter of its downstream target gene cell division cycle-associated protein 3 (CDCA3) and transactivate it, which in turn promoted the malignant phenotype of BLCA cells. Further investigations revealed that MYBL2 interacted with forkhead box M1 (FOXM1) to co-regulate the transcription of CDCA3. In addition, MYBL2/FOXM1 and CDCA3 might activate Wnt/β-catenin signaling, thereby promoting the malignant phenotype of BLCA cells. In conclusion, the current study identifies MYBL2 as an oncogene in BLCA. MYBL2 can accelerate the proliferation and metastasis of BLCA through the transactivation of CDCA3.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The microarray data could be downloaded from the NCBI Gene Expression Omnibus (GEO) database with accession number GSE13507, GSM1526875, GSM2797580, and GSE119971. Other bioinformatics analyses could be performed using the online databases: UALCAN, GEPIA, and JASPAR. Raw RNA sequencing data generated in this study have been submitted to the GEO database under accession number GSE201875 and GSE201979.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Rimar KJ, Tran PT, Matulewicz RS, Hussain M, Meeks JJ. The emerging role of homologous recombination repair and PARP inhibitors in genitourinary malignancies. Cancer. 2017;123:1912–24.
Pan CW, Liu H, Zhao Y, Qian C, Wang L, Qi J. JNK2 downregulation promotes tumorigenesis and chemoresistance by decreasing p53 stability in bladder cancer. Oncotarget. 2016;7:35119–31.
Shen D, Fang Y, Zhou F, Deng Z, Qian K, Wang G, et al. The inhibitory effect of silencing CDCA3 on migration and proliferation in bladder urothelial carcinoma. Cancer Cell Int. 2021;21:257.
Xiong YC, Wang J, Cheng Y, Zhang XY, Ye XQ. Overexpression of MYBL2 promotes proliferation and migration of non-small-cell lung cancer via upregulating NCAPH. Mol Cell Biochem. 2020;468:185–93.
Cicirò Y, Sala A. MYB oncoproteins: emerging players and potential therapeutic targets in human cancer. Oncogenesis. 2021;10:19.
Bayley R, Ward C, Garcia P. MYBL2 amplification in breast cancer: molecular mechanisms and therapeutic potential. Biochim Biophys Acta Rev Cancer. 2020;1874:188407.
Li Q, Wang M, Hu Y, Zhao E, Li J, Ren L, et al. MYBL2 disrupts the Hippo-YAP pathway and confers castration resistance and metastatic potential in prostate cancer. Theranostics. 2021;11:5794–812.
Hou X, Zhang Y, Han S, Hou B. A novel DNA methylation 10-CpG prognostic signature of disease-free survival reveal that MYBL2 is associated with high risk in prostate cancer. Expert Rev Anticancer Ther. 2020;20:1107–19.
Liu Q, Guo L, Qi H, Lou M, Wang R, Hai B, et al. A MYBL2 complex for RRM2 transactivation and the synthetic effect of MYBL2 knockdown with WEE1 inhibition against colorectal cancer. Cell Death Dis. 2021;12:683.
Yu R, Li C, Lin X, Chen Q, Li J, Song L, et al. Clinicopathologic features and prognostic implications of MYBL2 protein expression in pancreatic ductal adenocarcinoma. Pathol Res Pr. 2017;213:964–8.
Okumura F, Uematsu K, Byrne SD, Hirano M, Joo-Okumura A, Nishikimi A, et al. Parallel regulation of von Hippel-Lindau disease by pVHL-mediated degradation of B-Myb and hypoxia-inducible factor α. Mol Cell Biol. 2016;36:1803–17.
Heinrichs S, Conover LF, Bueso-Ramos CE, Kilpivaara O, Stevenson K, Neuberg D, et al. MYBL2 is a sub-haploinsufficient tumor suppressor gene in myeloid malignancy. Elife. 2013;2:e00825.
Han J, Xie R, Yang Y, Chen D, Liu L, Wu J, et al. CENPA is one of the potential key genes associated with the proliferation and prognosis of ovarian cancer based on integrated bioinformatics analysis and regulated by MYBL2. Transl Cancer Res. 2021;10:4076–86.
Sadasivam S, Duan S, DeCaprio JA. The MuvB complex sequentially recruits B-Myb and FoxM1 to promote mitotic gene expression. Genes Dev. 2012;26:474–89.
Wang Z, Ahmad A, Li Y, Banerjee S, Kong D, Sarkar FH. Forkhead box M1 transcription factor: a novel target for cancer therapy. Cancer Treat Rev. 2010;36:151–6.
Xue YJ, Xiao RH, Long DZ, Zou XF, Wang XN, Zhang GX, et al. Overexpression of FoxM1 is associated with tumor progression in patients with clear cell renal cell carcinoma. J Transl Med. 2012;10:200.
Engeland K. Cell cycle arrest through indirect transcriptional repression by p53: I have a DREAM. Cell Death Differ. 2018;25:114–32.
Fischer M, Grossmann P, Padi M, DeCaprio JA. Integration of TP53, DREAM, MMB-FOXM1 and RB-E2F target gene analyses identifies cell cycle gene regulatory networks. Nucleic Acids Res. 2016;44:6070–86.
Xie H, Miao N, Xu D, Zhou Z, Ni J, Yin F, et al. FoxM1 promotes Wnt/β-catenin pathway activation and renal fibrosis via transcriptionally regulating multi-Wnts expressions. J Cell Mol Med. 2021;25:1958–71.
Gong A, Huang S. FoxM1 and Wnt/β-catenin signaling in glioma stem cells. Cancer Res. 2012;72:5658–62.
Sheng Y, Yu C, Liu Y, Hu C, Ma R, Lu X, et al. FOXM1 regulates leukemia stem cell quiescence and survival in MLL-rearranged AML. Nat Commun. 2020;11:928.
Li M, Liu Y, Liu J, Li W, Li N, Xue D, et al. Circ_0006332 promotes growth and progression of bladder cancer by modulating MYBL2 expression via miR-143. Aging (Albany NY). 2019;11:10626–43.
Zhang X, Lv QL, Huang YT, Zhang LH, Zhou HH. Akt/FoxM1 signaling pathway-mediated upregulation of MYBL2 promotes progression of human glioma. J Exp Clin Cancer Res. 2017;36:105.
Ayad NG, Rankin S, Murakami M, Jebanathirajah J, Gygi S, Kirschner MW. Tome-1, a trigger of mitotic entry, is degraded during G1 via the APC. Cell. 2003;113:101–13.
Iness AN, Felthousen J, Ananthapadmanabhan V, Sesay F, Saini S, Guiley KZ, et al. The cell cycle regulatory DREAM complex is disrupted by high expression of oncogenic B-Myb. Oncogene. 2019;38:1080–92.
Guiley KZ, Iness AN, Saini S, Tripathi S, Lipsick JS, Litovchick L, et al. Structural mechanism of Myb-MuvB assembly. Proc Natl Acad Sci USA. 2018;115:10016–21.
Salik B, Yi H, Hassan N, Santiappillai N, Vick B, Connerty P, et al. Targeting RSPO3-LGR4 signaling for leukemia stem cell eradication in acute myeloid leukemia. Cancer Cell. 2020;38:263.
Choi BR, Cave C, Na CH, Sockanathan S. GDE2-dependent activation of canonical Wnt signaling in neurons regulates oligodendrocyte maturation. Cell Rep. 2020;31:107540.
He S, Tang S. WNT/β-catenin signaling in the development of liver cancers. Biomed Pharmacother. 2020;132:110851.
Bian J, Dannappel M, Wan C, Firestein R. Transcriptional regulation of Wnt/β-catenin pathway in colorectal cancer. Cells. 2020;9:2125.
Ge X, Wang X. Role of Wnt canonical pathway in hematological malignancies. J Hematol Oncol. 2010;3:33.
Ramirez JG, Smit DJ, Viol F, Schrader J, Ghadban T, Pantel K, et al. High serum levels of Wnt signaling antagonist dickkopf-related protein 1 are associated with impaired overall survival and recurrence in esophageal cancer patients. Cancers (Basel). 2021;13:4980.
Cao R, Wang G, Qian K, Chen L, Ju L, Qian G, et al. TM4SF1 regulates apoptosis, cell cycle and ROS metabolism via the PPARγ-SIRT1 feedback loop in human bladder cancer cells. Cancer Lett. 2018;414:278–93.
Chen G, Deng X. Cell synchronization by double thymidine block. Bio Protoc. 2018;8:e2994.
Association WM. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310:2191–4.
Acknowledgements
The excellent technical assistance of Ms. Yayun Fang and Ms. Danni Shan are gratefully acknowledged. Part of the analysis was performed on the High Performance Computing Platform of the Center for Life Science (Peking University). The study was funded by National Natural Science Foundation of China (82172985 and 81772730), Improvement Project for Theranostic Ability on Difficulty Miscellaneous Disease (Tumor) from National Health Commission of China (ZLYNXM202006), Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2020-PT320-004), Research Fund of Zhongnan Hospital of Wuhan University (ZNJC201915, SWYBK00-03, 413100049, and PTXM2020024), Science and Technology Department of Hubei Province Key Project (2022EJD001 and YYXKNL2022001).
Author information
Authors and Affiliations
Contributions
W.L., X.W., and Y.X. devised the study and wrote the manuscript. W.L., D.S., L.J., and R.Z. performed most experiments. G.W., K.Q., W.D., and W.J. helped with data collection and assembly. K.X. and Y.Z. performed data analysis and interpretation. X.W. and Y.X. corrected the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was performed in accordance with the Declaration of Helsinki [36] and was approved by the Institutional Ethics Committee of Zhongnan Hospital of Wuhan University (approval number: 2020102) and Experimental Animal Welfare and Ethics of Zhongnan Hospital (approval number: ZN2021065). Bioinformatics data involving humans were collected from publicly available databases with anonymous patient information.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, W., Shen, D., Ju, L. et al. MYBL2 promotes proliferation and metastasis of bladder cancer through transactivation of CDCA3. Oncogene 41, 4606–4617 (2022). https://doi.org/10.1038/s41388-022-02456-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-022-02456-x
This article is cited by
-
USP43 stabilizes c-Myc to promote glycolysis and metastasis in bladder cancer
Cell Death & Disease (2024)
-
DLGAP5 triggers proliferation and metastasis of bladder cancer by stabilizing E2F1 via USP11
Oncogene (2024)
-
From modulation of cellular plasticity to potentiation of therapeutic resistance: new and emerging roles of MYB transcription factors in human malignancies
Cancer and Metastasis Reviews (2024)
-
Molecular profile of metastasis, cell plasticity and EMT in pancreatic cancer: a pre-clinical connection to aggressiveness and drug resistance
Cancer and Metastasis Reviews (2024)
-
PLAGL2 promotes bladder cancer progression via RACGAP1/RhoA GTPase/YAP1 signaling
Cell Death & Disease (2023)